-
Name
2-Tetrahydrofurfurylamine
- EINECS 225-351-9
- CAS No. 4795-29-3
- Article Data43
- CAS DataBase
- Density 0.967 g/cm3
- Solubility miscible with water
- Melting Point 156 °C at 760 mmHg
- Formula C5H11NO
- Boiling Point 45.6 °C
- Molecular Weight 101.148
- Flash Point 114 °F
- Transport Information UN 2943 3/PG 3
- Appearance Colorless to light yellow liquid
- Safety 26-36-37/39-16
- Risk Codes 10-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Furfurylamine,tetrahydro- (6CI,7CI,8CI);(2-Tetrahydrofurylmethyl)amine;(RS)-(2-Tetrahydrofuryl)methylamine;(RS)-2-Tetrahydrofurfurylamine;(Tetrahydrofuran-2-yl)methanamine;(Tetrahydrofuran-2-ylmethyl)amine;1-(Tetrahydrofuran-2-yl)methanamine;2-(Aminomethyl)tetrahydrofuran;2-Tetrahydrofuranmethanamine;2-Tetrahydrofurfurylamine;C-(Tetrahydrofuran-2-yl)methylamine;N-[(Oxolan-2-yl)methyl]amine;NSC 76037;Tetrahydrofurfurylamine;2-aminomethyltetrahydrofuran;
- PSA 35.25000
- LogP 0.82440
Synthetic route

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen In tetrahydrofuran at 180℃; for 48h; Autoclave; | 94% |
| With ammonia; hydrogen In tetrahydrofuran at 180℃; for 48h; Autoclave; | 94 %Chromat. |
| Stage #1: (2-furyl)methyl alcohol With ammonia; nickel In tetrahydrofuran at 20℃; under 2625.26 Torr; for 0.333333h; Autoclave; Stage #2: With hydrogen In tetrahydrofuran at 180℃; under 7500.75 Torr; for 48h; Temperature; Pressure; Reagent/catalyst; |

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen In tetrahydrofuran at 180℃; for 12h; Autoclave; | A 5.1% B 91% |
| Multi-step reaction with 2 steps 1: ammonia; hydrogen / tetrahydrofuran / 2 h / 80 °C / Autoclave 2: ammonia; hydrogen / tetrahydrofuran / 24 h / 160 °C / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen In tetrahydrofuran at 180℃; for 12h; Autoclave; | A 40.7% B 53.8% |
| With ammonia; hydrogen In methanol at 100℃; under 22502.3 Torr; for 0.25h; Autoclave; Green chemistry; | A 34% B 49% |
| With ethanol; ammonia; nickel at 180℃; under 73550.8 Torr; Hydrogenation; |
-
-
88-14-2
2-furanoic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
110-89-4
piperidine

-
C
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
D
-
110-58-7
1-pentanamine

-
E
-
109-73-9
N-butylamine

| Conditions | Yield |
|---|---|
| Stage #1: 2-furanoic acid With cyclopentyl methyl ether; ammonia at 200℃; under 4500.45 Torr; Sealed tube; Green chemistry; Stage #2: With cyclopentyl methyl ether; ammonia; hydrogen at 200℃; under 42004.2 Torr; for 6.5h; Cooling with ice; Green chemistry; | A 13% B 9% C 43% D 6% E 10% |

-
-
98-01-1
furfural

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
B
-
19377-82-3
N-furfurylidenefurfurylamine

-
C
-
617-89-0
furan-2-ylmethanamine

| Conditions | Yield |
|---|---|
| With 0.5% Ru/activated carbon; ammonia; hydrogen In methanol at 100℃; under 22502.3 Torr; for 0.25h; Autoclave; Green chemistry; | A 6% B 8% C 40% |


| Conditions | Yield |
|---|---|
| With ammonia; hydrogen at 89.84℃; for 4h; High pressure; | 36% |
| Hydrogenation; | |
| With platinum(IV) oxide Hydrogenation; | |
| With palladium on activated charcoal; acetic acid Hydrogenation; | |
| With nickel at 115 - 140℃; under 58840.6 Torr; Hydrogenation; |
-
-
98-00-0
(2-furyl)methyl alcohol

