-
Name
PORPHINE
- EINECS 202-958-7
- CAS No. 101-60-0
- Article Data88
- CAS DataBase
- Density 1.353 g/cm3
- Solubility
- Melting Point 360°C
- Formula C20H14N4
- Boiling Point 804.4 °C at 760 mmHg
- Molecular Weight 310.358
- Flash Point 377.479 °C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Porphine(6CI,7CI,8CI);21,22,23,24-Tetraazapentacyclo[16.2.1.13,6.18,11.113,16]tetracosa-1,3,5,7,9,11(23),12,14,16,18(21),19-undecaene;Porphin;Porphyrin;
- PSA 56.30000
- LogP 1.19220
Synthetic route

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In n-heptan1ol | 80% |

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With sulfuric acid; water; sodium sulfate at 180℃; for 0.5h; | 77% |

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With sulfuric acid at 90℃; for 0.25h; | 74% |
| With sulfuric acid In isopropyl alcohol at 90℃; for 1h; | |
| With sulfuric acid In butan-1-ol at 90℃; for 0.25h; |
-
-
109-63-7
trifluoroborane diethyl ether

-
-
84-58-2
2,3-dicyano-5,6-dichloro-p-benzoquinone

-
-
1030624-07-7
3,5-dihexylbenzaldehyde

-
B
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| In chloroform | A n/a B 40% |

-
-
1046493-99-5
1-formyl-5-(3-pyridyl)dipyrromethane

-
-
36746-27-7
5-formyl-2,2'-dipyrrylmethane

-
B
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With air; 1,8-diazabicyclo[5.4.0]undec-7-ene; magnesium bromide In toluene at 115℃; microwave irradiation; | A 32% B 6% |

-
-
1046493-90-6
1-nicotinoyl-5-(3-pyridyl)dipyrromethane

-
-
36746-27-7
5-formyl-2,2'-dipyrrylmethane

-
A
-
40882-83-5
5,10,15,20-tetra(3-pyridyl)porphyrin

-
C
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With air; 1,8-diazabicyclo[5.4.0]undec-7-ene; magnesium bromide In toluene at 115℃; microwave irradiation; | A 3% B 28% C 13% |

-
-
1046493-94-0
1-picolinoyl-5-(4-pyridyl)dipyrromethane

-
-
36746-27-7
5-formyl-2,2'-dipyrrylmethane

-
B
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With air; 1,8-diazabicyclo[5.4.0]undec-7-ene; magnesium bromide In toluene at 115℃; microwave irradiation; | A 11% B 28% |

-
-
1046493-91-7
1-isonicotinoyl-5-(4-pyridyl)dipyrromethane

-
-
36746-27-7
5-formyl-2,2'-dipyrrylmethane

-
B
-
101-60-0
porphyrin

-
C
-
16834-13-2
5,10,15,20-tetra(4-pyridyl)-21H,23H-porphine

| Conditions | Yield |
|---|---|
| With air; 1,8-diazabicyclo[5.4.0]undec-7-ene; magnesium bromide In toluene at 115℃; for 1.5h; microwave irradiation; | A 27% B 16% C 14% |

-
-
1046493-89-3
1-picolinoyl-5-(2-pyridyl)dipyrromethane

-
-
36746-27-7
5-formyl-2,2'-dipyrrylmethane

-
B
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With air; 1,8-diazabicyclo[5.4.0]undec-7-ene; magnesium bromide In toluene at 115℃; microwave irradiation; | A 13% B 15% C 12% |

-
-
109-97-7
pyrrole

-
-
100-52-7
benzaldehyde

-
A
-
101-60-0
porphyrin

-
B
-
917-23-7
5,15,10,20-tetraphenylporphyrin

| Conditions | Yield |
|---|---|
| With acetic acid; propionic acid at 140℃; for 0.5h; | A 15% B n/a |

-
-
109-97-7
pyrrole

-
-
50-00-0
formaldehyd

-
-
158399-19-0
2,7-di-tert-butyl-4-formyl-5-methoxycarbonyl-9,9-dimethylxanthene

-
B
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| Stage #1: pyrrole; formaldehyd; 4-formyl-5-methoxycarbonyl-2,7-di-tert-butyl-9,9-dimethylxanthene With boron trifluoride diethyl etherate In chloroform for 1h; Darkness; Inert atmosphere; Stage #2: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In chloroform for 1h; | A 13% B 1% |

