-
Name
3,5-Dichloroaniline
- EINECS 210-948-9
- CAS No. 626-43-7
- Article Data68
- CAS DataBase
- Density 1.401 g/cm3
- Solubility water: 0.6 g/L (26 ºC)
- Melting Point 49 ºC
- Formula C6H5Cl2N
- Boiling Point 260.6 ºC at 760 mmHg
- Molecular Weight 162.018
- Flash Point 118.3 ºC
- Transport Information UN 3442 6.1/PG 2
- Appearance white to brown crystals
- Safety 28-36/37-45-60-61
- Risk Codes 23/24/25-33-50/53
-
Molecular Structure
-
Hazard Symbols
 T,
T, N,
N, Xi
Xi
- Synonyms Aniline,3,5-dichloro- (7CI,8CI);3,5-Dichlorobenzenamine;3,5-Dichlorophenylamine;
- PSA 26.02000
- LogP 3.15680
Synthetic route
-
-
39943-56-1
3,5-dichlorophenylhydrazine

-
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| aluminum nickel In methanol; water | 100% |

| Conditions | Yield |
|---|---|
| With hydrogen | 100% |


| Conditions | Yield |
|---|---|
| With stannous chloride In aniline | 100% |

| Conditions | Yield |
|---|---|
| aluminium bromide (AlBr3) In dodecane; water | 99.5% |

| Conditions | Yield |
|---|---|
| palladium/charcoal In tantalum; hydrogen | 99% |

| Conditions | Yield |
|---|---|
| A 97.9% B n/a |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; potassium iodide | 96.8% |

| Conditions | Yield |
|---|---|
| With C36H56Cl3CrN2O; magnesium; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In tetrahydrofuran at 60℃; for 24h; Inert atmosphere; chemoselective reaction; | 96% |
| With hydrogen In methanol; ethyl acetate at 60℃; for 0.333333h; Flow reactor; Green chemistry; chemoselective reaction; | 93% |
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; 1,10-Phenanthroline; isopropyl alcohol; potassium hydroxide at 100℃; for 15h; Inert atmosphere; Schlenk technique; | 81% |

| Conditions | Yield |
|---|---|
| palladium/charcoal In tantalum | 94% |

| Conditions | Yield |
|---|---|
| With ammonium sulfate; bis[chloro(1,2,3-trihapto-allylbenzene)palladium(II)]; ammonia; sodium t-butanolate; tert-butyl XPhos In 1,4-dioxane at 100℃; for 24h; Reagent/catalyst; Sealed tube; Inert atmosphere; | 93% |
| With lithium amide; ammonia at -50℃; | |
| With ammonia |

-
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With D-glucose; potassium hydroxide In water at 85℃; for 0.166667h; Green chemistry; chemoselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: benzo-phenone imine; 1,3,5-trichlorobenzene With sodium t-butanolate; tris-(dibenzylideneacetone)dipalladium(0); 1,1'-bis(diphenylphosphino) ferrocene at 136℃; for 3h; Heating / reflux; Stage #2: With hydrogenchloride In water; xylene at 70℃; for 0.75h; | 88% |
-
-
1228378-57-1
2-azido-N-(3,5-dichlorophenyl)-N-methyl-2-phenylacetamide

-
A
-
83490-71-5
N‑methyl‑2‑oxo‑2‑phenylacetamide

-
B
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With phosphate potassium salt; oxygen; copper diacetate In N,N-dimethyl-formamide at 80℃; under 760.051 Torr; for 9h; | A 53% B 29% |
-
-
19752-55-7
1-bromo-3,5-dichlorobenzene

-
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With dicyclohexyl(2',4',6'-triisopropyl-5-methoxy-3,4,6-trimethyl-[1,1'-biphenyl]-2-yl)phosphine; C50H70NO4PPdS; C50H70NO4PPdS; dicyclohexyl(2',4',6'-triisopropyl-4-methoxy-3,5,6-trimethyl-[1,1'-biphenyl]-2-yl)phosphine; ammonia; sodium t-butanolate In 1,4-dioxane at 100℃; for 24h; Inert atmosphere; | 48% |
-
-
611-06-3
2,4-dichloronitrobenzene

-
A
-
60930-56-5
3,3',5,5'-tetrachloroazoxybenzene

-
B
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With pyridine; hydrogen sulfide |

| Conditions | Yield |
|---|---|
| With sodium hypobromide |
-
-
85011-63-8
β-D-galactopyranosylmethyl 3,5-dichlorophenyltriazene

-
A
-
13964-15-3
2,6-anhydro-L-glycero-L-galacto-heptitol

-
B
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With Mg(2+)-free lacZ-β-galactosidase; sodium EDTA buffer at 25℃; Rate constant; |

