-
Name
2-Naphthyl benzoate
- EINECS 202-247-1
- CAS No. 93-44-7
- Article Data86
- CAS DataBase
- Density 1.199 g/cm3
- Solubility insoluble in water
- Melting Point 105-109 °C
- Formula C17H12O2
- Boiling Point 413.5 °C at 760 mmHg
- Molecular Weight 248.281
- Flash Point 174.6 °C
- Transport Information
- Appearance light brown crystalline powder
- Safety 24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 2-Naphthalenol,benzoate (9CI);2-Naphthol, benzoate (6CI,7CI,8CI);2-Benzoyloxynaphthalene;Benzonaphthol;Betabenzon;NSC 5537;Naphthalen-2-ylbenzoate;b-Naphthyl benzoate;2-Naphthalenol,2-benzoate;
- PSA 26.30000
- LogP 4.05900
Synthetic route

| Conditions | Yield |
|---|---|
| With TiO(acac)2 In xylene for 36h; Heating; | 100% |
| Stage #1: benzoic acid With trifluoroacetic anhydride; indium(III) chloride at 20℃; Stage #2: β-naphthol at 20℃; for 0.166667h; | 98% |
| With N,N-bis[2-oxo-3-oxazolidinyl]phosphorodiamidic chloride; triethylamine In dichloromethane for 1h; Ambient temperature; | 91% |

| Conditions | Yield |
|---|---|
| zirconium(IV) oxychloride In dichloromethane at 20℃; for 21h; | 98% |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; for 4h; Inert atmosphere; Schlenk technique; | 98% |
| at 60℃; for 2h; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: benzoic acid anhydride With molybdenium(VI) dioxodichloride In dichloromethane at 20℃; for 0.5h; Stage #2: β-naphthol In dichloromethane at 20℃; for 70h; | 98% |
| With bismuth(lll) trifluoromethanesulfonate In acetonitrile for 0.75h; Heating; | 95% |
| With bismuth oxide perchlorate In dichloromethane at 20℃; for 1h; | 92% |

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane In neat (no solvent) at 70 - 140℃; for 0.183333h; Microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethylene glycol monomethyl ether-3-methylimidazolium methanesulfonate at 25 - 35℃; | 97% |
-
-
18081-08-8
2-naphthyl trimethylsilyl ether

-
-
93-97-0
benzoic acid anhydride

-
-
93-44-7
2-naphthyl benzoate

| Conditions | Yield |
|---|---|
| bismuth(lll) trifluoromethanesulfonate In acetonitrile for 1.5h; Heating; | 95% |

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 70 - 80℃; for 5h; | 95% |

| Conditions | Yield |
|---|---|
| With bis(2,6-diisopropylphenyl)-3,4,5,6-tetrahydropyrimidin-1-ium chloride; palladium diacetate; sodium carbonate In 5,5-dimethyl-1,3-cyclohexadiene at 100℃; for 24h; Sealed tube; | 94% |
| With sodium cyanide; copper(l) iodide; sodium hydroxide In neat (no solvent) at 120℃; for 23h; | 89% |
| With iron(II) triflate; potassium tert-butylate; 1,3-bis[(2,6-diisopropyl)phenyl]imidazolinium chloride In 1,4-dioxane at 90℃; for 24h; | 83% |

| Conditions | Yield |
|---|---|
| With iron(II) triflate; potassium tert-butylate; oxygen; 1,3-bis[(2,6-diisopropyl)phenyl]imidazolinium chloride In 1,4-dioxane at 90℃; for 6h; | 92% |

| Conditions | Yield |
|---|---|
| With oxygen; cesium fluoride; bis[2-(diphenylphosphino)phenyl] ether; bis(dibenzylideneacetone)-palladium(0) In 5,5-dimethyl-1,3-cyclohexadiene at 60℃; under 760.051 Torr; for 12h; | 92% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; oxygen; palladium dichloride In acetonitrile at 60℃; for 5h; | 92% |

| Conditions | Yield |
|---|---|
| With [RhCl2(p-cymene)]2; oxygen; caesium carbonate; 1,3-bis[(2,6-diisopropyl)phenyl]imidazolinium chloride In 5,5-dimethyl-1,3-cyclohexadiene at 130℃; for 24h; | 90% |
| With oxygen; palladium diacetate; sodium carbonate; 1,3-bis[(2,6-diisopropyl)phenyl]imidazolinium chloride In 5,5-dimethyl-1,3-cyclohexadiene at 130℃; for 36h; | 88% |

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 75 - 80℃; for 6h; | 89% |
-
-
30784-04-4
2-naphthyl tetrahydro-2H-pyran-2-yl ether

-
-
93-97-0
benzoic acid anhydride

-
-
93-44-7
2-naphthyl benzoate

| Conditions | Yield |
|---|---|
| bismuth(lll) trifluoromethanesulfonate In acetonitrile for 2h; Heating; | 85% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; triphenylphosphine; potassium hydroxide In water at 80℃; for 12h; Suzuki Coupling; Inert atmosphere; | 84% |

