-
Name
perylene-3,4,9,10-tetracarboxylic acid
- EINECS 201-343-0
- CAS No. 81-32-3
- Article Data22
- CAS DataBase
- Density 1.739 g/cm3
- Solubility
- Melting Point
- Formula C24H12O8
- Boiling Point 912.4 °C at 760 mmHg
- Molecular Weight 428.354
- Flash Point 519.4 °C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 3,4,9,10-Tetracarboxyperylene;NSC 89768;
- PSA 149.20000
- LogP 4.53000
Synthetic route
-
-
128-69-8
perylene-3,4,9,10-tetracarboxylic acid 3,4:9,10-dianhydride

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water for 12h; Heating; | 99% |
| Stage #1: perylene-3,4,9,10-tetracarboxylic acid 3,4:9,10-dianhydride With potassium hydroxide In water at 20℃; for 6h; Stage #2: With hydrogenchloride In water pH=Ca. 7; | 80% |
| With sodium hydroxide |
-
-
118153-98-3
1,6,7,12-tetrachloroperylene-3,4,9,10-tetracarboxylic acid

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethylene glycol at 155 - 160℃; for 4h; | 91% |
-
-
107905-43-1
1,7-dibromoperylene-3,4,9,10-tetracarboxylic acid

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethylene glycol at 120℃; for 1h; | 82% |

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With alkaline sodium hypochlorite | |
| With chromium(III) oxide; sulfuric acid |
-
-
857785-32-1
3,10-diamino-dibenzo[cd,lm]perylene-1,8-dione

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium permanganate; sulfuric acid at 20℃; | |
| With sodium dichromate; sulfuric acid at 20℃; |
-
-
38250-80-5
C32H18O

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With nitric acid at 165 - 190℃; |
-
-
21020-04-2
1-Benzoyloxy-peropyren

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With nitric acid at 180 - 200℃; |
-
-
7664-93-9
sulfuric acid

-
-
81-33-4
3,4,9,10-perylene tetracarboxyxlic acid diimide

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| at 200℃; | |
| at 200℃; |

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid at 200℃; |
-
-
7664-93-9
sulfuric acid

-
-
857785-32-1
3,10-diamino-dibenzo[cd,lm]perylene-1,8-dione

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|


| Conditions | Yield |
|---|---|

-
-
7664-93-9
sulfuric acid

-
-
857785-32-1
3,10-diamino-dibenzo[cd,lm]perylene-1,8-dione

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|

-
-
118129-58-1
1,7-dichloroperylene-3,4,9,10-tetracarboxylic acid

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 59 percent / Cl2, 3percent oleum / 9 h / 70 - 75 °C 2: 91 percent / KOH / ethane-1,2-diol / 4 h / 155 - 160 °C View Scheme |

| Conditions | Yield |
|---|---|
| With sodalime at 450 - 500℃; stream of superheated steam; | 96% |
| bei der trocknen Destillation des Calciumsalzes; | |
| With potassium hydroxide at 250℃; beim Erhitzen des Kaliumsalzes im Autoklaven; |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
100-36-7
N,N-diethylethylenediamine

-
-
73528-90-2
N,N'-bis(2-(N'',N''-diethylamino)ethyl)perylene-3,4:9,10-bis(dicarboximide)

| Conditions | Yield |
|---|---|
| In water Heating; | 88% |
| 1.) DMF, 145 deg C - 150 deg C, 1 h, 2.) 125 deg C - 140 deg C, 2 h; Yield given. Multistep reaction; |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
100-44-7
benzyl chloride

-
-
698998-98-0
perylene 3,4,9,10-tetracarboxylic acid tetrabenzyl ester

| Conditions | Yield |
|---|---|
| With dmap; 18-crown-6 ether; potassium carbonate In N,N-dimethyl-formamide at 80℃; for 12h; | 86% |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
79750-27-9
1,12-sulfonyloxyperylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| 85% |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
109-73-9
N-butylamine

-
-
52000-75-6
2,9-dibutylanthra[2,1,9-def:6,5,10-d'e'f']diisoquinoline-1,3,8,10(2H,9H)-tetraone

| Conditions | Yield |
|---|---|
| With 1H-imidazole at 120℃; for 4h; | 80% |
-
-
6346-09-4
4-aminobutyrylaldehyde diethylacetal