-
A
-
97-99-4
Tetrahydrofurfuryl alcohol

-
B
-
4795-29-3
TETRAHYDROFURFURYLAMINE

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen In tetrahydrofuran at 160℃; for 24h; Autoclave; | A 12.6% B 34.2% |
| With ammonia; hydrogen In tetrahydrofuran at 160℃; for 24h; Autoclave; | A 15.5% B 11% |
| With ammonia; hydrogen In tetrahydrofuran at 160℃; for 24h; Autoclave; | A 15.5 %Chromat. B 11 %Chromat. |

| Conditions | Yield |
|---|---|
| With ammonia; water at 150℃; im versilberten Gefaess; |

| Conditions | Yield |
|---|---|
| With ammonia; nickel at 200℃; unter Druck; | |
| With nickel-aluminium barium hydroxide-catalyst; ammonia; hydrogen at 210℃; under 12503.6 Torr; |
-
-
494-47-3
hydrofuramide

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
B
-
18240-50-1
bis((furan-2-yl)methyl)amine

| Conditions | Yield |
|---|---|
| With kieselguhr; ethanol; nickel at 100℃; under 51485.6 - 80905.8 Torr; Hydrogenation; |
-
-
494-47-3
hydrofuramide

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
B
-
5343-16-8
di-(2-tetrahydrofurfurylmethyl)amine

| Conditions | Yield |
|---|---|
| With diethyl ether; ethanol; nickel at 125℃; under 73550.8 - 110326 Torr; Hydrogenation; | |
| With nickel; methyl cyclohexane at 125℃; under 73550.8 - 110326 Torr; Hydrogenation; | |
| With diethyl ether; nickel at 125℃; under 73550.8 - 110326 Torr; Hydrogenation; | |
| With ethanol; nickel at 125℃; under 73550.8 - 110326 Torr; Hydrogenation; |
-
-
97-99-4
Tetrahydrofurfuryl alcohol

-
A
-
110-89-4
piperidine

-
B
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
C
-
10324-58-0
1-pentylpiperidine

-
D
-
1199-81-1
1-(tetrahydrofuran-2-ylmethyl)piperidine

-
E
-
5343-16-8
di-(2-tetrahydrofurfurylmethyl)amine

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen; CuO/ZnO/Al2O3 at 230℃; under 26252.1 Torr; Product distribution; other heterogenous Cu-, Ni-, Co- and Fe-containing bifunctional catalyst systems; | A 4.0 % Chromat. B 37 % Chromat. C 2.0 % Chromat. D 2.0 % Chromat. E 20.0 % Chromat. |


-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

| Conditions | Yield |
|---|---|
| With Pd-BaSO4; ethanol Hydrogenation; |
-
-
64-17-5
ethanol

-
-
494-47-3
hydrofuramide

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
B
-
18240-50-1
bis((furan-2-yl)methyl)amine

| Conditions | Yield |
|---|---|
| at 100℃; under 73550.8 Torr; Hydrogenation; |

-
-
23247-31-6
(E)-tetrahydrofuran-2-carbaldehyde oxime

-
-
64-17-5
ethanol

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
B
-
2508-29-4
5-hydroxypentylamine


| Conditions | Yield |
|---|---|
| With ethanol; sodium |

| Conditions | Yield |
|---|---|
| With 1,4-dioxane; copper oxide-chromium oxide at 250℃; under 147102 - 220652 Torr; Hydrogenation; | |
| With 1,4-dioxane; copper oxide-chromium oxide at 250℃; under 147102 - 220652 Torr; Hydrogenation; |
-
-
60-29-7
diethyl ether

-
-
494-47-3
hydrofuramide

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
B
-
5343-16-8
di-(2-tetrahydrofurfurylmethyl)amine

| Conditions | Yield |
|---|---|
| at 200℃; under 73550.8 - 110326 Torr; Hydrogenation; |