-
-
591-50-4
iodobenzene

-
-
199620-14-9, 13007-92-6
chromium(0) hexacarbonyl

-
-
135-19-3
β-naphthol

-
-
93-44-7
2-naphthyl benzoate

| Conditions | Yield |
|---|---|
| With triethylamine; nickel dichloride In N,N-dimethyl-formamide at 100℃; for 3.7h; | 83% |

-
-
201230-82-2
carbon monoxide

-
-
59-88-1
phenylhydrazine hydrochloride

-
-
135-19-3
β-naphthol

-
-
93-44-7
2-naphthyl benzoate

| Conditions | Yield |
|---|---|
| With bis(diphenylphosphino)propanepalladium(II) dichloride; oxygen; sodium hydroxide at 100℃; under 760.051 Torr; for 12h; Schlenk technique; Molecular sieve; Green chemistry; | 82% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In toluene at 120℃; for 12h; Sealed tube; | 81% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1,4-dioxane at 120℃; for 48h; Green chemistry; | 79% |
-
-
32316-92-0
naphthalene-2-boronic acid

-
-
74542-54-4
N,N-phenyl-p-toluenesulfonylbenzamide

-
-
93-44-7
2-naphthyl benzoate

| Conditions | Yield |
|---|---|
| With allylchloro[1,3-bis(2,6-di-isopropylphenyl)imidazol-2-ylidine]palladium(II); oxygen; cesium fluoride In 5,5-dimethyl-1,3-cyclohexadiene at 65℃; for 15h; | 77% |
| With dmap; potassium phosphate In 1,2-dimethoxyethane at 40℃; for 24h; Schlenk technique; Irradiation; | 75% |
-
-
85909-02-0
N-benzyl-N-(tert-butoxycarbonyl)-benzamide

-
-
135-19-3
β-naphthol

-
-
93-44-7
2-naphthyl benzoate

| Conditions | Yield |
|---|---|
| With sodium t-butanolate In toluene at 150℃; for 24h; Sealed tube; | 77% |
-
-
15226-74-1, 61091-28-9, 61117-58-6
dicobalt octacarbonyl

-
-
591-50-4
iodobenzene

-
-
135-19-3
β-naphthol

-
-
93-44-7
2-naphthyl benzoate

| Conditions | Yield |
|---|---|
| With triethylamine In toluene at 100℃; for 15h; Catalytic behavior; | 73% |


| Conditions | Yield |
|---|---|
| With Ni(NTf2)2·xH2O; 4-mercapto-4-oxide-dinaphtho<2,1-d:1',2'-f><1,3,2>dioxaphosphepin; 9-(2-mesityl)-10-methylacridinium perchlorate In dichloromethane at 20℃; for 48h; Inert atmosphere; Irradiation; | 65% |
-
-
500312-07-2
O-(2-naphthyl) O-phenyl thiocarbonate

-
A
-
93-44-7
2-naphthyl benzoate

-
B
-
82408-29-5
phenyl naphthalene-2-carboxylate

| Conditions | Yield |
|---|---|
| With triethylsilane; di-tert-butyl peroxide In benzene at 135℃; for 5h; Inert atmosphere; Sealed tube; regioselective reaction; | A 15% B 61% |

| Conditions | Yield |
|---|---|
| With oxygen; copper diacetate In acetonitrile at 20 - 120℃; for 24h; Schlenk technique; Sealed tube; | 60% |
-
-
111730-61-1
1-benzoyloxy-5-chloro-3,6-diethyl-1,2-dihydro-2-oxopyrazine

-
-
108-93-0
cyclohexanol

-
A
-
93-44-7
2-naphthyl benzoate

-
B
-
72876-06-3
5-chloro-3,6-diethyl-2-hydroxypyrazine-1-oxide

| Conditions | Yield |
|---|---|
| With triethylamine In benzene for 3h; Heating; | A 38% B n/a |


| Conditions | Yield |
|---|---|
| With iron(III)-acetylacetonate In n-heptane at 105℃; for 24h; Inert atmosphere; | 32% |
-
-
98-07-7
Benzotrichlorid

-
-
135-19-3
β-naphthol

-
A
-
6333-07-9
1-benzoyl-2-naphthol

-
B
-
93-44-7
2-naphthyl benzoate

| Conditions | Yield |
|---|---|
| With iron(III) chloride on silica at 70 - 72℃; | A 27.41% B n/a |


| Conditions | Yield |
|---|---|
| With potassium carbonate In 1,4-dioxane at 120℃; for 48h; Green chemistry; | 20% |
-
-
93-44-7
2-naphthyl benzoate