-
-
1046493-92-8
1-picolinoyl-5-(3-pyridyl)dipyrromethane

-
-
36746-27-7
5-formyl-2,2'-dipyrrylmethane

-
C
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With air; 1,8-diazabicyclo[5.4.0]undec-7-ene; magnesium bromide In toluene at 115℃; microwave irradiation; | A 9% B 11% C 7% |


| Conditions | Yield |
|---|---|
| Stage #1: 2-pyrrole aldehyde; tripyrromethane With trifluoroacetic acid In dichloromethane at 20℃; Stage #2: With chloranil In dichloromethane Heating; | A 7% B 10% |


| Conditions | Yield |
|---|---|
| Stage #1: 2-hydroxymethylpyrrole; tripyrromethane With trifluoroacetic acid In dichloromethane at 20℃; for 1.5h; Stage #2: With chloranil In dichloromethane for 1h; Heating; | A n/a B 10% |

-
-
109-97-7
pyrrole

-
-
179076-07-4
2,5-bis(2',2',3',3',4',4',4'-heptafluoro-1'-trimethylsilyloxybutyl)pyrrole

-
B
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In dichloromethane | A 9.1% B n/a |
| With toluene-4-sulfonic acid In dichloromethane | A 9.1% B n/a |


| Conditions | Yield |
|---|---|
| Stage #1: pyrrole; formaldehyd With boron trifluoride diethyl etherate In chloroform at 20℃; for 24h; Inert atmosphere; Darkness; Stage #2: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In chloroform for 1h; Inert atmosphere; Reflux; Stage #3: With triethylamine In chloroform at 20℃; Inert atmosphere; | 1.5% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-pyrrole aldehyde; di(pyrrol-2-yl)methane With methyl 2,2-dimethyl-3-(trimethylsiloxy)-1-cyclopropanecarboxylate; trifluoroacetic acid In dichloromethane at 20℃; for 21h; Inert atmosphere; Darkness; Stage #2: With triethylamine; 2,3-dicyano-5,6-dichloro-p-benzoquinone In dichloromethane Inert atmosphere; | 1% |
-
-
27472-36-2
2-hydroxymethylpyrrole

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; magnesium acetate | |
| With acetic acid In xylene at 110 - 115℃; | |
| With acetic acid In xylene at 110 - 115℃; Heating; | |
| Multi-step reaction with 2 steps 1: aq. sodium dodecyl sulfate, HCl / 0.17 h 2: 2,3-dichloro-5,6-dicyano-1,4-benzoquinone / H2O; tetrahydrofuran / 0.5 h View Scheme |

| Conditions | Yield |
|---|---|
| With diethyl ether; iodine; magnesium unter Luftzutritt; |


-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| at 160℃; im Vakuum; |

| Conditions | Yield |
|---|---|
| Stage #1: n-butyllithium; porphyrin In tetrahydrofuran at 20℃; for 0.5h; Stage #2: With water In tetrahydrofuran at 20℃; for 0.25h; Stage #3: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In tetrahydrofuran at 20℃; for 30h; | 99% |
-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With triethylamine; magnesium bromide In dichloromethane at 60℃; | 97% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Reflux; Inert atmosphere; | 94% |
-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With copper(II) acetate monohydrate In methanol; chloroform for 0.5h; Reflux; | 93% |
-
-
157131-39-0
1,3,5-tris{2-(pyridin-4-yl)-vinyl}benzene

-
-
1039768-31-4
[Ru2(η6-p-cymene)2(C6H2O4)Cl2]

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| In methanol for 24h; Reflux; | 91% |

-
-
157131-39-0
1,3,5-tris{2-(pyridin-4-yl)-vinyl}benzene


-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| In methanol for 24h; Reflux; | 83% |


| Conditions | Yield |
|---|---|
| Stage #1: porphyrin; phenyllithium In tetrahydrofuran at -78 - 20℃; for 0.25h; Stage #2: With water In tetrahydrofuran for 0.0833333h; Stage #3: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In tetrahydrofuran | 75% |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide Reflux; Inert atmosphere; | 75% |
| In N,N-dimethyl-formamide Inert atmosphere; Schlenk technique; Reflux; | 75% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In chloroform at 2 - 5℃; for 0.0833333h; | A 71% B 8% |
| With bromine In chloroform at 2 - 5℃; for 0.0833333h; | A 54% B 15% |
-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| With potassium tetrachloroaurate(III); sodium acetate In acetic acid at 80℃; for 0.75 - 2.25h; Product distribution / selectivity; Heating / reflux; | 70% |
| With tetrabutylammonium tetrachloroaurate(III); sodium acetate In acetic acid at 80℃; for 0.75 - 2.25h; Product distribution / selectivity; Heating / reflux; | 70% |
-
-
14873-63-3, 15617-19-3, 51921-56-3
bis(benzonitrile)dichloroplatinum(II)

