-
Name
2-DIMETHYLAMINOETHANETHIOL HYDROCHLORIDE
- EINECS
- CAS No. 13242-44-9
- Article Data2
- CAS DataBase
- Density g/cm3
- Solubility
-
Melting Point
150-160 °C(lit.)
- Formula C4H11 N S . Cl H
- Boiling Point 121.9°Cat760mmHg
- Molecular Weight 141.665
- Flash Point 27.5°C
- Transport Information
- Appearance white to off-white crystalline powder
- Safety Poison by intraperitoneal route. When heated to decomposition it emits very toxic fumes of Cl−, SOx, and NOx.
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols

- Synonyms Ethanethiol,2-(dimethylamino)-, hydrochloride (6CI,7CI,8CI,9CI);(2-Mercaptoethyl)dimethylammonium chloride; 1-(Dimethylamino)-2-mercaptoethanehydrochloride; 2-(Dimethylamino)ethanethiol hydrochloride;2-(Dimethylamino)ethyl mercaptan hydrochloride;2-(N,N-Dimethylamino)ethanethiol hydrochloride; Captamine hydrochloride; MEDA;N,N-Dimethyl-2-aminoethanethiol hydrochloride; N,N-Dimethylcysteaminehydrochloride; N-(2-Mercaptoethyl)dimethylamine hydrochloride; NSC 45463;Thiofluor
- PSA 42.04000
- LogP 1.27980
Synthetic route
-
-
4584-46-7
(2-chloroethyl)dimethylamine hydrochloride

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: (2-chloroethyl)dimethylamine hydrochloride With sodium thiosulfate In water for 10h; Heating; Stage #2: With hydrogenchloride at 85℃; for 2h; Further stages.; |
-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
199983-80-7
1-(Chloromethyl-dimethyl-silanyl)-2-{tris-[2-(chloromethyl-dimethyl-silanyl)-ethyl]-silanyl}-ethane

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 80℃; for 9.5h; | 99% |
-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
10027-07-3
suberoyl chloride

-
-
143434-02-0
(2-thiosuberoyloxydiethyl)-bis(dimethylamine) dihydrochloride

| Conditions | Yield |
|---|---|
| In dichloromethane for 24h; Heating; | 98.6% |
-
-
7787-60-2
bismuth(III) chloride

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; ethanol pptn.; recrystn. (water); elem. anal.; | 97% |
-
-
7787-60-2
bismuth(III) chloride

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
200342-37-6
BiCl3(SCH2CH2N(CH3)2H)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; ethanol (N2); elem. anal.; | 97% |

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran; toluene; mineral oil Heating; | 95% |
-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
118646-39-2
2-(dimethylamino)ethane-1-sulfonyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| With chlorine In methanol; water at -5 - 0℃; for 1.5h; | 94% |
-
-
20350-15-6
(+)-brefeldin A

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

| Conditions | Yield |
|---|---|
| With N,N,N',N'-tetramethyl-1,8-diaminonaphthalene In methanol; water for 2h; Ambient temperature; | 94% |

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-2-phenylacetophenone In tetrahydrofuran; methanol for 4h; Irradiation; regioselective reaction; | 93% |
-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
79-11-8
chloroacetic acid

-
-
1415981-68-8
2-((2-(dimethylamino)ethyl)thio)acetic acid hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 2-(N,N-dimethylamino)ethylthiol hydrochloride; chloroacetic acid With potassium methanolate In methanol at 40℃; for 2h; Inert atmosphere; Stage #2: With hydrogenchloride In isopropyl alcohol pH=2; | 93% |
-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
199983-94-3
1-{Bis-[2-(chloromethyl-dimethyl-silanyl)-ethyl]-methyl-silanyl}-2-[tris-(2-{bis-[2-(chloromethyl-dimethyl-silanyl)-ethyl]-methyl-silanyl}-ethyl)-silanyl]-ethane

