-
Name
3,3'-Diaminobenzophenone
- EINECS 210-281-3
- CAS No. 611-79-0
- Article Data18
- CAS DataBase
- Density 1.233 g/cm3
- Solubility Insoluble in water
- Melting Point 150-151 °C
- Formula C13H12N2O
- Boiling Point 469.4 °C at 760 mmHg
- Molecular Weight 212.251
- Flash Point 237.7 °C
- Transport Information
- Appearance yellow acicular crystal
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
- Hazard Symbols
- Synonyms Benzophenone,3,3'-diamino- (6CI,7CI,8CI);3,3'-Diaminobenzophenone;NSC 113058;m,m'-Diaminobenzophenone;bis(3-aminophenyl)methanone;3,3'-Diaminobenzophenone;Bis(3-aminophenyl)methanon;Bis(3-aminophenyl)methanone;Methanone, bis(3-aminophenyl)-;
- PSA 69.11000
- LogP 3.24440
Synthetic route
-
-
21222-05-9
3,3'-dinitrobenzophenone

-
-
611-79-0
3,3'-diaminobenzophenone

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; tin(ll) chloride at 90℃; for 0.416667h; | 91% |
| With tin(ll) chloride In ethanol at 70℃; for 0.5h; | 89% |
| With hydrogenchloride; acetic acid; tin(ll) chloride |

-
-
611-79-0
3,3'-diaminobenzophenone

| Conditions | Yield |
|---|---|
| With ammonium persulfate; Montmorillonite K-10 clay for 0.0666667h; microwave irradiation; | 87% |

| Conditions | Yield |
|---|---|
| Stage #1: benzophenone With dinitrogen pentoxide In dichloromethane at 30℃; for 5h; Molecular sieve; Stage #2: With hydrogen; nickel In dichloromethane under 11251.1 Torr; for 5h; Temperature; Pressure; | 83% |
| Multi-step reaction with 2 steps 1: nitric acid 2: tin; hydrochloric acid View Scheme | |
| Multi-step reaction with 2 steps 1: nitric acid 2: tin; hydrochloric acid View Scheme | |
| Multi-step reaction with 2 steps 1: sulfuric acid; nitric acid / 2 h / 60 °C 2: hydrogenchloride; water; tin(ll) chloride / ethyl acetate / 4 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: sulfuric acid; nitric acid / 1 h / 75 °C 2: iron; ammonium chloride / ethanol; water / 4 h / 80 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: concentrated sulfuric acid / 130 - 140 °C / Reinigung durch Behandeln mit Aceton 2: glacial acetic acid; water; chromic acid 3: tin dichloride; hydrochloric acid; glacial acetic acid View Scheme | |
| Multi-step reaction with 3 steps 1: sulfuric acid / 135 °C 2: glacial acetic acid; water; chromic acid 3: tin dichloride; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: glacial acetic acid; water; chromic acid 2: tin dichloride; hydrochloric acid; glacial acetic acid View Scheme | |
| Multi-step reaction with 2 steps 1: glacial acetic acid; water; chromic acid 2: tin dichloride; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: concentrated sulfuric acid / 130 - 140 °C / Reinigung durch Behandeln mit Aceton 2: glacial acetic acid; water; chromic acid 3: tin dichloride; hydrochloric acid; glacial acetic acid View Scheme | |
| Multi-step reaction with 3 steps 1: sulfuric acid / 135 °C 2: glacial acetic acid; water; chromic acid 3: tin dichloride; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; magnesium sulfate; palladium In benzene |


-
-
611-79-0
3,3'-diaminobenzophenone

| Conditions | Yield |
|---|---|
| With hydrogen; palladium In benzene |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; palladium/active carbon In 1,4-dioxane |
-
-
83704-08-9
3,3'-dinitro-4-bromo benzophenone

-
-
611-79-0
3,3'-diaminobenzophenone

| Conditions | Yield |
|---|---|
| With hydrogen; palladium/active carbon In 1,4-dioxane |

-
-
611-79-0
3,3'-diaminobenzophenone

| Conditions | Yield |
|---|---|
| With hydrogen; sodium carbonate; palladium In ethanol |

