Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Sinotech Import&Export Corporation
Properties: CAS No.: 109-67-1 UN No.: 1108 Molecular formula: C5H10 GB number: 31006 Product name: 1-pentene Alias: 1-Pentylene; HSDB 1086; Propylethylene; alpha-Amylene; alpha-n-Amylene; pent-1-ene; 2-methylbut-2-ene
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:109-67-1
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Henan Tianfu Chemical Co., Ltd.
1.Our services:A.Supply sampleB.The packing also can be according the customers` requirmentC.Any inquiries will be replied within 24 hoursD.we provide Commerical Invoice, Packing List, Bill of loading, COA , Health certificate and Origin certificate.
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:109-67-1
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:109-67-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryChemsigma International Co.,Ltd.
bulk?production Application:Pharmaceutical intermediates
HENAN SUNLAKE ENTERPRISE CORPORATION
Henan Sunlake Enterprise Corporation Our Advantages 1, Any inquiry about ch
Hebei Ruishun Trade Co.,Ltd
Hebei ruishun trade co.,ltd, registered capital one million,have a production of pharmaceutical raw materials, pharmaceutical raw materials factory reagent r&d center,seek development by credit reputation.our products have a large price advanta
Qingdao Sigma Chemical Co., Ltd.
Qingdao Sigma Chemical Ltd is is a global chemical industry manufacturers and suppliers of pharmaceuticals and intermediates, peptide,Nootropis etc API, food and feed additives, herbal extracts, agrochemicals and fine chemicals etc. Our Labo
Hangzhou Share Chemical Co., Ltd
At Share Chemical Company, we scrupulously abide by our policy of “Excellent Quality at a Reasonable Price”. We strive to satisfy all of our customers by providing the finest quality products supported by the finest in customer servi
Shanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Chungking Joyinchem Co., Ltd.
Joyinchem have been committed to chemical supply for several years and have built good cooperation records with multinational chemical corporations and importers from all over the world. Our services include:-Spot goods-Contract manufacturing-Custom
Shaanxi Cuicheng Biomedical Technology Co., Ltd.
Shaanxi Cuicheng Biomedical Technology Co., Ltd. is located in Chuangzhi Industrial Park, Economic and Technological Development Zone, Hanzhong City, Shaanxi Province, China. It is a company specializing in natural medicines and high-throughput scree
Shanghai Run-Biotech Co., Ltd.
Shanghai, Run-Biotech Co., Ltd is a leading domestic pharmaceutical, biopharmaceutical, and health care products R & D outsourcing services company. As an innovation-driven and customer-focused company, Run Biotech provides a broad and integrated por
Nanjing Raymon Biotech Co., Ltd.
1-Pentene [Standard Material for GC]Appearance:Off white to slight yellow solid Storage:Store in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/Pharmaceutical intermediates Transportation:By Sea,by
Cas:109-67-1
Min.Order:0
Negotiable
Type:Trading Company
inquiryLangchem
1.We are specializing in providing customers with complete chemical raw materials.2.Superior quality, moderate price & quick delivery.3. Extensive international cooperation experience.4. Outstanding communication skills. Application:contact us for de
Changzhou Foreign Trade Corp
CFTC?PharmaChem?is?a?service?based?company?in?China,?a?pharmachem?division?of?Changzhou?Foreign?Trade?Corp.,supplying?raw?materials,?intermediates,?APIs?and?fine?chemicals?for?the?pharmaceutical?and?specialty?chemical?industries?worldwide. Applicatio
Chemical Co.Ltd
1-Pentene [Standard Material for GC]Appearance:Off white to slight yellow solid Storage:Stored in shaded, cool and dry places Package:1L 5L 10L 25L bottle Application:pharma intermediate Transportation:Handle with cares to avoid damaging the packages
Cas:109-67-1
Min.Order:0
Negotiable
Type:Trading Company
inquirySigma-Aldrich Chemie GmbH
Package:25, 100 mL in glass bottle Application:1-Pentene can be used as a component in gasoline surrogates. It can also be used in the synthesis of (+/-)-byssochlamic acid and rugulactone.
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen In methanol at 20℃; under 760.051 Torr; for 4h; Green chemistry; | 96% |
| With hydrogen In methanol under 760.051 Torr; for 5h; | 96% |
| With piperazine; hydrogen In ethanol at 80℃; under 4500.45 Torr; for 20h; | 88% |
-
-
20449-21-2
2-(but-3-en-1-yl)-2-methyl-1,3-dioxolane