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; isopropyl alcohol at 85℃; for 11h; | 92% |
-
-
1421372-94-2
N-(4-fluoro-2-methoxy-5-nitrophenyl)-4-(1-methyl-1H-indole-3-yl)-pyrimidine-2-amine

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 2-(N,N-dimethylamino)ethylthiol hydrochloride With sodium hydride In N,N-dimethyl-formamide at 20℃; for 0.75h; Inert atmosphere; Stage #2: N-(4-fluoro-2-methoxy-5-nitrophenyl)-4-(1-methyl-1H-indole-3-yl)-pyrimidine-2-amine In N,N-dimethyl-formamide Inert atmosphere; | 90.42% |
| With sodium hydride In N,N-dimethyl-formamide at 60℃; for 4h; | 71% |
| Stage #1: 2-(N,N-dimethylamino)ethylthiol hydrochloride With sodium hydride In hexane; N,N-dimethyl-formamide for 0.75h; Inert atmosphere; Stage #2: N-(4-fluoro-2-methoxy-5-nitrophenyl)-4-(1-methyl-1H-indole-3-yl)-pyrimidine-2-amine In hexane; N,N-dimethyl-formamide Inert atmosphere; | 2.5 g |

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-2-phenylacetophenone In tetrahydrofuran; methanol for 4h; Irradiation; regioselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-2-phenylacetophenone In tetrahydrofuran; methanol for 4h; Irradiation; regioselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-2-phenylacetophenone In tetrahydrofuran; methanol for 4h; Irradiation; regioselective reaction; | 89% |
-
-
74815-81-9
6,7-dibromo-2,3-naphthalenedicarbonitrile

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
1335029-25-8
2,3-dicyano-6,7-bis[(2-dimethylamino)ethylthio]naphthalene

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In N,N-dimethyl acetamide at 20℃; for 1h; | 88% |

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-2-phenylacetophenone In tetrahydrofuran; methanol for 4h; Irradiation; regioselective reaction; | 88% |

| Conditions | Yield |
|---|---|
| With triethylamine In water at -20℃; for 24h; Michael addition; | 87% |
-
-
90-46-0
9-hydroxyxanthene

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
78264-38-7
9-<<2-(dimethylamino)ethyl>thio>-9H-xanthene hydrochloride

| Conditions | Yield |
|---|---|
| In acetonitrile for 1.25h; Heating; | 86% |
-
-
1401456-65-2
C50H70Cl3NP2Si4

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
1401456-67-4
(Ph2PCH2)2N(C6H4-4-Si(CH2CH2CH2SiMe2(CH2S(CH2)2NMe2))3)

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water at 80℃; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-2-phenylacetophenone In tetrahydrofuran; methanol for 3h; Inert atmosphere; Irradiation; | 86% |
-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
103-80-0
phenylacetyl chloride

-
-
143434-00-8
(2-phenylthioacetoxyethyl)dimethylammonium chloride

| Conditions | Yield |
|---|---|
| In dichloromethane a.) RT, 45 min, b.) reflux, 24 h; | 85.88% |
-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
83726-75-4
2-chloro-4-(thien-2'-yl)pyrimidine

-
-
83726-78-7
N,N-dimethyl-2-<<4'-(thien-2''yl)pyrimidin-2'-yl>thio>ethylamine

| Conditions | Yield |
|---|---|
| Stage #1: 2-(N,N-dimethylamino)ethylthiol hydrochloride With sodium ethanolate at 20℃; for 0.25h; Stage #2: 2-chloro-4-(thien-2'-yl)pyrimidine at 70℃; for 12h; Further stages.; | 85% |
| With sodium ethanolate In ethanol for 5h; Heating; | 1.25 g |
-
-
1426827-54-4
C29H45NO2Si3

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-2-phenylacetophenone In tetrahydrofuran; methanol for 3h; Inert atmosphere; Irradiation; | 84% |

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-2-phenylacetophenone In tetrahydrofuran; methanol for 4h; Irradiation; regioselective reaction; | 84% |