-
-
101-60-0
porphyrin

-
-
30040-00-7
(porphinato)platinum(II)

| Conditions | Yield |
|---|---|
| With sodium proprionate In chlorobenzene under 760.051 Torr; for 3.5h; Reflux; | 70% |
-
-
42333-78-8
2,4,6-tri(4-pyridyl)-1,3,5-triazine

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane for 24h; Reflux; | 69% |

-
-
101-60-0
porphyrin

-
-
71-48-7, 917-69-1, 5931-89-5, 55881-15-7, 68931-68-0, 68931-69-1, 93029-27-7
cobalt(II) acetate

| Conditions | Yield |
|---|---|
| In methanol; chloroform for 12h; Heating / reflux; | 68% |

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid diethyl ether; N,N,N,N-tetraethylammonium tetrafluoroborate In dichloromethane; acetonitrile Electrolysis; Inert atmosphere; | 68% |
-
-
157131-39-0
1,3,5-tris{2-(pyridin-4-yl)-vinyl}benzene

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| In methanol for 24h; Reflux; | 67% |


| Conditions | Yield |
|---|---|
| In benzonitrile under 760.051 Torr; for 10h; Reflux; | 66% |
-
-
42333-78-8
2,4,6-tri(4-pyridyl)-1,3,5-triazine

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane for 24h; Reflux; | 65% |

-
-
42333-78-8
2,4,6-tri(4-pyridyl)-1,3,5-triazine

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| In methanol; dichloromethane for 24h; Reflux; | 62% |


| Conditions | Yield |
|---|---|
| Stage #1: n-hexyllithium; porphyrin In tetrahydrofuran at -78 - 20℃; for 0.25h; Stage #2: With water In tetrahydrofuran for 0.0833333h; Stage #3: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In tetrahydrofuran | 61% |
| Stage #1: n-hexyllithium; porphyrin In tetrahydrofuran; hexane at 20℃; for 15h; Stage #2: With water In tetrahydrofuran; hexane at 20℃; for 0.25h; Stage #3: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In tetrahydrofuran; hexane at 20℃; for 0.5h; | 61% |
-
-
101-60-0
porphyrin

-
-
22843-73-8
5,10,15,20-tetrakis(4-nitrophenyl)porphyrin

-
-
22112-84-1
tetrakis(4-aminophenyl)porphyrin

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin(II) chloride hydrate In water at 80℃; for 6h; | 59.1% |
-
-
101-60-0
porphyrin

-
A
-
67066-10-8
5-monobromoporphin

-
B
-
85416-22-4
5,15-dibromoporphin

-
C
-
85416-23-5
5,10,15-tribromoporphin

| Conditions | Yield |
|---|---|
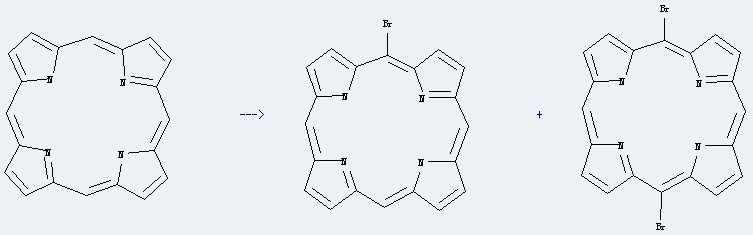
| With bromine In water; acetic acid for 2h; Product distribution; Ambient temperature; reactions of porphin with different bromination agents were investigated; | A 54% B 15% C n/a |
| With bromine In water; acetic acid for 2h; Ambient temperature; |

-
-
42333-78-8
2,4,6-tri(4-pyridyl)-1,3,5-triazine

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| Stage #1: (Cp*Rh)2(dhnq)Cl2; silver trifluoromethanesulfonate In methanol at 20℃; for 4h; Stage #2: 2,4,6-tri(4-pyridyl)-1,3,5-triazine; porphyrin In methanol for 15h; Reflux; | 52% |


| Conditions | Yield |
|---|---|
| Stage #1: n-butyllithium; porphyrin In tetrahydrofuran at 20℃; for 0.5h; Stage #2: With water In tetrahydrofuran at 20℃; for 0.25h; Stage #3: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In tetrahydrofuran at 20℃; for 30h; | 48% |

| Conditions | Yield |
|---|---|
| Stage #1: n-hexyllithium; porphyrin In tetrahydrofuran; hexane at 20℃; for 15h; Stage #2: water In tetrahydrofuran; hexane at 20℃; for 0.25h; Stage #3: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In tetrahydrofuran; hexane at 20℃; for 0.5h; | 48% |
-
-
42333-78-8
2,4,6-tri(4-pyridyl)-1,3,5-triazine