-
A
-
81972-21-6
(2S,3R,4R,5R,6R)-2-(2-Hydroxy-ethyl)-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol

-
B
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With Mg(2+)-free lacZ-β-galactosidase; sodium EDTA buffer at 25℃; Rate constant; |
-
-
85013-24-7
n-propyl-3,5-dichlorophenyltriazene

-
A
-
103322-03-8
Propane-1-diazonium

-
B
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With potassium chloride; tris hydrochloride In acetonitrile at 25℃; different pH-s, other reagents; |
-
-
119421-08-8
(3,5-Dichloro-phenylsulfinamoyl)-acetic acid tert-butyl ester

-
A
-
104311-66-2
sodium t-butoxycarbonylmethanesulphinate

-
B
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With potassium chloride In ethanol; water at 19.9℃; Rate constant; Mechanism; pH 9.30; |
-
-
62584-33-2
3-(3,5-dichloro-phenyl)-ureidoacetic acid

-
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With Pseudomonas paucimobilis; water In various solvent(s) at 30℃; for 200h; pH=6.5; Kinetics; Further Variations:; Reagents; Hydrolysis; Microbiological reaction; |
-
-
36734-19-7
Iprodione

-
A
-
62584-33-2
3-(3,5-dichloro-phenyl)-ureidoacetic acid

-
B
-
75-31-0
isopropylamine

-
C
-
626-43-7
3,5-Dichloroaniline

-
D
-
27387-87-7
3-(3',5'-Dichlorophenyl)imidazolidine-2,4-dione

| Conditions | Yield |
|---|---|
| With Pseudomonas fluoerescens; water In various solvent(s) at 30℃; for 200h; pH=6.5; Kinetics; Further Variations:; Reagents; Hydrolysis; Microbiological reaction; |

-
-
152723-53-0, 152723-54-1
3-(3,5-dichlorophenyl)-5-methyl-5-vinyloxazolidine-2,4-dione

-
A
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With buffer pH 4.5 at 35℃; Product distribution; |

-
-
152723-53-0, 152723-54-1
3-(3,5-dichlorophenyl)-5-methyl-5-vinyloxazolidine-2,4-dione

-
A
-
34893-92-0
3,4-dichlorophenyl isocyanate

-
B
-
626-43-7
3,5-Dichloroaniline

-
C
-
24261-46-9
3-(3,5-Dichlorophenyl)-5-methyl-5-methyl-oxazolidine-2,4-dione

| Conditions | Yield |
|---|---|
| humic acids In water for 8.33333h; Kinetics; Product distribution; Further Variations:; Solvents; Catalysts; wave length; Decomposition; UV-irradiation; |

-
-
152723-53-0, 152723-54-1
3-(3,5-dichlorophenyl)-5-methyl-5-vinyloxazolidine-2,4-dione

-
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| In methanol; water for 0.166667h; Decomposition; UV-irradiation; formation of xenobiotics; |
-
-
152723-53-0, 152723-54-1
3-(3,5-dichlorophenyl)-5-methyl-5-vinyloxazolidine-2,4-dione

-
A
-
2150-88-1
methyl N-(3-chlorophenyl)carbamate

-
B
-
51422-77-6
(3-methoxyphenyl)carbamic acid methyl ester

-
C
-
83015-12-7
methyl N-(3,5-dimethoxyphenyl)carbamate

-
D
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| In methanol; water for 0.166667h; Kinetics; Product distribution; Further Variations:; Solvents; Catalysts; time; wave length; Decomposition; UV-irradiation; |


-
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| With phosphoric acid; sulfuric acid; acetic acid anschliessend mit NaHPO2 und Cu2O in H2O; |
-
-
79-04-9
chloroacetyl chloride

-
-
626-43-7
3,5-Dichloroaniline

-
-
33560-48-4
2-chloro-N-(3,5-dichlorophenyl)acetamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 25℃; | 100% |
| With triethylamine In dichloromethane at 0 - 25℃; | 100% |
| With triethylamine In dichloromethane at 20℃; for 5h; | 99% |

| Conditions | Yield |
|---|---|
| In dichloromethane | 100% |

| Conditions | Yield |
|---|---|
| indium(III) triflate In acetonitrile at 50℃; for 0.191667h; Product distribution / selectivity; Microwave irradiation; | 100% |
| montmorillonit KSF In acetonitrile at 50℃; for 0.191667h; Product distribution / selectivity; Microwave irradiation; | 79% |
| ytterbium(III) triflate In acetonitrile at 50℃; for 0.191667h; Product distribution / selectivity; Microwave irradiation; | 77% |