-
-
592-99-4, 7642-15-1, 14850-23-8
4-octene

-
A
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| aluminum oxide; tetramethylstannane; rhenium(VII) oxide In chlorobenzene at 25℃; for 3h; Yields of byproduct given; | A n/a B 88.6% |

-
-
142-62-1
hexanoic acid

-
A
-
124-18-5
decane

-
B
-
109-67-1
1-penten

-
C
-
109-66-0
pentane

-
D
-
109-68-2
2-pentene

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water pH=5.4 - 9.4; Concentration; pH-value; Kolbe Electrolysis; Electrochemical reaction; | A 82% B 44% C 30% D 26% |
| With potassium hydroxide In water pH=5.8 - 9; Kolbe Electrolysis; Electrochemical reaction; | A 39% B 14% C 5% D 7% |


| Conditions | Yield |
|---|---|
| With ethene; Mo(NiPrPh)-(CHCMe2Ph)(Me2Pyrr)(OBitet) In benzene-d6 at 22℃; under 3040.2 Torr; for 0.25h; Inert atmosphere; stereoselective reaction; | A 79% B n/a |
-
-
627-19-0
1-Pentyne

-
A
-
627-20-3
(Z)-pent-2-ene

-
B
-
646-04-8
(E)-pent-2-ene

-
C
-
109-67-1
1-penten

-
D
-
109-66-0
pentane

| Conditions | Yield |
|---|---|
| With hydrogen; palladium dichloride In N,N-dimethyl-formamide under 18751.5 Torr; for 0.3h; Product distribution; Ambient temperature; various time; | A n/a B n/a C 78.5% D 2.4% |
| With [Ru4(μ-H)4(CO)12] In benzene at 25℃; for 23h; Irradiation; | A 10% B 13% C 73% D 4% |
| With [Ru4(μ-H)4(CO)12]; hydrogen In benzene at 25℃; under 517.1 Torr; for 24h; Irradiation; | A 13% B 10% C 66% D 11% |


| Conditions | Yield |
|---|---|
| With carbon monoxide In (2)H8-toluene heated to 150°C for 2.5 h under CO; cooled to 10°C; NMR, GC, GC-MS; | A 78% B 54% C 13% |

-
-
627-21-4
2-Pentyne

-
A
-
627-20-3
(Z)-pent-2-ene

-
B
-
646-04-8
(E)-pent-2-ene

-
C
-
109-67-1
1-penten

-
D
-
109-66-0
pentane

| Conditions | Yield |
|---|---|
| With [Ru4(μ-H)4(CO)12] In benzene at 25℃; for 23h; Irradiation; | A 75% B 1% C 24% D n/a |
| With [Ru4(μ-H)4(CO)12]; hydrogen In benzene at 25℃; under 517.1 Torr; for 24h; Irradiation; | A 19% B 73% C 2% D 6% |
| With [Ru4(μ-H)4(CO)12]; hydrogen In benzene at 25℃; for 24h; Irradiation; | A 19% B 73% C 2% D 6% |
| With hydrogen; dodecacarbonyltetrarhodium(0) at 80℃; under 760 Torr; for 3h; Product distribution; other alkynes, other carbonyl-rhodium catalyst system; | A 64 % Chromat. B 17 % Chromat. C 2 % Chromat. D 12 % Chromat. |

-
-
67969-82-8
tetrafluoroboric acid diethyl ether

-
A
-
12078-28-3, 38979-86-1
dicarbonylcyclopentadienyliodoiron(II)