-
-
2923-28-6
silver trifluoromethanesulfonate

-
-
101-60-0
porphyrin

| Conditions | Yield |
|---|---|
| Stage #1: (η6-Cp*Ir)2(μ4-5,8-dihydroxy-1,4-naphthoquinone)Cl2; silver trifluoromethanesulfonate In methanol at 20℃; for 4h; Stage #2: 2,4,6-tri(4-pyridyl)-1,3,5-triazine; porphyrin In methanol for 15h; Reflux; | 48% |


| Conditions | Yield |
|---|---|
| Stage #1: porphyrin; phenyllithium In tetrahydrofuran at 20℃; for 0.5h; Stage #2: With water Stage #3: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In tetrahydrofuran at 20℃; for 0.5h; | A 17% B 43% |

21H,23H-Porphine Specification
The 21H,23H-Porphine, with the CAS registry number 101-60-0, is also known as Porphine. It belongs to the product categories of Porphyrins; Synthetic Porphyrins. This chemical's molecular formula is C20H14N4 and molecular weight is 310.35. What's more, its systematic name is porphyrin.
Physical properties of 21H,23H-Porphine are: (1)ACD/LogP: 3.07; (2)# of Rule of 5 Violations: 0; (3)#H bond acceptors: 4; (4)#H bond donors: 2; (5)#Freely Rotating Bonds: 0; (6)Polar Surface Area: 57.36 Å2; (7)Index of Refraction: 1.72; (8)Molar Refractivity: 90.584 cm3; (9)Molar Volume: 229.261 cm3; (10)Polarizability: 35.91 × 10-24 cm3; (11)Surface Tension: 74.658 dyne/cm; (12)Density: 1.354 g/cm3; (13)Flash Point: 377.479 °C; (14)Enthalpy of Vaporization: 112.898 kJ/mol; (15)Boiling Point: 804.44 °C at 760 mmHg; (16)Vapour Pressure: 0 mmHg at 25°C.
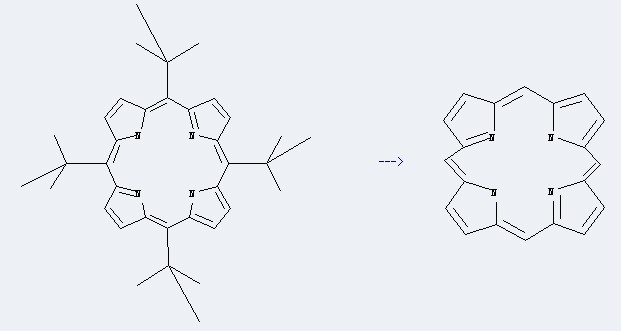
Preparation: this chemical can be prepared by meso-tetra(tert-butyl)porphyrin at the temperature of 90 °C. This reaction will need reagent conc.H2SO4 with the reaction time of 15 min. The yield is about 74%.
Uses of 21H,23H-Porphine: it can be used to produce 5-monobromoporphin and 5,15-dibromoporphin at the temperature of 2-5 °C. It will need reagent N-bromosuccinimide and solvent CHCl3 with the reaction time of 5 min. The yield is about 71%.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: c1c/2[nH]c(c1)/cc/3\nc(/cc/4\[nH]/c(c\c5n/c(c2)/C=C5)/cc4)C=C3
(2)InChI: InChI=1/C20H14N4/c1-2-14-10-16-5-6-18(23-16)12-20-8-7-19(24-20)11-17-4-3-15(22-17)9-13(1)21-14/h1-12,21,24H/b13-9-,14-10-,15-9-,16-10-,17-11-,18-12-,19-11-,20-12-
(3)InChIKey: RKCAIXNGYQCCAL-CEVVSZFKBA
Related Products
- 21H,23H-Porphine
- 21H,23H-Porphine-2,18-dipropanoic acid, 8-(1-hydroxyethyl)-13-(1-methoxyethyl)-3,7,12,17-tetramethyl-
- 21H-Biline-8,12-dipropanoicacid, 3,18-diethenyl-1,19,22,24-tetrahydro-2,7,13,17-tetramethyl-1,19-dioxo-,dimethyl ester
- 101601-00-7
- 101601-80-3
- 10160-24-4
- 1016-05-3
- 101607-39-0
- 10160-87-9
- 101-61-1
- 10161-33-8
- 10161-34-9
- 1016167-62-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View