-
-
100124-06-9
4-dibenzofurylboronic acid

-
-
626-43-7
3,5-Dichloroaniline

-
-
298-12-4
Glyoxilic acid

-
-
745013-41-6
(dibenzofuran-4-yl)-(3,5-dichlorophenylamino)acetic acid

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; | 100% |


| Conditions | Yield |
|---|---|
| 100% |
-
-
598-21-0
2-Bromoacetyl bromide

-
-
626-43-7
3,5-Dichloroaniline

-
-
57339-11-4
2-bromo-N-(3,5-dichlorophenyl)acetamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; for 1.25h; Inert atmosphere; | 100% |
| In dichloromethane at 20℃; for 0.666667h; | 65% |
| With triethylamine In dichloromethane at 0 - 20℃; for 3h; |
-
-
105-36-2
ethyl bromoacetate

-
-
626-43-7
3,5-Dichloroaniline

-
-
501008-39-5
ethyl 2-((3,5-dichlorophenyl)amino)acetate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In acetonitrile at 80℃; for 16h; | 100% |
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 90℃; for 24h; | 88% |
| In ethanol Reflux; | |
| With potassium carbonate Reflux; |
-
-
626-43-7
3,5-Dichloroaniline

-
-
98-74-8
4-Nitrobenzenesulfonyl chloride

-
-
296275-16-6
4-nitro-benzenesulfonic acid-(3,5-dichloro-anilide)

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; | 99% |
| With pyridine | |
| With pyridine |
-
-
933-88-0
ortho-toluoyl chloride

-
-
626-43-7
3,5-Dichloroaniline

-
-
97146-53-7
1-(3,5-dichlorophenyl)-3-(o-tolyl)urea

| Conditions | Yield |
|---|---|
| Stage #1: ortho-toluoyl chloride With pyridine; trimethylsilylazide In N,N-dimethyl-formamide at 20℃; Curtius rearrangement; Microflow reaction; Inert atmosphere; Stage #2: 3,5-Dichloroaniline With acetic acid In N,N-dimethyl-formamide at 110℃; Microflow reaction; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With ammonium formate In water; isopropyl alcohol at 20℃; for 3h; | 99% |
| With Raney aluminium-nickel alloy; water; sodium hydroxide In dimethoxymethane at 25℃; for 17h; |
-
-
124-38-9
carbon dioxide

-
-
626-43-7
3,5-Dichloroaniline

-
-
75-56-9, 16033-71-9
methyloxirane

-
A
-
57-55-6
propylene glycol

| Conditions | Yield |
|---|---|
| With C24H25N4O3(1+)*I(1-); 1,8-diazabicyclo[5.4.0]undec-7-ene at 90℃; under 3750.38 Torr; for 4h; Autoclave; | A n/a B 99% |

-
-
858235-96-8
(4-chloro-quinazolin-2-yl)-(3-piperidino-propyl)-amine

-
-
626-43-7
3,5-Dichloroaniline

-
-
76005-31-7
N4-(3,5-Dichloro-phenyl)-N2-(3-piperidin-1-yl-propyl)-quinazoline-2,4-diamine; hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In isopropyl alcohol for 3h; Heating; | 98% |
-
-
1402920-06-2
2-methylamino-4-mexylamino-6-chloro-1,3,5-triazine

-
-
626-43-7
3,5-Dichloroaniline

-
-
1402920-22-2
C18H18Cl2N6

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 72h; Reflux; | 98% |
-
-
532-55-8
Benzoyl isothiocyanate

-
-
626-43-7
3,5-Dichloroaniline

-
-
425660-07-7
N-benzoyl-N’-(3,5-dichlorophenyl)thiourea

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 60 - 65℃; for 0.166667h; Microwave irradiation; | 98% |
| In acetone at 40 - 50℃; | 92% |
| In acetone | |
| In acetone |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: acetic anhydride; 3,5-Dichloroaniline With acetic acid at 12 - 40℃; Large scale; Stage #2: With nitric acid at 12℃; Large scale; | 97.8% |
-
-
911681-37-3
tert-butyl 4-(2-chloroquinazolin-4-ylamino)piperidine-1-carboxylate

-
-
626-43-7
3,5-Dichloroaniline

| Conditions | Yield |
|---|---|
| Stage #1: tert-butyl 4-(2-chloroquinazolin-4-ylamino)piperidine-1-carboxylate; 3,5-Dichloroaniline In 1,4-dioxane at 120℃; for 10h; Stage #2: With potassium carbonate In water | 97% |
-
-
108-31-6
maleic anhydride

-
-
626-43-7
3,5-Dichloroaniline

-
-
55198-61-3
(Z)-4-((3,5-dichlorophenyl)amino)-4-oxobut-2-enoic acid

| Conditions | Yield |
|---|---|
| In diethyl ether at 20℃; for 2.5h; | 97% |
| With acetic acid at 20℃; for 0.166667h; |
-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
626-43-7
3,5-Dichloroaniline