-
B
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| With carbon monoxide In dichloromethane-d2 (Cp2(CO)3Fe2(CHCHCH2CH2CH3))PF6, LiI, HBF4*Et2O, and CD2Cl2 sealed in NMR tube ubder 1 atm of CO, slow reaction at 35°C monitored by NMR for 10 days; 1-pentene determined by gas chromy., Cp(CO)2FeI isolated by Et2O extn. of residue; | A 48% B 72% |

-
-
110-53-2
1-Bromopentane

-
-
95903-96-1
triethylbenzylammonium ethanolate

-
A
-
17952-11-3
1-ethoxypentane

-
B
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| at 20 - 25℃; for 1h; | A 67% B 6% |


| Conditions | Yield |
|---|---|
| With triethylbenzylammonium ethanolate at 20 - 25℃; for 1h; | A 67% B 6% |
-
-
2004-70-8
1-methylbuta-1,3-diene

-
A
-
627-20-3
(Z)-pent-2-ene

-
B
-
646-04-8
(E)-pent-2-ene

-
C
-
109-67-1
1-penten

-
D
-
109-66-0
pentane

| Conditions | Yield |
|---|---|
| With hydrogen; palladium dichloride In N,N-dimethyl-formamide under 18751.5 Torr; for 0.116667h; Product distribution; Ambient temperature; various time; | A 11.7% B 62.1% C 15.3% D 1.2% |
| With hydrogen; complex 1 under 684 Torr; for 6h; Product distribution; Variation of complex catalysts, time.; | |
| With hydrogen; Pd-containing polymer In methanol at 20℃; Product distribution; catalytic properties of palladium fixed on poly(m- and p-)hydroxyphenylbenzoxazoleterephthalamides, hydrogenation and isomerization of olefins; | |
| With hydrogen; <2> In dichloromethane for 5h; Product distribution; other alkene; |

-
-
628-17-1
amyl iodide

-
A
-
646-04-8
(E)-pent-2-ene

-
B
-
6032-29-7
(+/-)-2-pentanol

-
C
-
584-02-1
2-pentanol

-
D
-
71-41-0
pentan-1-ol

-
E
-
109-67-1
1-penten

-
F
-
109-66-0
pentane

| Conditions | Yield |
|---|---|
| In acetonitrile for 0.1h; Irradiation; | A 22.18% B 5.91% C 9.26% D 3.15% E 53.6% F 1.41% |


| Conditions | Yield |
|---|---|
| In benzene for 18h; Irradiation; | A 49% B n/a |
| In benzene for 18h; Mechanism; Product distribution; Irradiation; oxygen purged,; | A 32% B n/a |
| Irradiation; | A 52 % Chromat. B n/a |

| Conditions | Yield |
|---|---|
| With hydrogen In (2)H8-toluene at 0℃; for 6.41667h; Irradiation; | A 44% B 13% |

| Conditions | Yield |
|---|---|
| With hydrogen In toluene Fe complex in toluene was photolyzed (black light bulb, emission max. 366 nm) at 0°C in an H2 atmosphere for 6 h; monitored by gas chromy. (AgNO3 column), heptane as internal standard; | A 44% B 13% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water pH=5.2 - 7.5; Kolbe Electrolysis; Electrochemical reaction; | A 44% B 30% C 26% |
| With potassium hydroxide In water pH=5 - 7.1; Kolbe Electrolysis; Electrochemical reaction; | A 38% B 28% C 34% |

-
-
627-20-3
(Z)-pent-2-ene

-
A
-
34557-54-5
methane

-
B
-
107-01-7
butene-2

-
C
-
74-84-0
ethane

-
D
-
109-67-1
1-penten

-
E
-
563-45-1
3-Methyl-1-butene

-
F
-
106-99-0
buta-1,3-diene

| Conditions | Yield |
|---|---|
| hydrogen sulfide at 469.9℃; Rate constant; Kinetics; Product distribution; mechanism; effects of temperature, concentration; further products; | A 43.3% B 2.9% C 1.6% D 4.5% E 0.5% F 39.5% |