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 2-(N,N-dimethylamino)ethylthiol hydrochloride With sodium hydroxide In methanol for 0.0833333h; Inert atmosphere; Stage #2: (NEt4)[Ni(2-(N-(dimethylaminoethyl)aminocarbonyl)benzenethiolate)Cl] In methanol for 24h; Inert atmosphere; | 84% |
-
-
783349-34-8
6-chloro-7-fluoro-8-nitro-9H-β-carboline

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
783349-57-5
[2-(6-chloro-8-nitro-9H-β-carbolin-7-ylsulfanyl)-ethyl]-dimethyl-amine

| Conditions | Yield |
|---|---|
| Stage #1: 6-chloro-7-fluoro-8-nitro-9H-β-carboline; 2-(N,N-dimethylamino)ethylthiol hydrochloride With n-butyllithium In DMF (N,N-dimethyl-formamide) at 20℃; for 0.583333h; Stage #2: With sodium hydrogencarbonate In water at 20℃; | 83% |
| Stage #1: 2-(N,N-dimethylamino)ethylthiol hydrochloride With n-butyllithium In DMF (N,N-dimethyl-formamide); hexane at 20℃; for 0.0833333h; Stage #2: 6-chloro-7-fluoro-8-nitro-9H-β-carboline In DMF (N,N-dimethyl-formamide); hexane at 20℃; for 0.5h; Stage #3: With sodium hydrogencarbonate In DMF (N,N-dimethyl-formamide); hexane; water at 20℃; | 83% |

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
1338246-79-9
[Pt(SCH2CH2NMe2)(8-methoxycyclooct-4-ene-1-yl)]

| Conditions | Yield |
|---|---|
| With NaOMe In methanol (N2); using Schlenk techniques; addn. of NaOMe in MeOH to methanolic soln. of Me2NCH2CH2SH*HCl, addn. of Pt2(μ-Cl)2(C8H12OMe)2, stirring for 15 min; evapn. to dryness, extn. with CH2Cl2; elem. anal.; | 83% |
-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
1072-11-3
bis(N,N-dimethyl-2-aminoethyl)disulfide

| Conditions | Yield |
|---|---|
| With sodium chlorite In methanol; water at 25℃; for 0.5h; Dimerization; | 82% |
| Stage #1: 2-(N,N-dimethylamino)ethylthiol hydrochloride With iodine In methanol Stage #2: With sodium hydroxide In water pH=> 10; | 80% |
| With sodium chlorite In methanol; water at 0℃; Dimerization; |
-
-
12092-47-6
di-μ-chloro-bis(1,5-cyclooctadiene)dirhodium

-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In methanol stirring (30 min), evaporation; drying, extracting (CH2Cl2), filtration, concentration, addition (CH3CN), coo;ing (-20°C), filtration (with cool CH3CN), drying (vac.); elem. anal.; | 82% |
-
-
13242-44-9
2-(N,N-dimethylamino)ethylthiol hydrochloride