-
-
64-17-5
ethanol

-
-
494-47-3
hydrofuramide

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
B
-
5343-16-8
di-(2-tetrahydrofurfurylmethyl)amine

| Conditions | Yield |
|---|---|
| Hydrogenation; |

-
-
494-47-3
hydrofuramide

-
-
82166-21-0
methyl cyclohexane

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
B
-
5343-16-8
di-(2-tetrahydrofurfurylmethyl)amine

| Conditions | Yield |
|---|---|
| at 200℃; under 73550.8 - 110326 Torr; Hydrogenation; |

-
-
97-99-4
Tetrahydrofurfuryl alcohol

-
-
7664-41-7
ammonia

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
C
-
5343-16-8
di-(2-tetrahydrofurfurylmethyl)amine

| Conditions | Yield |
|---|---|
| at 210℃; under 12503.6 Torr; |

-
-
97-99-4
Tetrahydrofurfuryl alcohol

-
-
7664-41-7
ammonia

-
A
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
C
-
5343-16-8
di-(2-tetrahydrofurfurylmethyl)amine

| Conditions | Yield |
|---|---|
| at 160℃; |


| Conditions | Yield |
|---|---|
| With ethanol; nickel at 150℃; Hydrogenation.unter Druck; |

| Conditions | Yield |
|---|---|
| With ethanol; sodium |

| Conditions | Yield |
|---|---|
| With 1,4-dioxane; copper oxide-chromium oxide at 250℃; under 147102 - 220652 Torr; Hydrogenation; | |
| With 1,4-dioxane; copper oxide-chromium oxide at 250℃; under 147102 - 220652 Torr; Hydrogenation; |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
79-04-9
chloroacetyl chloride

-
-
39089-62-8
2-chloro-N-[(tetrahydrofuran-2-yl)methyl]acetamide

| Conditions | Yield |
|---|---|
| In dichloromethane | 100% |
| With potassium carbonate In dichloromethane Heating; | 97% |
| With trimethylamine In dichloromethane at 0 - 20℃; for 4h; | 76% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
1034926-02-7
C11H12BrF2NO3S

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 100% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
1034926-01-6
C11H13BrFNO3S

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 100% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
1034926-05-0
C11H13BrClNO3S

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 100% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
1173799-71-7
1,1,1-trifluoro-N-((1S,2S)-2-isothiocyanatocyclohexyl)methanesulfonamide

-
-
1221716-46-6
1,1,1-trifluoro-N-((1S,2S)-2-(3-((tetrahydrofuran-2-yl)methyl)thioureido)cyclohexyl)methanesulfonamide

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 2h; Inert atmosphere; | 100% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
68836-19-1
5-bromo-3-(methylthio)-1,4-diphenyl-1,2,4-triazolium bromide

-
-
76116-95-5
5-(tetrahydrofurfurylimino)-3-methylthio-1,4-diphenyl-Δ2-1,2,4-triazole

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform at 0℃; for 0.166667h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: TETRAHYDROFURFURYLAMINE With triethylamine In methanol at 0℃; for 0.166667h; Inert atmosphere; Stage #2: 2,4-dichloroquinazoline In methanol; dichloromethane at 0 - 20℃; for 0.5h; Inert atmosphere; | 99% |
| Stage #1: TETRAHYDROFURFURYLAMINE With triethylamine In methanol at 0℃; for 0.166667h; Inert atmosphere; Stage #2: 2,4-dichloroquinazoline In methanol; dichloromethane at 0 - 20℃; for 0.5h; Inert atmosphere; | 99% |
| With sodium acetate In tetrahydrofuran; water at 65℃; | 82% |
| With sodium hydroxide 1.) THF, 30 min, 2.) THF, 30 min; Multistep reaction; | |
| With sodium acetate In tetrahydrofuran; water at 60℃; |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 99% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In dichloromethane at 20℃; for 16h; | 99% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

| Conditions | Yield |
|---|---|
| With triethylamine In ethylene glycol at 130℃; for 16h; | 99% |

