-
Name
3,5-Dimethoxyaniline
- EINECS 233-616-5
- CAS No. 10272-07-8
- Article Data36
- CAS DataBase
- Density 1.096 g/cm3
- Solubility insoluble in water
- Melting Point 54-57 ºC
- Formula C8H11NO2
- Boiling Point 178 ºC (20 mmHg)
- Molecular Weight 153.181
- Flash Point 135 ºC
- Transport Information UN 2811
- Appearance White crystal or Colorless liquid
- Safety 26-36-36/37/39-22
- Risk Codes 22-36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms 3,5-Dimethoxy-phenylamine;3,5-Dimethoxybenzeneamine;Benzenamine, 3,5-dimethoxy-;3,5-dimethoxy aniline;5-Aminoresorcinol dimethylether;
- PSA 44.48000
- LogP 1.86720
Synthetic route
-
-
85862-68-6
1-azido-3,5-dimethoxybenzene

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; palladium In water for 1.5h; Inert atmosphere; | 99% |
| With sodium sulfide; water for 0.5h; Reflux; | 96% |
| With ammonium hydroxide at 90℃; for 0.5h; | 90% |
| With triethylamine; tris[tert-butyl]phosphonium tetrafluoroborate In tetrahydrofuran; water at 20℃; for 15h; Staudinger Azide Reduction; | 94 %Spectr. |
-
-
85657-94-9
N-(3,5-dimethoxyphenyl)-2,2,2-trifluoroacetamide

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With water; potassium carbonate In methanol for 20h; Ambient temperature; | 94% |
-
-
106018-88-6
2-Trimethylsilanyl-ethanesulfonic acid (3,5-dimethoxy-phenyl)-amide

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With cesium fluoride In N,N-dimethyl-formamide at 95℃; for 40h; | 93% |
-
-
7051-16-3
1-chloro-3,5-dimethoxybenzene

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; potassium phosphate In dimethyl sulfoxide at 80℃; UV-irradiation; | 93% |
| With ammonium hydroxide; potassium phosphate; copper(l) iodide; N1,N2-bis(5-methyl-[1,1'-biphenyl]-2-yl)oxalamide In water; dimethyl sulfoxide at 110℃; for 24h; Inert atmosphere; | 88% |
| With ammonium hydroxide; potassium phosphate; copper(l) iodide; N1,N2-bis(5-methyl-[1,1'-biphenyl]-2-yl)oxalamide In dimethyl sulfoxide at 120℃; for 24h; Inert atmosphere; Schlenk technique; | 88% |
-
-
79257-61-7
N-(3,5-dimethoxy-phenyl)-acetamide

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Stage #1: N-(3,5-dimethoxy-phenyl)-acetamide With Schwartz's reagent In tetrahydrofuran at 20℃; for 0.05h; Inert atmosphere; Stage #2: With water In tetrahydrofuran Inert atmosphere; chemoselective reaction; | 86% |
| With alkaline solution |
-
-
192182-54-0
3,5-dimethoxyphenylboronic acid

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydroxylamine-O-sulfonic acid In acetonitrile at 20℃; for 16h; | 82% |
-
-
20469-65-2
1-bromo-3,5-dimethoxybenzene

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Stage #1: 1-bromo-3,5-dimethoxybenzene With magnesium In tetrahydrofuran Inert atmosphere; Stage #2: With C10H17NO In tetrahydrofuran; toluene at -78℃; for 2h; Inert atmosphere; Stage #3: With ammonium chloride In tetrahydrofuran; water; toluene Inert atmosphere; | 78% |
| With ammonium hydroxide; copper(l) iodide; N,N'-bis(3,5-dimethoxyphenyl)cyclopentane-1,1-dicarboxamide; caesium carbonate In dimethyl sulfoxide at 90℃; for 24h; Inert atmosphere; Sealed tube; | 68% |
| With ammonia; sodium; ferric nitrate |
-
-
25245-27-6
1-iodo-3,5-dimethoxybenzene

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; copper(l) iodide; N,N'-bis(3,5-dimethoxyphenyl)cyclopentane-1,1-dicarboxamide; caesium carbonate In dimethyl sulfoxide at 20℃; for 24h; Inert atmosphere; Sealed tube; | 75% |
-
-
500-99-2
3,5-dimethoxyphenol

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Stage #1: 3,5-dimethoxyphenol With Nonafluorobutanesulfonyl fluoride; tris(trimethylsilyl)amine; potassium hexamethylsilazane In tetrahydrofuran at 60℃; for 5h; Schlenk technique; Inert atmosphere; Stage #2: With hydrogenchloride In water at 20℃; for 0.0833333h; pH=3 - 4; Reagent/catalyst; Solvent; | 75% |
-
-
1132-21-4
3,5-dimethoxybenzoic acid