-
-
124959-28-0
2-chloro-4-(2'-furanyl)pyrimidine

-
-
109628-19-5
N,N-dimethyl-2-<(4'-furan-2''-ylpyrimidin-2'-yl)thio>ethylamine

| Conditions | Yield |
|---|---|
| Stage #1: 2-(N,N-dimethylamino)ethylthiol hydrochloride With sodium ethanolate at 20℃; for 0.25h; Stage #2: 2-chloro-4-(2'-furanyl)pyrimidine at 70℃; for 12h; Further stages.; | 81% |
| Stage #1: 2-(N,N-dimethylamino)ethylthiol hydrochloride With sodium ethanolate In ethanol at 20℃; for 0.25h; Inert atmosphere; Stage #2: 2-chloro-4-(2'-furanyl)pyrimidine In ethanol at 70℃; Inert atmosphere; | 81% |
2-Dimethylamino ethanethiol hydrochloride Chemical Properties
Molecular Structure of 2-Dimethylamino ethanethiol hydrochloride (CAS NO.13242-44-9):
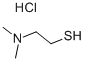
EINECS: 236-221-6
IUPAC Name: 2-(Dimethylamino)ethanethiol hydrochloride
Molecular Formula: C4H12ClNS
Molecular Weight: 141.662780 g/mol
H-Bond Donor: 1
H-Bond Acceptor: 1
Canonical SMILES: CN(C)CCS.Cl
InChI: InChI=1S/C4H11NS.ClH/c1-5(2)3-4-6;/h6H,3-4H2,1-2H3;1H
InChIKey: NRVFDGZJTPCULU-UHFFFAOYSA-N
Flash Point: 27.5 °C
Enthalpy of Vaporization: 35.99 kJ/mol
Boiling Point: 121.9 °C at 760 mmHg
Vapour Pressure: 14.3 mmHg at 25 °C
Melting Point: 150-160 °C(lit.)
Storage Temp.: Refrigerator (+4°C)
2-Dimethylamino ethanethiol hydrochloride Toxicity Data With Reference
| 1. | ipr-mus LD50:280 mg/kg | YKKZAJ Yakugaku Zasshi. Journal of Pharmacy. 93 (1973),25. |
2-Dimethylamino ethanethiol hydrochloride Consensus Reports
Reported in EPA TSCA Inventory.
2-Dimethylamino ethanethiol hydrochloride Safety Profile
Safety Information of 2-Dimethylamino ethanethiol hydrochloride (CAS NO.13242-44-9):
Hazard Codes: Xi 
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin
Safety Statements: 26-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
S36:Wear suitable protective clothing
WGK Germany: 3
RTECS: KJ1051000
F: 3-10-13-23
Poison by intraperitoneal route. When heated to decomposition it emits very toxic fumes of Cl−, SOx, and NOx.
2-Dimethylamino ethanethiol hydrochloride Specification
2-Dimethylamino ethanethiol hydrochloride with CAS registry number of 13242-44-9 is white to off-white crystalline powder, also known as Captamine hydrochloride [USAN] ; (2-Mercaptoethyl)dimethylammonium chloride ; (Dimethylamino)ethanethiol hydrochloride ; 1-(Dimethylamino)-2-thioethane hydrochloride ; 2-(Dimethylamino)ethanethiol hydrochloride ; 2-Dimethylamino ethanethiol hydrochloride ; Captamine HCl ; Captamine hydrochloride ; MEDA ; N,N-Dimethyl-2-aminoethanethiol hydrochloride ; N,N-Dimethylcysteamine hydrochloride ; N-(2-Mercaptoethyl)dimethylamine hydrochloride ; N-Dimethylcysteamine hydrochloride ; NSC 45463 ; NSC-45463 ; UNII-M43AX41U87 ; Ethanethiol, 2-(dimethylamino)-, hydrochloride ; Ethanethiol, 2-(dimethylamino)-, hydrochloride (1:1) . It is incompatible with strong oxidizing agents. When heated to decomposition, it yields Hydrogen chloride , Carbon monoxide , oxides of Nitrogen , oxides of sulfur, Carbon dioxide , Hydrogen sulfide .
Related Products
- 2-Dimethylamino ethanethiol hydrochloride
- 2-Dimethylamino-1,3-dioxolane
- 2-DIMETHYLAMINO-1,4-NAPHTHOQUINONE
- 2-Dimethylamino-2-Methyl-1-Propanol
- 2-Dimethylamino-2-phenylbutan-1-ol
- 2-Dimethylaminobutan-1-ol
- 2-Dimethylaminoethanol (+)-bitartrate salt
- 2-Dimethylaminoethanol hydrogen L-(+)-tartrate
- 2-Dimethylaminoethyl (1H-indol-3-yl)acetate hydrochloride
- 2-DIMETHYLAMINOETHYL 1-NAPHTHYL-ACETATE HYDROCHLORIDE
- 13242-53-0
- 13242-55-2
- 132426-19-8
- 132431-09-5
- 132435-02-0
- 1324-35-2
- 132435-65-5
- 132437-90-2
- 13244-33-2
- 13244-35-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View