-
-
611-79-0
3,3'-diaminobenzophenone

-
-
611-79-0
3,3'-diaminobenzophenone

| Conditions | Yield |
|---|---|
| With triethylamine |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol Reflux; | 98% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol Reflux; | 98% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol Reflux; | 97% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol Reflux; | 95% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol Reflux; | 92% |
-
-
611-79-0
3,3'-diaminobenzophenone

-
-
129993-66-4
2-hydroxy-4-[4-(decyloxy)benzoyloxy]benzaldehyde

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol for 4h; Reflux; | 90% |
-
-
611-79-0
3,3'-diaminobenzophenone

-
-
868620-29-5
4-(2`hydroxy-4`-dodecylhydroxybenzoylhydroxy)benzaldehyde

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol for 4h; Reflux; | 90% |
-
-
611-79-0
3,3'-diaminobenzophenone

-
-
389621-89-0
4-Tetradecyloxy-benzoic acid 4-formyl-3-hydroxy-phenyl ester

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol for 4h; Reflux; | 90% |
-
-
611-79-0
3,3'-diaminobenzophenone

-
-
177424-35-0
4-formyl-3-hydroxyphenyl 4-(n-hexadecanyloxy)-benzoate

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol for 4h; Reflux; | 90% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol Reflux; | 90% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol for 4h; Reflux; | 86% |
-
-
611-79-0
3,3'-diaminobenzophenone

-
-
58845-21-9
3,3'-diaminobenzhydrol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol Heating; | 72% |
| With potassium hydroxide | |
| With sodium amalgam; ethanol | |
| With sodium amalgam; ethanol |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 2h; | 62% |

-
-
611-79-0
3,3'-diaminobenzophenone

| Conditions | Yield |
|---|---|
| In ethanol Reflux; | 55% |

| Conditions | Yield |
|---|---|
| Stage #1: biotin With benzotriazol-1-ol In N,N-dimethyl-formamide at 45℃; Molecular sieve; Heating; Inert atmosphere; Stage #2: With dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 3h; Inert atmosphere; Stage #3: 3,3'-diaminobenzophenone With dmap at 20 - 60℃; for 28h; Inert atmosphere; | 53% |
-
-
19810-31-2
Benzyloxyacetyl chloride

-
-
611-79-0
3,3'-diaminobenzophenone

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 2h; | 46% |
-
-
611-79-0
3,3'-diaminobenzophenone

-
-
1111-67-7, 15192-76-4, 26656-82-6
copper(I) thiocyanate

-
-
333-20-0
potassium thioacyanate

| Conditions | Yield |
|---|---|
| Stage #1: 3,3'-diaminobenzophenone With hydrogenchloride; sodium nitrite In water at 0℃; for 0.25h; Stage #2: copper(I) thiocyanate; potassium thioacyanate In water at 20℃; for 3h; | 38% |


| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran | 23% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine; sodium t-butanolate In toluene at 100℃; for 24h; | 10.6% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; sodium nitrite at 0℃; und anschliessend mit wss.Phosphorsaeure unter starker Kuehlung und Erhitzen des mit Harnstoff versetzten Reaktionsgemisches mit Wasser; | |
| With phosphoric acid; sulfuric acid; sodium nitrite In water at 0℃; Wittig reaction; |
-
-
611-79-0
3,3'-diaminobenzophenone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; ethanol; hydroxylamine hydrochloride |
-
-
611-79-0
3,3'-diaminobenzophenone

-
-
79-04-9
chloroacetyl chloride

-
-
59635-68-6
3,3'-bis-(2-chloro-acetylamino)-benzophenone

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; acetone |

| Conditions | Yield |
|---|---|
| With sodium hydroxide man fuehrt das erhaltene methylschwefelsaure Salz mit Kaliumjodid in das entsprechende Jodid ueber und erhitzt letzteres im Vakuum; |
-
-
611-79-0
3,3'-diaminobenzophenone