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
1446681-44-2
N,N'-bis(4,4-diethoxybutyl)perylene-3,4,9,10-tetracarboxylic diimide

| Conditions | Yield |
|---|---|
| With 1H-imidazole at 120℃; for 4h; | 78% |
-
-
24131-32-6
3,5-bis(benzyloxy)benzyl bromide

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With dmap; 18-crown-6 ether; potassium carbonate In N,N-dimethyl-formamide at 85℃; for 12h; | 76% |

| Conditions | Yield |
|---|---|
| With dmap; 18-crown-6 ether; potassium carbonate In N,N-dimethyl-formamide at 85℃; for 12h; | 72% |

| Conditions | Yield |
|---|---|
| In water at 20℃; for 0.5h; | 72% |

-
-
74654-07-2
2-[2-(2-methoxyethoxy)ethoxy]ethylamine

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| With 1H-imidazole; zinc diacetate at 160℃; for 18h; Inert atmosphere; | 47% |
-
-
546-68-9
titanium(IV) isopropylate

-
-
66-71-7
1,10-Phenanthroline

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

| Conditions | Yield |
|---|---|
| In toluene at 40℃; for 144h; Inert atmosphere; Sealed tube; | 38% |

-
-
956092-70-9
(2S,3S)-3-amino-4-{4,4’-dimethoxytrityloxy}butan-2-ol

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
75-31-0
isopropylamine

-
-
1260145-35-4
C52H42N2O8

| Conditions | Yield |
|---|---|
| Stage #1: (2S,3S)-3-amino-4-{4,4’-dimethoxytrityloxy}butan-2-ol; perylene-3,4,9,10-tetracarboxylic acid With zinc diacetate; triethylamine In pyridine for 24h; Molecular sieve; Reflux; Stage #2: isopropylamine With zinc diacetate; triethylamine In pyridine for 24h; Molecular sieve; Reflux; | 28% |

-
-
1402226-20-3
7-amino-1,12-tridecadiene

-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
1402226-21-4
N,N'-di(1,12-tridecadiene-7-yl)perylene-3,4,9,10,tetracarboxylic acid bisimide

| Conditions | Yield |
|---|---|
| With zinc diacetate In quinoline at 230℃; for 5h; Inert atmosphere; | 12% |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
141-43-5
ethanolamine

-
-
80689-49-2
N-hydroxyethyl-3,4:9,10-perylenetetracarboxylic-3,4-anhydride-9,10-imide

| Conditions | Yield |
|---|---|
| Stage #1: perylene-3,4,9,10-tetracarboxylic acid; ethanolamine In water at 0℃; for 5h; Reflux; Stage #2: With sulfuric acid In water at 90℃; for 0.5h; | 10% |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
593-51-1
methylamine hydrochloride

-
-
5521-31-3
N,N'-Dimethylperylene-3,4,9,10-biscarboximide

| Conditions | Yield |
|---|---|
| at 180 - 200℃; |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
81-33-4
3,4,9,10-perylene tetracarboxyxlic acid diimide

| Conditions | Yield |
|---|---|
| at 200 - 230℃; durch Erhitzen des Tetraammoniumsalzes mit Ammoniumcarbonat; |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
128-69-8
perylene-3,4,9,10-tetracarboxylic acid 3,4:9,10-dianhydride

| Conditions | Yield |
|---|---|
| With acetic anhydride | |
| With sodium hydroxide | |
| With sulfuric acid at 20℃; for 12h; |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
106-47-8
4-chloro-aniline

-
-
2379-77-3
3,4,9,10-perylenetetracarboxylic acid N,N'-di(4-chlorophenyl)diimide

| Conditions | Yield |
|---|---|
| at 100 - 120℃; |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
62-53-3
aniline

-
-
128-65-4
2,9-diphenylanthra[2,1,9-def:6,5,10-d'e'f']diisoquinoline-1,3,8,10(2H,9H)-tetraone

| Conditions | Yield |
|---|---|
| With acetic acid at 100 - 120℃; | |
| at 100 - 120℃; |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
-
108-00-9
N,N-dimethylethylenediamine

-
-
73528-89-9
2,9-bis(2-(dimethylamino)ethyl)anthra[2,1,9-def:6,5,10-d′e′f ′]-diisoquinoline-1,3,8,10(2H,9H)-tetraone

| Conditions | Yield |
|---|---|
| 1.) DMF, 145 deg C - 150 deg C, 1 h, 2.) 125 deg C - 140 deg C, 2 h; Yield given. Multistep reaction; |
-
-
81-32-3
perylene-3,4,9,10-tetracarboxylic acid