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Stage #1: 3,5-dimethoxybenzoic acid With diphenylphosphoranyl azide; triethylamine In toluene at 80℃; Stage #2: With sodium hydroxide In tetrahydrofuran; toluene at 20℃; for 2h; | 43% |
| Stage #1: 3,5-dimethoxybenzoic acid With diphenylphosphoranyl azide; triethylamine In toluene at 80℃; Stage #2: With sodium hydroxide In tetrahydrofuran for 2h; | 43% |
-
-
710311-79-8
N-hydroxy-3,5-dimethoxybenzamide

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Stage #1: N-hydroxy-3,5-dimethoxybenzamide With acetic anhydride; potassium carbonate In dimethyl sulfoxide at 50℃; for 2h; Lossen Rearrangement; Stage #2: With hydrogenchloride In water; dimethyl sulfoxide at 0℃; | 3% |
| Stage #1: N-hydroxy-3,5-dimethoxybenzamide With potassium carbonate In dimethyl sulfoxide at 90℃; for 3h; Lossen Rearrangement; Stage #2: With hydrogenchloride In water; dimethyl sulfoxide at 20℃; for 0.0833333h; |
-
-
17715-69-4
1-Bromo-2,4-dimethoxybenzene

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With ammonia; sodium amide in fluessiges NH3; |
-
-
16147-07-2
1,3-dimethoxy-5-nitrobenzene

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin | |
| With hydrogenchloride; tin; tin(ll) chloride at 40℃; Reagens 5: Essigsaeure; | |
| With ethanol; nickel Hydrogenation; | |
| With hydrogen; palladium on activated charcoal In ethyl acetate |
-
-
17213-58-0
3,5-dimethoxybenzamide

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With sodium hypobromide at -15℃; | |
| Yield given; | |
| With sodium hypochlorite; sodium hydroxide at 80℃; for 5h; | 4.1 g |
-
-
17275-82-0
ethyl 3,5-dimethoxybenzoate

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 78 percent / hydrazine hydrate / 2 h / Heating 2: 63 percent / sodium nitrite, hydrochloric acid (2M), acetic acid (glacial) / H2O / 1 h / -10 °C 3: 93 percent / toluene / 2.5 h / Heating 4: 8.7 g / sodium hydroxide (20percent) / 60 °C View Scheme |
-
-
99-10-5
3,5-Dihydroxybenzoic acid

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 84 percent / sodium hydroxide / heating View Scheme | |
| Multi-step reaction with 2 steps 1.1: potassium carbonate / acetone / 55 °C 2.1: triethylamine; diphenylphosphoranyl azide / toluene / 80 °C 2.2: 2 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: potassium carbonate / acetone / 55 °C 2.1: triethylamine; diphenylphosphoranyl azide / toluene / 80 °C 2.2: 2 h View Scheme |
-
-
75996-26-8
azido(3,5-dimethoxyphenyl)methanone

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 93 percent / toluene / 2.5 h / Heating 2: 8.7 g / sodium hydroxide (20percent) / 60 °C View Scheme | |
| Multi-step reaction with 2 steps 1: CH2Cl2 / 60 h / Heating 2: 94 percent / H2O, K2CO3 / methanol / 20 h / Ambient temperature View Scheme |
-
-
51707-38-1
3,5-dimethoxy-benzoic acid hydrazide

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 63 percent / sodium nitrite, hydrochloric acid (2M), acetic acid (glacial) / H2O / 1 h / -10 °C 2: 93 percent / toluene / 2.5 h / Heating 3: 8.7 g / sodium hydroxide (20percent) / 60 °C View Scheme |
-
-
17213-57-9
3,5-dimethoxybenzoyl chloride

-
-
10272-07-8
3,5-Dimethoxyaniline
-
-
54132-76-2
3,5-dimethoxyphenyl isocyanate

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 8.7 g / sodium hydroxide (20percent) / 60 °C View Scheme | |
| With benzoic acid; sodium hydroxide In tetrahydrofuran at 20℃; for 0.5h; Curtius Rearrangement; |
-
-
20734-67-2
3,5-dihydroxyphenylamine

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: aqueous dioxane 2: aqueous NaOH 3: aq.-ethanolic alkaline solution View Scheme |
-
-
73164-69-9
N-(3,5-dihydroxyphenyl)acetamide

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aqueous NaOH 2: aq.-ethanolic alkaline solution View Scheme |
-
-
2150-37-0
methyl 3,5-dimethoxybenzoate