-
-
78998-23-9
α-keto-octanoic acid 2-(2-naphthyl)ethylester

-
A
-
109-67-1
1-penten

-
B
-
78998-25-1
2-Hydroxy-acrylic acid 2-naphthalen-2-yl-ethyl ester

| Conditions | Yield |
|---|---|
| In benzene Irradiation; | A 40% B n/a |
| In benzene for 18h; Mechanism; Product distribution; Irradiation; oxigen purged; | A 39% B n/a |
| In benzene for 18h; Irradiation; |
-
-
19493-09-5
Methylenetriphenylphosphorane

-
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| for 24h; Ambient temperature; | 40% |
-
-
187737-37-7
propene

-
A
-
763-29-1
2-Methyl-1-pentene

-
B
-
2213-32-3
2,4-dimethyl-1-pentene

-
C
-
691-37-2
4-Methyl-1-pentene

-
D
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| With Al#dotZr; C40H36Cl2Zr In toluene at 80℃; under 4500.45 Torr; for 0.5h; Reagent/catalyst; Temperature; Autoclave; | A 39.9% B 14.9% C 16.3% D 28.9% |
| With Al#dotZr; C40H40Cl2Zr In toluene at 40℃; under 4500.45 Torr; for 0.5h; Autoclave; | A 33.9% B 12.6% C 19.2% D 34.3% |
| With Zr#dotHf; C40H40Cl2Hf In toluene at 80℃; under 4500.45 Torr; for 0.5h; Reagent/catalyst; Temperature; Autoclave; | A 19.7% B 22.9% C 32.8% D 24.6% |

-
-
1574-41-0
Z-piperylene

-
A
-
627-20-3
(Z)-pent-2-ene

-
B
-
646-04-8
(E)-pent-2-ene

-
C
-
109-67-1
1-penten

-
D
-
109-66-0
pentane

| Conditions | Yield |
|---|---|
| With hydrogen; palladium dichloride In N,N-dimethyl-formamide under 18751.5 Torr; for 0.1h; Product distribution; Ambient temperature; various time; | A 26% B 34.2% C 28.7% D 0.8% |
| With hydrogen; complex 1 under 684 Torr; for 24h; Product distribution; Variation of complex catalysts, time, H2 pressure.; | |
| With polyethyleneimine; ruthenium trichloride; hydrogen In ethanol at 50℃; under 3800 Torr; Product distribution; | A 30 % Chromat. B 40 % Chromat. C 13 % Chromat. D 17 % Chromat. |
| With (η5-C5H5)NiOs3(μ-H)3(CO)9; hydrogen In octane at 120℃; under 684 Torr; for 1h; Product distribution; also trans-1,3-pentadiene; also heterogeneous catalysis (gas-chromatographic materials as a support); var. temp. and times; | |
| With hydrogen; (η5-C5H5)NiRu3(μ-H)3(CO)9 In octane for 120h; Product distribution; effect of the reaction time, comparison with the osmium-containing complex; |

-
-
592-99-4, 7642-15-1, 14850-23-8
4-octene

-
-
1576-85-8
4-pentenyl acetate

-
A
-
109-67-1
1-penten

-
B
-
150462-80-9
acetoxy-1 octene-4

-
C
-
76293-63-5
1,8-diacetoxy-4-octene

| Conditions | Yield |
|---|---|
| tetramethylstannane; tungsten(VI) chloride at 70℃; for 16h; | A n/a B 31.8% C 7.9% |

-
-
75-09-2
dichloromethane

-
-
693-04-9
butyl magnesium bromide

-
A
-
111-84-2
nonane

-
B
-
110-54-3
hexane

-
C
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| With C31H37ClN3NiO2(1-)*Li(1+) In tetrahydrofuran at 25℃; for 0.333333h; Concentration; Temperature; Inert atmosphere; Overall yield = 64 %; | A 20% B 14% C 29.9% |
| With C31H37ClFeN3O2 In tetrahydrofuran at 25℃; for 0.0833333h; Temperature; Concentration; Inert atmosphere; Overall yield = 100 %; |