-
A
-
198-55-0
PERYLENE

-
B
-
7350-88-1
Perylene-3-carboxylic acid

| Conditions | Yield |
|---|---|
| at 190 - 250℃; im Autoklaven; |

3,4,9,10-Perylenetetracarboxylicacid Specification
The 3,4,9,10-Perylenetetracarboxylicacid, with the CAS registry number 81-32-3, is also known as tri-n-Propylamine. Its EINECS registry number is 201-343-0. This chemical's molecular formula is C24H12O8 and molecular weight is 428.34728. What's more, both its IUPAC name and systematic name are the same which is called Perylene-3,4,9,10-tetracarboxylic acid.
Physical properties about 3,4,9,10-Perylenetetracarboxylicacid are: (1)ACD/LogP: 3.46; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -2.46; (4)ACD/LogD (pH 7.4): -2.68; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 8; (10)#H bond donors: 4; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 105.2 Å2; (13)Index of Refraction: 1.939; (14)Molar Refractivity: 118.02 cm3; (15)Molar Volume: 246.2 cm3; (16)Surface Tension: 112.5 dyne/cm; (17)Density: 1.739 g/cm3; (18)Flash Point: 519.4 °C; (19) Enthalpy of Vaporization: 138.97 kJ/mol; (20)Boiling Point: 912.4 °C at 760 mmHg; (21)Vapour Pressure: 0 mmHg at 25 °C.
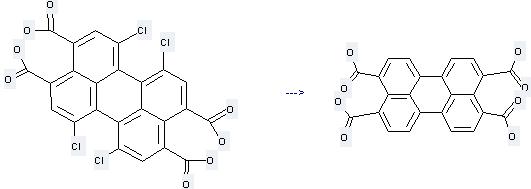
Preparation of 3,4,9,10-Perylenetetracarboxylicacid: this chemical can be prepared by 1,6,7,12-Tetrachloroperylene-3,4,9,10-tetracarboxylic acid. This reaction needs reagent KOH and solvent Ethane-1,2-diol at temperature of 155 - 160 °C. The reaction time is 4 hours. The yield is 91 %.
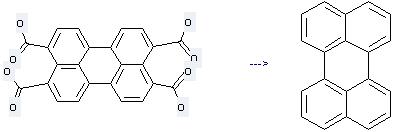
Uses of 3,4,9,10-Perylenetetracarboxylicacid: it is used to produce other chemicals. For example, it is used to produce Perylene. The reaction occurs with reagent KOH-solution at temperature of 250 °C and other condition of beim Erhitzen des Kaliumsalzes im Autoklaven.
You can still convert the following datas into molecular structure:
(1) SMILES: O=C(O)c3c5c2c(c1ccc(c4c1c(c2cc3)ccc4C(=O)O)C(=O)O)ccc5C(=O)O
(2) InChI: InChI=1/C24H12O8/c25-21(26)13-5-1-9-10-2-6-15(23(29)30)20-16(24(31)32)8-4-12(18(10)20)11-3-7-14(22(27)28)19(13)17(9)11/h1-8H,(H,25,26)(H,27,28)(H,29,30)(H,31,32)
(3) InChIKey: FVDOBFPYBSDRKH-UHFFFAOYAV
Related Products
- 3,10-Diaminotricyclo(5.2.1.0(sup 2,6))decane
- 3,10-Dinitrophenanthrene
- 3-((10-ETHYL-11-(p-HYDROXYPHENYL)DIBENZ-(B,F)OXEPIN-3-YL)OXY)-1,2-PROPANEDIOL HYDRATE (4:1)
- 3-(1,1,2,2-Tetrafluoroethoxy)aniline
- 3-(1,1,2,2-Tetrafluoroethoxy)benzaldehyde
- 3-(1,1,2,2-Tetrafluoroethoxy)bromobenzene
- 3-(1,1,2,2-Tetrafluoroethoxy)toluene
- 3-[1,1'-Biphenyl]-4-yl-1,2,3,4-tetrahydro-1-naphthol
- 3,11-Dichloro-6,11-dihydro-6-methyldibenzo[c,f][1,2]thiazepine 5,5-dioxide
- 3-[(1,1-Dimethyl-2-hydroxyethyl)amino]-2-hydroxypropanesulfonicacid
- 81323-58-2
- 81325-81-7
- 81329-90-0
- 81-33-4
- 81334-34-1
- 81335-37-7
- 81335-47-9
- 81335-77-5
- 81336-74-5
- 81336-78-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View