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: liquid ammonia 2: aqueous sodium hypobromite / -15 °C View Scheme | |
| Multi-step reaction with 3 steps 1: sodium hydroxide / 1,4-dioxane / 5 h / 20 °C 2: triethylamine; diphenyl phosphoryl azide / 80 °C 3: sodium hydroxide; benzoic acid / tetrahydrofuran / 0.5 h / 20 °C View Scheme |
-
-
500-99-2
3,5-dimethoxyphenol

-
-
5150-42-5
2,3-dimethoxyphenol

-
-
1972-28-7
diethylazodicarboxylate

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With triphenylphosphine In tetrahydrofuran |

-
-
2150-44-9
1-carbomethoxy-3,5-dihydroxybenzene

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: potassium carbonate / acetonitrile / 20 °C 2: sodium hydroxide / 1,4-dioxane / 5 h / 20 °C 3: triethylamine; diphenyl phosphoryl azide / 80 °C 4: sodium hydroxide; benzoic acid / tetrahydrofuran / 0.5 h / 20 °C View Scheme |
-
-
7051-16-3
1-chloro-3,5-dimethoxybenzene

-
A
-
1445972-42-8
bis(3,5-dimethoxyphenyl)amine

-
B
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With dicyclohexyl(2',4',6'-triisopropyl-5-methoxy-3,4,6-trimethyl-[1,1'-biphenyl]-2-yl)phosphine; C50H70NO4PPdS; C50H70NO4PPdS; dicyclohexyl(2',4',6'-triisopropyl-4-methoxy-3,5,6-trimethyl-[1,1'-biphenyl]-2-yl)phosphine; ammonia; sodium t-butanolate In 1,4-dioxane at 50℃; for 24h; Inert atmosphere; Overall yield = 81 %; |

| Conditions | Yield |
|---|---|
| With phosphoric acid; ammonia In ethylene glycol at 145℃; for 5h; | A 95 %Spectr. B 84 %Spectr. |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
384793-29-7
(3,5-dimethoxyphenyl)-carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; Inert atmosphere; | 100% |
| Stage #1: 3.5-dimethoxyaniline With triethylamine In dichloromethane at 20℃; Stage #2: di-tert-butyl dicarbonate In dichloromethane at 20℃; | 99% |
| at 90℃; for 3h; | 98% |
-
-
79-22-1
methyl chloroformate

-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
83015-12-7
methyl N-(3,5-dimethoxyphenyl)carbamate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; for 2h; Inert atmosphere; | 100% |
| With pyridine In dichloromethane at 0 - 20℃; for 2h; Inert atmosphere; | 94% |
| In toluene for 2h; Acylation; Heating; | 81% |
-
-
190728-25-7
4-((6,7-dimethoxyquinolin-4-yl)oxy)aniline

-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
144-55-8
sodium hydrogencarbonate

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With triethylamine In toluene | 100% |

-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
920-46-7
Methacryloyl chloride

-
-
134354-28-2
N-(3,5-dimethoxyphenyl)-2-methylacrylamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; | 100% |
| In dichloromethane at 0 - 20℃; | |
| In dichloromethane at 0 - 20℃; | |
| In dichloromethane at 0 - 20℃; | |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; |
-
-
598-21-0
2-Bromoacetyl bromide

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; for 1.25h; Inert atmosphere; | 100% |
| at -77℃; |

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In tetrahydrofuran at 20℃; Inert atmosphere; | 99.9% |
-
-
58518-08-4
1,1,1-trifluoro-4-phenylbut-3-yn-2-one

-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
129633-19-8
(Z)-4-(3,5-Dimethoxy-phenylamino)-1,1,1-trifluoro-4-phenyl-but-3-en-2-one

| Conditions | Yield |
|---|---|
| In methanol Ambient temperature; | 99% |
-
-
403-29-2
2-bromo-4'-fluoroacetophenone

-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
161139-70-4
2-[(3,5-dimethoxyphenyl)amino]-1-(4-fluorophenyl)ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In neat (no solvent) at 20℃; for 2h; | 99% |
| With sodium hydrogencarbonate In ethanol for 4h; Heating; |
-
-
81051-55-0, 53256-20-5
(Z)-ethyl 3-(methylamino)-3-phenylacrylate

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane for 30h; Heating; | 99% |
-
-
84761-77-3
2-formylphenyl triflate

-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
111043-16-4
1,3-dimethoxyacridine

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate; XPhos In toluene at 105℃; for 13h; Mechanism; Reagent/catalyst; Solvent; Temperature; Schlenk technique; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With acetic acid; zinc at 20 - 60℃; for 2.5h; | 99% |
-
-
174265-12-4
2-bromo-5-chlorobenzaldehyde