-
-
115-11-7
isobutene

-
A
-
106-98-9
1-butylene

-
B
-
590-18-1
(Z)-2-Butene

-
C
-
513-35-9
2-methyl-but-2-ene

-
D
-
1574-41-0
Z-piperylene

-
E
-
2004-70-8
1-methylbuta-1,3-diene

-
F
-
187737-37-7
propene

-
G
-
34557-54-5
methane

-
H
-
624-64-6
trans-2-Butene

-
I
-
627-20-3
(Z)-pent-2-ene

-
J
-
646-04-8
(E)-pent-2-ene

-
K
-
74-84-0
ethane

-
L
-
74-98-6
propane

-
M
-
75-28-5
Isobutane

-
N
-
78-78-4
methylbutane

-
O
-
74-85-1
ethene

-
P
-
109-67-1
1-penten

-
Q
-
287-92-3
Cyclopentane

-
R
-
563-46-2
2-Methyl-1-butene

-
S
-
563-45-1
3-Methyl-1-butene

-
T
-
142-29-0
cyclopentene

-
U
-
106-97-8
n-butane

-
V
-
109-66-0
pentane

| Conditions | Yield |
|---|---|
| CBV1502 at 579.84℃; under 900.09 Torr; Product distribution / selectivity; | A 2.6% B 2.4% C 1.29% D 0.05% E 0.03% F 24.95% G 0.73% H 3.19% I 0.32% J 0.58% K 0.36% L 2.08% M 2.15% N 0.34% O 9.61% P 0.23% Q 0.4% R 0.71% S 0.14% T 0.14% U 1.8% V 0.16% |
| CBV28014 at 509.84℃; under 900.09 Torr; Product distribution / selectivity; | A 6.71% B 7.3% C 5.62% D 0.02% E 0.03% F 23.29% G 0.09% H 9.97% I 1.1% J 2.06% K 0.07% L 1.24% M 1.95% N 0.59% O 3.25% P 0.7% Q 0.31% R 2.72% S 0.47% T 0.21% U 1.37% V 0.26% |

-
-
187737-37-7
propene

-
A
-
591-93-5
1,4-Pentadiene

-
B
-
34557-54-5
methane

-
C
-
74-98-6
propane

-
D
-
109-67-1
1-penten

-
E
-
563-45-1
3-Methyl-1-butene

-
F
-
75-19-4
cyclopropane

| Conditions | Yield |
|---|---|
| at -78.1℃; Product distribution; excited by the impact of low-energy electrons; | A 0.12% B 0.57% C 0.99% D 0.094% E 0.032% F 0.21% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride In tetrahydrofuran for 1h; | A 0.25% B 0.447% C 0.084% |
| With hydrogenchloride In diethyl ether at 0℃; | A 0.006% B 0.08% C 0.013% |
| With hydrogenchloride In tetrahydrofuran for 1h; | A 0.25% B 0.447% C 0.084 mmol |


-
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| With synthetic air at 750℃; Formation of xenobiotics; | 0.001% |
| at 750℃; Formation of xenobiotics; | 0.003% |

| Conditions | Yield |
|---|---|
| at -25℃; |

-
-
513-35-9
2-methyl-but-2-ene

-
A
-
109-67-1
1-penten

-
B
-
563-46-2
2-Methyl-1-butene

-
C
-
563-45-1
3-Methyl-1-butene

-
D
-
109-68-2
2-pentene

| Conditions | Yield |
|---|---|
| at 200 - 380℃; Gleichgewichtsbestimmungen bei der Umlagerung ueber Silicagel; |


| Conditions | Yield |
|---|---|
| With H3Ni4(C5H5)4; hydrogen at 40℃; under 760 Torr; other olefins; var. times; | 100% |
| With hydrogen In N,N-dimethyl-formamide at 25℃; under 760.051 Torr; for 10h; | 87% |
| With C14H15NZr In toluene at 25℃; Reagent/catalyst; | 11% |

| Conditions | Yield |
|---|---|
| With iodine In water; ethyl acetate at 20℃; for 1.33333h; iodosulfonization; | 100% |
| With iodine In water; ethyl acetate for 2h; Ambient temperature; |

| Conditions | Yield |
|---|---|
| With 1,2,3-trimethoxybenzene; Grubbs catalyst first generation In dichloromethane for 6h; Heating / reflux; | 100% |

| Conditions | Yield |
|---|---|
| Irradiation (UV/VIS); UV-irradiation at 25-50 °C for 18 h;; | 100% |
| Irradiation (UV/VIS); UV-irradiation at 25-50 °C for 18 h;; | 100% |
-
-
109-67-1
1-penten