-
-
137-07-5
2-amino-benzenethiol

-
A
-
883-93-2
2-Phenylbenzothiazole

-
B
-
135-19-3
β-naphthol

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one; potassium carbonate at 100℃; for 0.75h; Hydrolysis; cyclization; debenzoylation; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In 1-methyl-pyrrolidin-2-one for 0.5h; Heating; | 100% |
| With potassium fluoride; thiophenol In 1-methyl-pyrrolidin-2-one for 0.5h; Heating; | 92% |
| With HEPES buffer; human liver microsomal carboxylesterase 1 In dimethyl sulfoxide pH=7.4; Enzyme kinetics; Further Variations:; Reagents; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In 1,4-dioxane at 120℃; for 48h; Green chemistry; | 100% |
-
-
93-44-7
2-naphthyl benzoate

-
-
72771-44-9
1-nitonaphthalen-2-yl benzoate

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In acetic acid at 70 - 80℃; for 0.5h; | 95% |

| Conditions | Yield |
|---|---|
| for 0.116667h; Rearrangement; microwave irradiation; | 92% |
| With sodium dodecyl-sulfate for 7h; Irradiation; | 75% |
| With aluminium trichloride In chlorobenzene at 110℃; under 760 Torr; for 0.0833333h; Irradiation; microwave oven; | 72% |
-
-
93-44-7
2-naphthyl benzoate

-
-
108-98-5
thiophenol

-
A
-
884-09-3
phenyl thiobenzoate

-
B
-
882-33-7
diphenyldisulfane

-
C
-
135-19-3
β-naphthol

| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one; potassium carbonate for 0.5h; Hydrolysis; debenzoylation; oxidation; substitution; Heating; | A n/a B n/a C 90% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In 1,4-dioxane at 120℃; for 48h; Green chemistry; | 89% |
2-Naphthyl Benzoate Chemical Properties
IUPAC Name: Naphthalen-2-yl Benzoate
CAS:93-44-7
EINECS:202-247-1
Product Categories: Pharmaceutical Raw Materials ; Aromatic Ester ; Functional Materials ; Liquid Crystals & Related Compounds ; Phenyl Esters (Liquid Crystals).
Following is the Molecular Structure of 2-Naphthyl Benzoate (93-44-7):
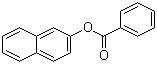
SMILES: O=C(Oc2ccc1c(cccc1)c2)c3ccccc3
InChI: InChI=1/C17H12O2/c18-17(14-7-2-1-3-8-14)19-16-11-10-13-6-4-5-9-15?(13)12-16/h1-12H
InChIKey: DWJIJRSTYFPKGD-UHFFFAOYAH
Std. InChI: InChI=1S/C17H12O2/c18-17(14-7-2-1-3-8-14)19-16-11-10-13-6-4-5-9-1?5(13)12-16/h1-12H Std. InChIKey: DWJIJRSTYFPKGD-UHFFFAOYSA-N
Molecular Formula:C17H12O2
Molar Volume: 206.9cm3
Melting Point: 105-109℃
Boiling Point: 413.5℃ at 760mmHg
Flash Point: 174.6℃
Density: 1.199g/cm3
Molar Refractivity: 75.74cm3
Polarizability: 30.02 10-24cm3
Enthalpy of Vaporization: 66.63kJ/mol
Vapour Pressure: 4.77E-07mmHg at 25℃
2-Naphthyl Benzoate Uses
2-Naphthyl Benzoate (93-44-7) can be used as hardening agent for paraffin.
2-Naphthyl Benzoate Safety Profile
Safety Description: S22 S24/25.
S22 Do not breathe dust.
S24 Avoid contact with skin.
S25 Avoid contact with eyes.
Ingestion: May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation: May cause respiratory tract irritation.
Skin: May cause skin irritation.
Eyes: May cause eye irritation.
2-Naphthyl Benzoate Specification
2-Naphthyl Benzoate (93-44-7),which also can be called for Benzoic Acid 2-Naphthyl Ester ; Benzonaphthol ; Beta-Naphthyl Benzoate ; Beta-Naphthyl Benzoate ; 2 Benzoxynaphthalene ; 2-Naphthol Bezoate ; 2-Naphthyl Benzoate ; 2-Benzoyloxy Naphthalene . 2-Naphthyl Benzoate (93-44-7) is a kind of light brown crystalline powder. This chemical is stable under normal temperatures and pressures.After decomposition it turned into Carbon Monoxide and Carbon Dioxide . 2-Naphthyl Benzoate (93-44-7) should be stored in a tightly closed container of cool,dry places. When you are handling this chemical,take care of avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Related Products
- 2-Naphthyl acetate
- 2-Naphthyl acrylate
- 2-Naphthyl alpha-D-glucopyranoside
- 2-Naphthyl Benzoate
- 2-Naphthyl beta-D-glucopyranoside
- 2-Naphthyl diphenyl phosphate
- 2-Naphthylacetic acid
- 2-Naphthylacetonitrile
- 2-Naphthylacrylic acid
- 2-Naphthylamine, 1-methoxy-, hydrochloride
- 934476-98-9
- 934-48-5
- 93449-32-2
- 934505-32-5
- 934506-10-2
- 934-53-2
- 934537-52-7
- 934538-04-2
- 934538-12-2
- 934541-31-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View