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In diethyl ether at 20℃; for 2h; Condensation; | 98% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 160℃; for 0.5h; | 98% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
351003-54-8
4-bromo-2,6-dichlorobenzenesulfonyl chloride

-
-
1034926-06-1
C11H12BrCl2NO3S

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 24h; | 98% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
490-11-9
3,4-pyridinecarboxylic acid

-
-
950445-43-9
2-((tetrahydrofuran-2-yl)methyl)-2H-pyrrolo[3,4-c]pyridine-1,3-dione

| Conditions | Yield |
|---|---|
| for 0.0333333h; Microwave irradiation; | 98% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
98-61-3
4-iodobenzenesulfonyl chloride

-
-
326885-27-2
4-iodo-N-(tetrahydro-2-furanylmethyl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; Cooling with ice/water; Inert atmosphere; | 98% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
1417785-60-4
N-(tetrahydrofuran-2-ylmethyl)acetamidine hydrobromide

| Conditions | Yield |
|---|---|
| In ethanol at 0 - 20℃; | 98% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
14631-43-7
2-tetrahydrofuran-2-carbonitrile

| Conditions | Yield |
|---|---|
| With pyridine; Oxone; 4-acetylamino-2,2,6,6-tetramethyl-1-piperidinoxy; Pyridine hydrobromide In dichloromethane at 20℃; for 12h; Green chemistry; | 98% |
| With dmap; copper(l) iodide; 9-azabicyclo[3.3.1]nonane N-oxyl; oxygen; 4,4'-di-tert-butyl-2,2'-bipyridine In acetonitrile at 20℃; under 760.051 Torr; for 15h; Reagent/catalyst; | 47 %Spectr. |

| Conditions | Yield |
|---|---|
| With 14C2H2F3O(1-)*6C4H8O*La2Na8(14+) at 80℃; for 6h; Inert atmosphere; | 98% |
| With bis(1,5-cyclooctadiene)nickel (0); 1,3-bis(2,6-diisopropylphenyl)-1H-imidazol-3-ium In toluene at 140℃; for 16h; Glovebox; Inert atmosphere; Sealed tube; | 88% |
| With C18H15IMnN3O3; sodium t-butanolate In toluene at 120℃; for 18h; Inert atmosphere; Schlenk technique; | 76% |
| With zirconocene dichloride In toluene at 110℃; for 20h; | 57% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
7623-09-8
2-chloropropionyl chloride

-
-
596807-96-4
2-chloro-N-(tetrahydro-furan-2-ylmethyl)-propionamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane Heating; | 97% |
| With potassium carbonate In dichloromethane for 4h; Heating; |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
36082-50-5
2,4-dichloro-5-bromopyrimidine

| Conditions | Yield |
|---|---|
| With triethylamine In methanol at 0 - 20℃; Inert atmosphere; | 97% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
27631-29-4
2-chloro-6,7-dimethoxy-3H-quinazolin-4-one

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 18h; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: TETRAHYDROFURFURYLAMINE; formaldehyd; acetic acid at 15℃; for 0.5h; Stage #2: Glyoxal at 20℃; | 97% |

-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
82140-87-2
3,3-Diazido-2-cyan-acrylsaeure-methylester

-
A
-
130148-77-5
3-Azido-2-cyan-3-(tetrahydro-2-furylmethylamino)acrylsaeure-methylester

| Conditions | Yield |
|---|---|
| In dichloromethane at -30℃; for 16h; | A 96% B n/a |

-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
769956-97-0
(4-nitrophenyl) 3-O-benzyl-estradiol-16-carboxylate