-
-
219499-66-8
4-(3,5-dichloroanilino)-2,6-dichloro-1,3,5-triazine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In acetone at -20 - 0℃; for 2h; | 96% |
3,5-Dichloroaniline Specification
The 3,5-Dichloroaniline, with the CAS registry number 626-43-7, is also known as Benzenamine, 3,5-dichloro-. It belongs to the product categories of Anilines, Aromatic Amines and Nitro Compounds; Amines; C2 to C6; Nitrogen Compounds; Alpha Sort; Aromatics Pesticides & Metabolites; Chemical Class; D; DAlphabetic; DIA - DIC. Its EINECS registry number is 210-948-9. This chemical's molecular formula is C6H5Cl2N and molecular weight is 162.02. What's more, its IUPAC name is the same with its product name.
Physical properties about 3,5-Dichloroaniline are: (1)ACD/LogP: 2.70; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.7; (4)ACD/LogD (pH 7.4): 2.7; (5)ACD/BCF (pH 5.5): 66.78; (6)ACD/BCF (pH 7.4): 66.85; (7)ACD/KOC (pH 5.5): 704.04; (8)ACD/KOC (pH 7.4): 704.71; (9)#H bond acceptors: 1; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 3.24 Å2; (13)Index of Refraction: 1.613; (14)Molar Refractivity: 40.27 cm3; (15)Molar Volume: 115.6 cm3; (16)Surface Tension: 48.3 dyne/cm; (17)Density: 1.401 g/cm3; (18)Flash Point: 118.3 °C; (19)Enthalpy of Vaporization: 49.83 kJ/mol; (20)Boiling Point: 260.6 °C at 760 mmHg; (21)Vapour Pressure: 0.0121 mmHg at 25 °C.
Preparation of 3,5-Dichloroaniline: this chemical can be prepared by 1,3,5-Trichlorobenzene.
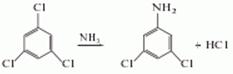
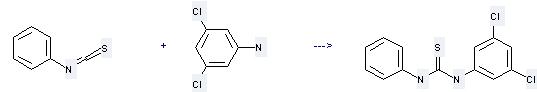
Uses of 3,5-Dichloroaniline: (1) this chemical is used as intermediate of pesticide and pharmaceutical. It is also used as a raw material for agricultural pesticides. It can also be used for synthetic herbicides, plant growth regulator; (2) it is used to produce other chemicals. For example, it can react with Isothiocyanatobenzene to get N-(3,5-Dichloro-phenyl)-N'-phenyl-thiourea. The reaction occurs with solvent ethanol and other condition of heating for 2 hours. The yield is 75 %.
When you are dealing with this chemical, you should be very careful. This chemical may present an immediate or delayed danger to one or more components of the environment. It is toxic by inhalation, in contact with skin and if swallowed. It is very toxic to aquatic organisms and may cause long-term adverse effects in the aquatic environment. So you should wear suitable protective clothing and gloves. This material and its container must be disposed of as hazardous waste and you should avoid releasing to the environment. In case of accident or if you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
(1) SMILES: Clc1cc(N)cc(Cl)c1
(2) InChI: InChI=1S/C6H5Cl2N/c7-4-1-5(8)3-6(9)2-4/h1-3H,9H2
(3) InChIKey: UQRLKWGPEVNVHT-UHFFFAOYSA-N
Related Products
- 3,10-Diaminotricyclo(5.2.1.0(sup 2,6))decane
- 3,10-Dinitrophenanthrene
- 3-((10-ETHYL-11-(p-HYDROXYPHENYL)DIBENZ-(B,F)OXEPIN-3-YL)OXY)-1,2-PROPANEDIOL HYDRATE (4:1)
- 3-(1,1,2,2-Tetrafluoroethoxy)aniline
- 3-(1,1,2,2-Tetrafluoroethoxy)benzaldehyde
- 3-(1,1,2,2-Tetrafluoroethoxy)bromobenzene
- 3-(1,1,2,2-Tetrafluoroethoxy)toluene
- 3-[1,1'-Biphenyl]-4-yl-1,2,3,4-tetrahydro-1-naphthol
- 3,11-Dichloro-6,11-dihydro-6-methyldibenzo[c,f][1,2]thiazepine 5,5-dioxide
- 3-[(1,1-Dimethyl-2-hydroxyethyl)amino]-2-hydroxypropanesulfonicacid
- 6264-40-0
- 626-44-8
- 6264-66-0
- 6264-67-1
- 6264-69-3
- 626-48-2
- 6264-98-8
- 6265-05-0
- 6265-07-2
- 6265-16-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View