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Stage #1: 3.5-dimethoxyaniline With 1,1'-bis-(diphenylphosphino)ferrocene; tris-(dibenzylideneacetone)dipalladium(0); potassium carbonate In toluene at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: 2-bromo-5-chlorobenzaldehyde In toluene at 80℃; for 12h; Inert atmosphere; | 99% |
-
-
84459-32-5
2-bromo-5-nitrobenzaldehyde

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Stage #1: 3.5-dimethoxyaniline With 1,1'-bis-(diphenylphosphino)ferrocene; tris-(dibenzylideneacetone)dipalladium(0); potassium carbonate In toluene at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: 2-bromo-5-nitrobenzaldehyde In toluene at 80℃; for 12h; Inert atmosphere; | 99% |
-
-
360575-28-6
2-bromo-6-fluorobenzaldehyde

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Stage #1: 3.5-dimethoxyaniline With 1,1'-bis-(diphenylphosphino)ferrocene; tris-(dibenzylideneacetone)dipalladium(0); potassium carbonate In toluene at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: 2-bromo-6-fluorobenzaldehyde In toluene at 80℃; for 12h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 3.5-dimethoxyaniline With 1,1'-bis-(diphenylphosphino)ferrocene; tris-(dibenzylideneacetone)dipalladium(0); potassium carbonate In toluene at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: ortho-bromobenzaldehyde In toluene at 80℃; for 12h; Reagent/catalyst; Solvent; Inert atmosphere; | 99% |
| Stage #1: 3.5-dimethoxyaniline With 1,1'-bis-(diphenylphosphino)ferrocene; tris-(dibenzylideneacetone)dipalladium(0); potassium carbonate In toluene at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: ortho-bromobenzaldehyde In toluene at 80℃; for 12h; Reagent/catalyst; | 99% |
-
-
20469-65-2
1-bromo-3,5-dimethoxybenzene

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With chloro[(tri-tert-butylphosphine)-2-(2-aminobiphenyl)]palladium (II); sodium t-butanolate In tetrahydrofuran at 70℃; for 24h; Inert atmosphere; Schlenk technique; | 99% |
| With chloro[(tri-tert-butylphosphine)-2-(2-aminobiphenyl)]palladium (II); sodium t-butanolate In tetrahydrofuran at 60℃; for 19h; Inert atmosphere; | 81% |
| With chloro[(tri-tert-butylphosphine)-2-(2-aminobiphenyl)]palladium (II); sodium t-butanolate In tetrahydrofuran at 60℃; Inert atmosphere; | 75% |
-
-
40891-33-6
3,5-dimethoxyaniline hydrochloride

-
-
119-52-8
4,4'-dimethoxybenzoin

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| at 130 - 140℃; for 1h; | 99% |

-
-
869294-86-0
ethyl 6-iodo-4-oxo-3,4-dihydroquinazoline-2-carboxylate

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| Stage #1: 3.5-dimethoxyaniline With trimethylaluminum In n-heptane; 1,2-dichloro-ethane at 0 - 20℃; for 0.5h; Stage #2: ethyl 6-iodo-4-oxo-3,4-dihydroquinazoline-2-carboxylate In n-heptane; 1,2-dichloro-ethane for 2h; Reflux; | 99% |

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); sodium carbonate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In water; toluene for 4h; Reflux; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In isopropyl alcohol at 20 - 90℃; for 16h; Inert atmosphere; | 98.4% |
-
-
108-24-7
acetic anhydride

-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
79257-61-7
N-(3,5-dimethoxy-phenyl)-acetamide

| Conditions | Yield |
|---|---|
| With pyridine at 0 - 20℃; | 98% |
| In toluene at 20℃; for 15h; Time; Inert atmosphere; | 98% |
| In toluene at 25℃; for 20h; | 97% |
-
-
485-47-2
indan-1,2,3-trione hydrate

-
-
10272-07-8
3,5-Dimethoxyaniline

| Conditions | Yield |
|---|---|
| In acetic acid for 16h; Ambient temperature; | 98% |

| Conditions | Yield |
|---|---|
| With titanium(IV) isopropylate; MS 4 Angstroem; palladium diacetate; triphenylphosphine In benzene at 80℃; for 3h; Alkylation; N-allylation; | 98% |
-
-
106-38-7
para-bromotoluene