-
-
154013-02-2
6,6-dimethyl-5-oxo-5,6,7,8-tetrahydronaphthalen-2-yl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| Stage #1: 1-penten With 9-bora-bicyclo[3.3.1]nonane In tetrahydrofuran at 20℃; Stage #2: 6,6-dimethyl-5-oxo-5,6,7,8-tetrahydronaphthalen-2-yl trifluoromethanesulfonate With potassium phosphate; tetrakis(triphenylphosphine) palladium(0); potassium bromide In tetrahydrofuran; water at 68℃; for 2h; | 100% |

| Conditions | Yield |
|---|---|
| With Wilkinson's catalyst at 60℃; for 6h; | 99.4% |

| Conditions | Yield |
|---|---|
| With peracetic acid; C80H84Mn2N8O4*4ClO4(1-) In acetonitrile at 0℃; for 0.5h; | 99% |
| With tert.-butylhydroperoxide; 2C13H10N3O2(1-)*MoO2(2+) In methanol; dichloromethane for 1h; Catalytic behavior; Reagent/catalyst; | 87% |
| With tert.-butylhydroperoxide; [dioxidomolybdenum(VI)(2-bromo-N'-(2-hydroxo-3-methoxybenzylidene)benzohydrazone)(H2O)] In methanol; dichloromethane at 79.84℃; for 1h; Catalytic behavior; Reagent/catalyst; Reflux; | 84.2% |

| Conditions | Yield |
|---|---|
| With oxygen; Bi-Mo oxide (1/1) at 400℃; Rate constant; Kinetics; also without O2; other temperature; | 99% |
| With multi-component bismuth molybdate at 320℃; |

| Conditions | Yield |
|---|---|
| With 3,3',5,5'-tetramethylbiphenyl-2,2',6,6'-tetrakis(dipyrrolylphosphinite); dicarbonyl(acetylacotonato)rhodium(I); hydrogen In isopropyl alcohol; toluene at 125℃; for 8h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; Glovebox; Green chemistry; regioselective reaction; | A 98.6% B n/a |
| With dodecacarbonyl-triangulo-triruthenium; 2-(dicyclohexylphosphanyl)-1-(2-methoxyphenyl)-1H-imidazole; hydrogen In methanol; toluene at 130℃; under 45004.5 Torr; for 20h; Autoclave; regioselective reaction; | A 80% B n/a |
| With Rh(IMes)(cod)Cl; hydrogen In tetrahydrofuran at 95℃; under 45003.6 Torr; for 12h; Product distribution; Further Variations:; Solvents; Temperatures; Pressures; |

-
-
109-67-1
1-penten

-
-
38857-88-4
bis(2,2,2-trichloroethyl)azodicarboxylate

| Conditions | Yield |
|---|---|
| With copper(II) bis(trifluoromethanesulfonate) In dichloromethane at 20℃; for 20h; | 98% |

-
-
109-67-1
1-penten

-
-
1354795-35-9
[(2,2'-bis(di-tert-butylphosphino)biphenyl)(AuCl)2]

| Conditions | Yield |
|---|---|
| In dichloromethane treatment mixt. of gold compd. and silver compd. with pentene in CH2Cl2,stirring for 2 h at room temp. in dark; filtration through celite, evapn., elem. anal.; | 98% |