-
-
333-20-0
potassium thioacyanate

-
-
53357-16-7
Bis-(5-amino-2-thiocyanato-phenyl)-methanone

| Conditions | Yield |
|---|---|
| With bromine |
-
-
611-79-0
3,3'-diaminobenzophenone

-
-
71969-93-2
C26H24N6

| Conditions | Yield |
|---|---|
| With hydrazine hydrate; zinc(II) chloride |

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid; sodium nitrite 1.) 30 min; 2.) toluene, reflux; Yield given. Multistep reaction; |
3,3'-Diaminobenzophenone Specification
The 3,3'-Diaminobenzophenone, with the CAS registry number 611-79-0 and EINECS registry number 210-281-3, has the systematic name of bis(3-aminophenyl)methanone. And the molecular formula of this chemical is C13H12N2O. It belongs to the following product categories: Benzophenones (for High-Performance Polymer Research); Functional Materials; Reagent for High-Performance Polymer Research.
The physical properties of 3,3'-Diaminobenzophenone are as following: (1)ACD/LogP: 1.92; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.91; (4)ACD/LogD (pH 7.4): 1.92; (5)ACD/BCF (pH 5.5): 16.63; (6)ACD/BCF (pH 7.4): 16.83; (7)ACD/KOC (pH 5.5): 259.41; (8)ACD/KOC (pH 7.4): 262.55; (9)#H bond acceptors: 3; (10)#H bond donors: 4; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 23.55 Å2; (13)Index of Refraction: 1.673; (14)Molar Refractivity: 64.51 cm3; (15)Molar Volume: 172.1 cm3; (16)Polarizability: 25.57×10-24cm3; (17)Surface Tension: 59.7 dyne/cm; (18)Density: 1.233 g/cm3; (19)Flash Point: 237.7 °C; (20)Enthalpy of Vaporization: 73.18 kJ/mol; (21)Boiling Point: 469.4 °C at 760 mmHg; (22)Vapour Pressure: 5.51E-09 mmHg at 25°C.
Preparation of 3,3'-Diaminobenzophenone: It can be prepared by 3,3'-dinitro-benzophenone. The reaction will need reagent alcoholic ammonium sulfide.
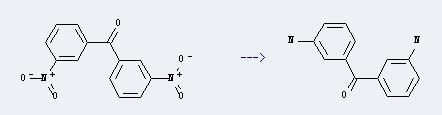
You should be cautious while dealing with this chemical. It irritates eyes, respiratory system and skin. Therefore, you had better take the following instructions: Wear suitable protective clothing, and in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(c1cc(N)ccc1)c2cccc(N)c2
(2)InChI: InChI=1/C13H12N2O/c14-11-5-1-3-9(7-11)13(16)10-4-2-6-12(15)8-10/h1-8H,14-15H2
(3)InChIKey: TUQQUUXMCKXGDI-UHFFFAOYAF
Related Products
- 3,10-Diaminotricyclo(5.2.1.0(sup 2,6))decane
- 3,10-Dinitrophenanthrene
- 3-((10-ETHYL-11-(p-HYDROXYPHENYL)DIBENZ-(B,F)OXEPIN-3-YL)OXY)-1,2-PROPANEDIOL HYDRATE (4:1)
- 3-(1,1,2,2-Tetrafluoroethoxy)aniline
- 3-(1,1,2,2-Tetrafluoroethoxy)benzaldehyde
- 3-(1,1,2,2-Tetrafluoroethoxy)bromobenzene
- 3-(1,1,2,2-Tetrafluoroethoxy)toluene
- 3-[1,1'-Biphenyl]-4-yl-1,2,3,4-tetrahydro-1-naphthol
- 3,11-Dichloro-6,11-dihydro-6-methyldibenzo[c,f][1,2]thiazepine 5,5-dioxide
- 3-[(1,1-Dimethyl-2-hydroxyethyl)amino]-2-hydroxypropanesulfonicacid
- 6117-91-5
- 61183-61-7
- 61189-99-9
- 61190-10-1
- 611-91-6
- 611-92-7
- 6119-47-7
- 611-94-9
- 611-95-0
- 6119-53-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View