-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
255835-89-3
N-(4-tolyl)-3,5-dimethoxyaniline

| Conditions | Yield |
|---|---|
| With C60H86O4P2Pd2(2+)*2BF4(1-); sodium t-butanolate In 1,4-dioxane at 80℃; for 1h; | 98% |
| With sodium t-butanolate; 1,1'-bis(diphenylphosphino)ferrocene; tris(dibenzylideneacetone)palladium In toluene at 90℃; for 5h; |
-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
407-25-0
trifluoroacetic anhydride

-
-
85657-94-9
N-(3,5-dimethoxyphenyl)-2,2,2-trifluoroacetamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane for 1h; | 98% |
| With triethylamine In dichloromethane at 0 - 20℃; for 2.5h; | 98% |
| With triethylamine In toluene at 15 - 20℃; for 1.5h; Temperature; Solvent; | 85.4% |
| With triethylamine In toluene at 18 - 25℃; for 2h; | |
| With triethylamine In dichloromethane at 20℃; for 2.5h; Cooling with ice; |
-
-
383-63-1
ethyl trifluoroacetate,

-
-
10272-07-8
3,5-Dimethoxyaniline

-
-
85657-94-9
N-(3,5-dimethoxyphenyl)-2,2,2-trifluoroacetamide

| Conditions | Yield |
|---|---|
| With dmap In tetrahydrofuran for 48h; Heating / reflux; | 98% |
| With dmap In tetrahydrofuran for 48h; Reflux; | 98% |
3,5-Dimethoxyaniline Chemical Properties
Molecular Weight:153.18
Appearance:Beige crystals
Solubility in water:Soluble
Melting Point:66 – 68
Boiling Point:303
3,5-Dimethoxyaniline`s Synonyms:BENZENAMINE, 3,5-DIMETHOXY-;3,5-DIMETHOXYANILINE;3,5-DIMETHOXYPHENYLAMINE;5-AMINORESORCINOL DIMETHYL ESTER;5-AMINORESORCINOL DIMETHYL ETHER;AKOS 91392;3,5-dimethoxy-benzenamin;3,5-dimethoxy-Benzenamine
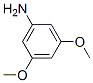
Molecular Structure:
3,5-Dimethoxyaniline Toxicity Data With Reference
3,5-Dimethoxyaniline Consensus Reports
First Aid Measures:
Ingestion: If victim is conscious and alert, give 2-4 cupfuls of milk or water. Never give anything by mouth to an unconscious person. Get medical aid immediately.
Inhalation: Remove from exposure to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical ai
Skin: Get medical aid. Flush skin with plenty of soap and water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately.
Storage:Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Handling:Wash thoroughly after handling. Use only in a well ventilated area. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
3,5-Dimethoxyaniline Safety Profile
Risk Statements: 22-36/37/38-20/21/22
Safety Statements: 26-36-36/37/39-22
WGK Germany: 3
Hazard Note: Irritant
Risk Phrases:
R 20/21/22 Harmful by inhalation, in contact with skin and if swallowed.
R 36/37/38 Irritating to eyes, respiratory system and skin.
Safety Phrases:
S 22 Do not breathe dust.
S 26 In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S 36/37/39 Wear suitable protective clothing, gloves and eye/face protection.
3,5-Dimethoxyaniline Specification
Fire Fighting: Wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. To extinguish fire, use water fog, dry chemical, carbon dioxide, or regular foam.
Personal Protection: Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166. Skin: Wear appropriate protective gloves to prevent skin exposure. Clothing: Wear appropriate protective clothing to prevent skin exposure.
Related Products
- 3,10-Diaminotricyclo(5.2.1.0(sup 2,6))decane
- 3,10-Dinitrophenanthrene
- 3-((10-ETHYL-11-(p-HYDROXYPHENYL)DIBENZ-(B,F)OXEPIN-3-YL)OXY)-1,2-PROPANEDIOL HYDRATE (4:1)
- 3-(1,1,2,2-Tetrafluoroethoxy)aniline
- 3-(1,1,2,2-Tetrafluoroethoxy)benzaldehyde
- 3-(1,1,2,2-Tetrafluoroethoxy)bromobenzene
- 3-(1,1,2,2-Tetrafluoroethoxy)toluene
- 3-[1,1'-Biphenyl]-4-yl-1,2,3,4-tetrahydro-1-naphthol
- 3,11-Dichloro-6,11-dihydro-6-methyldibenzo[c,f][1,2]thiazepine 5,5-dioxide
- 3-[(1,1-Dimethyl-2-hydroxyethyl)amino]-2-hydroxypropanesulfonicacid
- 102728-64-3
- 1027-28-7
- 102735-53-5
- 102735-84-2
- 102735-90-0
- 1027375-80-9
- 10273-90-2
- 102743-85-1
- 102747-84-2
- 102748-61-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View