-
-
136918-14-4
phthalimide

-
-
109-67-1
1-penten

-
-
1074-82-4
potassium phtalimide

-
-
71510-39-9
2-pentyl-1H-isoindole-1,3(2H)-dione

| Conditions | Yield |
|---|---|
| In toluene at 20 - 120℃; for 4h; Temperature; Solvent; Darkness; Cooling with ice; Irradiation; | 97.85% |


| Conditions | Yield |
|---|---|
| With Sodium thiosulfate pentahydrate; 2,2'-azobis-(2,4-dimethylvaleronitrile) In water at 100℃; for 3h; Autoclave; | 97.4% |
-
-
109-67-1
1-penten

-
-
79409-46-4
Methyl (2S,3R,4S)-4-(1,3-Dioxolan-2-ylmethyl)-3,4-dihydro-2-(2,3,4,6-tetraacetyl-β-D-glucopyranosyloxy)-3-vinyl-2H-pyran-5-carboxylate

-
-
881198-83-0
(4S,5R,6S)-4-[1,3]Dioxolan-2-ylmethyl-5-((E)-pent-1-enyl)-6-((2S,3R,4S,5R,6R)-3,4,5-triacetoxy-6-acetoxymethyl-tetrahydro-pyran-2-yloxy)-5,6-dihydro-4H-pyran-3-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| tricyclohexylphosphine[1,3-bis(2,4,6-trimethylphenyl)-4,5-dihydroimidazol-2-ylidine][benzylidene]ruthenium(II) dichloride In toluene at 100℃; | 97% |
-
-
932735-47-2
methyl 8-(2-iodo-5-oxocyclopent-1-enyl)octanoate

-
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| With triethylamine; triphenylphosphine; palladium diacetate at 40℃; for 96h; Heck reaction; | 97% |
-
-
126816-56-6
(+/-)-trans-(η2-ethene)dichloro(4-decyloxy-4'-stilbazole)platinum(II)

-
-
109-67-1
1-penten

-
-
126808-23-9
(+/-)-trans-(η2-pent-1-ene)dichloro(4-decyloxy-4'-stilbazole)platinum(II)

| Conditions | Yield |
|---|---|
| In diethyl ether suspn. of Pt-complex in Et2O, addn. of olefine, stirring for 15 min; filtration, addn. of pentane, slow removal of solvent; elem. anal.; | 97% |

-
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| In chlorobenzene addn. of 1-pentene to freshly prepd. Re-compd. with stirring (after 15 min, -45°C), in C6H5Cl, removal of cold bath, stirring at 100°C for 12 h; dropwise addn. of the soln. to hexane, collection of ppt. by filtn., washing with pentane, drying under oil pump vac.; ratio RS,SR/RR,SS >99:<1; elem. anal.; | 97% |
| In chlorobenzene addn. of 1-pentene to freshly prepd. Re-compd. with stirring (after 15 min, -45°C), in C6H5Cl, removal of cold bath, react. for 12 h; dropwise addn. of the soln. to hexane, collection of ppt. by filtn., washing with pentane, drying under oil pump vac.; ratio RS,SR/RR,SS 64:36; | 87% |
| In chlorobenzene addn. of 1-pentene to freshly prepd. Re-compd. in an NMR tube (after 15 min, -45°C), in C6H5Cl, shaking, transferring quickly to a -45°C probe; after 6 h addn. of the soln. to cold pentane (-80°C), collection of ppt. by filtn., drying under oil pump vac.; ratio RS,SR/RR,SS 39:61; | 81% |

| Conditions | Yield |
|---|---|
| With C21H35N3PRu*F6P(1-) In acetone at 25℃; for 0.25h; | 95% |
| beim Behandeln mit Alkyl- oder Aryl-natrium-Verbindungen; | |
| Isomerisierung bei Einwirkung verschiedener Alkyl- und Aryl-natrium-Verbindungen; |
-
-
109-67-1
1-penten

-
-
61485-47-0
Tetrathiooxalsaeure-dimethylester

-
-
76503-70-3
5,6-Dihydro-2,3-bis(methylthio)-5-propyl-1,4-dithiin

| Conditions | Yield |
|---|---|
| for 48h; Ambient temperature; | 95% |
-
-
109-67-1
1-penten