-
-
769956-98-1
JAC01163-3

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; | 96% |
| In acetonitrile at 20℃; |

| Conditions | Yield |
|---|---|
| With benzoic acid In para-xylene at 130℃; for 8h; Inert atmosphere; Schlenk technique; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-amino-4-bromobenzoic acid With N-ethyl-N,N-diisopropylamine; HATU In dichloromethane at 20℃; for 0.25h; Stage #2: TETRAHYDROFURFURYLAMINE In dichloromethane at 20℃; for 2h; | 96% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
454-89-7
3-Trifluoromethylbenzaldehyde

-
-
912291-16-8
1-(tetrahydrofuran-2-yl)-N-(3-(trifluoromethyl)benzyl)methanamine

| Conditions | Yield |
|---|---|
| Stage #1: TETRAHYDROFURFURYLAMINE; 3-Trifluoromethylbenzaldehyde In chloroform at 60℃; Stage #2: With sodium tetrahydroborate In methanol at 25℃; | 96% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
85-44-9
phthalic anhydride

-
-
26116-10-9
2-((tetrahydrofuran-2-yl)methyl)isoindoline-1,3-dione

| Conditions | Yield |
|---|---|
| In chloroform Heating; | 95.2% |
| at 120℃; for 3h; | 95% |
| In chloroform Reflux; | 95.2% |
| With sodium hydrogencarbonate In chloroform; water |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

| Conditions | Yield |
|---|---|
| In tetrahydrofuran | 95% |
-
-
4795-29-3
TETRAHYDROFURFURYLAMINE

-
-
570-22-9
4,5-imidazoledicarboxylic acid

-
-
1204831-04-8
5-((tetrahydrofuran-2-yl)methyl)pyrrolo[3,4-d]imidazole-4,6(1H,5H)-dione

| Conditions | Yield |
|---|---|
| for 0.0666667h; Microwave irradiation; | 95% |
2-Tetrahydrofurfurylamine Consensus Reports
2-Tetrahydrofurfurylamine Standards and Recommendations
2-Tetrahydrofurfurylamine Specification
The Tetrahydrofurfurylamine, also known as 2-Tetrahydrofurfurylamine, is an organic compound with the formula C5H11NO. It belongs to the product categories of Furan & Benzofuran; Amines. Its EINECS registry number is 225-351-9. With the CAS registry number 4795-29-3, its IUPAC name is oxolan-2-ylmethanamine.
Physical properties of Tetrahydrofurfurylamine: (1)ACD/LogP: -0.66; (2)#H bond acceptors: 2; (3)#H bond donors: 2; (4)#Freely Rotating Bonds: 2; (5)Index of Refraction: 1.453; (6)Molar Refractivity: 28.28 cm3; (7)Molar Volume: 104.5 cm3; (8)Surface Tension: 37.2 dyne/cm; (9)Density: 0.967 g/cm3; (10)Flash Point: 45.6 °C; (11)Enthalpy of Vaporization: 39.27 kJ/mol; (12)Boiling Point: 156 °C at 760 mmHg; (13)Vapour Pressure: 2.95 mmHg at 25°C.
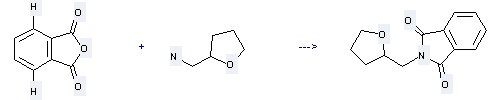
Uses of Tetrahydrofurfurylamine: it can be used to produce N-tetrahydrofurfuryl-phthalimide with phthalic acid anhydride by heating. This reaction will need solvent CHCl3. The yield is about 95.2%.
When you are using this chemical, please be cautious about it as the following:
This chemical may cause inflammation to the skin or other mucous membranes. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. Whenever you will contact it, please wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: C1CC(OC1)CN
(2)InChI: InChI=1S/C5H11NO/c6-4-5-2-1-3-7-5/h5H,1-4,6H2
(3)InChIKey: YNOGYQAEJGADFJ-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 200mg/kg (200mg/kg) | National Technical Information Service. Vol. AD277-689, |
Related Products
- 2-Tetrahydrofurfurylamine
- 479543-46-9
- 479547-57-4
- 47956-45-6
- 479586-24-8
- 479-59-4
- 479611-85-3
- 479613-16-6
- 479-61-8
- 479630-08-5
- 479633-63-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View