-
-
80594-34-9
1-Oxo-1-(trimethylsilyl)-2-propanone

| Conditions | Yield |
|---|---|
| In Cyclopentane Irradiation; | 95% |
-
-
67969-82-8
tetrafluoroboric acid diethyl ether

-
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| With C6H5Cl In chlorobenzene addn. of HBF4*Et2O to mixt. of the rhenium complex and C6H5Cl (under N2, -45°C, with stirring, 15 min), addn. of pent-1-ene, after 30 min cold bath removed, stirred (20 h); mixt. filtered into hexane, ppt. collected, washed with pentane, dried in vac.; 2 diastereomers: (RS,SR)/(RR,SS) = 67:33; | 95% |
| With CH2Cl2 In dichloromethane addn. of HBF4*Et2O to mixt. of the rhenium complex and CH2Cl2 (under N2, -80°C), addn. of pent-1-ene, after 30 min cold bath removed, stirred; mixt. filtered into hexane, ppt. collected, washed with pentane, dried in vac.; 2 diastereomers: (RS,SR)/(RR,SS) = 63:37; | 94% |
| In dichloromethane cooling of soln. of Re-compd. (-78°C), addn. of HBF4*Et2O, stirring (0.5 h), addn. of 1-penten, stirring (-78°C, 1 h), warming (room temp.), stirring (2 h); removal of solvent (vac.), extn. (THF), addn. of hexene, filtn., drying (vac.), mixt of diastereomers not sepd.; elem. anal.; | 91% |


| Conditions | Yield |
|---|---|
| Stage #1: diphenyldisulfane With triphenylphosphine Heating; Stage #2: 1-penten With tetrakis(triphenylphosphine) palladium(0) at 100℃; | 95% |
-
-
353261-89-9
2-(2-oxoquinoxaline-1(2H)-yl)acetic acid methyl ester

-
-
109-67-1
1-penten

| Conditions | Yield |
|---|---|
| With trimethylsilylazide; bis-[(trifluoroacetoxy)iodo]benzene In dichloromethane; 1,2-dichloro-ethane at 23℃; for 0.0166667h; | 95% |

| Conditions | Yield |
|---|---|
| With trimethylsilylazide; 9-(2-mesityl)-10-methylacridinium perchlorate In dichloromethane at 20℃; for 0.25h; Catalytic behavior; Solvent; Reagent/catalyst; Irradiation; | 95% |
| With trimethylsilylazide; bis-[(trifluoroacetoxy)iodo]benzene In 1,2-dichloro-ethane at 20℃; for 0.0166667h; Mechanism; Solvent; Reagent/catalyst; | 87% |
-
-
109-67-1
1-penten

-
-
1423875-77-7
(2,6-bis(4,4-dimethyloxazolinyl)-3,5-dimethylphenyl)Ir(acetate)(H)

| Conditions | Yield |
|---|---|
| With sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate In 1,4-dioxane; benzene-d6 at 20℃; under 760.051 Torr; for 1h; Inert atmosphere; Schlenk technique; | 94% |

| Conditions | Yield |
|---|---|
| With trimethylsilylazide; bis-[(trifluoroacetoxy)iodo]benzene In dichloromethane; 1,2-dichloro-ethane at 23℃; for 0.0166667h; Mechanism; Reagent/catalyst; Solvent; Temperature; | 94% |

| Conditions | Yield |
|---|---|
| With trimethylsilylazide; bis-[(trifluoroacetoxy)iodo]benzene In dichloromethane; 1,2-dichloro-ethane at 23℃; for 0.0166667h; | 94% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-penten With zirconocene dichloride; cyclopentylmagnesium bromide In tetrahydrofuran; toluene at 0 - 25℃; for 1.5h; Stage #2: With 2,4-dimethylpentan-3-one In tetrahydrofuran; toluene at 25℃; for 3h; Stage #3: benzaldehyde In hexane; toluene at 25℃; for 5h; | 93% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View






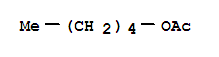









 F+,
F+,  Xn,
Xn,  F,
F,  Xi
Xi