-
Name
3-Carboxyphenylboronic acid
- EINECS -0
- CAS No. 25487-66-5
- Article Data18
- CAS DataBase
- Density 1.406 g/cm3
- Solubility 2.5 g/100 mL in water
- Melting Point 243-247 °C(lit.)
- Formula C7H7BO4
- Boiling Point 434.512 °C at 760 mmHg
- Molecular Weight 165.941
- Flash Point 216.585 °C
- Transport Information
- Appearance off-white crystalline powder
- Safety 37/39-26-36
- Risk Codes 36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, Xn
Xn
- Synonyms Benzoic acid, m-borono- (6CI,7CI,8CI);Benzoic acid, 3-borono- (9CI);(3-formyloxyphenyl)boronic acid;Benzeneboronic acid, m-carboxy-;m-Carboxyphenylboronic acid;3-16-00-01283 (Beilstein Handbook Reference);m-Carboxybenzeneboronic acid;3-Boronobenzoic acid;3-hydroxyphenylboronic acid;
- PSA 77.76000
- LogP -0.93540
Synthetic route
-
-
269409-73-6
3-carboxyphenylboronic acid pinacol ester

-
-
13061-96-6
dihydroxy-methyl-borane

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane at 20℃; | 99% |
-
-
150255-96-2
(3‐cyanophenyl)boronic acid

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: (3‐cyanophenyl)boronic acid With potassium hydroxide In ethylene glycol at 175℃; for 3h; Stage #2: With hydrogenchloride In water; ethylene glycol pH=2 - 3; | 89% |
-
-
7722-64-7
potassium permanganate

-
-
17933-03-8
m-tolylboronic acid

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 30 - 40℃; Temperature; | 61.2% |
-
-
13675-18-8
tetrahydroxydiboron

-
-
99-05-8
meta-aminobenzoic acid

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: meta-aminobenzoic acid With hydrogenchloride; sodium nitrite In water at 0℃; for 0.25h; Stage #2: tetrahydroxydiboron With sodium acetate In water at 20℃; for 0.333333h; | 52.5% |
| Stage #1: meta-aminobenzoic acid With hydrogenchloride; sodium nitrite In water at 0℃; for 0.25h; Stage #2: tetrahydroxydiboron With potassium carbonate In water at 20℃; for 0.333333h; |

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-Diiodobenzene With n-butyllithium In diethyl ether; hexane at -70℃; Inert atmosphere; Stage #2: With Triisopropyl borate In diethyl ether; hexane at -70℃; for 0.25h; Inert atmosphere; Stage #3: carbon dioxide Further stages; | 50% |
-
-
13675-18-8
tetrahydroxydiboron

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; for 0.25h; | 39% |
-
-
17933-03-8
m-tolylboronic acid

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With alkaline aqueous potassium permanganate | |
| With potassium permanganate In sodium hydroxide | |
| With permanganate(VII) ion In sodium hydroxide oxidation in alkaline soln.;; | |
| With permanganate In sodium hydroxide aq. NaOH; oxidation in alkaline soln.;; |
-
-
535-80-8
3-chlorobenzoate

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrahydroxydiboron; (chloro(2-dicyclohexylphosphino-2′,4′,6′-triisopropyl-1,1′-biphenyl)[2-(2′-amino-1,1′-biphenyl)]palladium(II)); potassium acetate; XPhos In ethanol at 80℃; for 5h; Inert atmosphere; |
-
-
99-05-8
meta-aminobenzoic acid

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: ethanol; water / 0.03 h / 20 °C 1.2: 1.25 h / 0 - 20 °C 2.1: N,N-dimethyl-formamide / 0.25 h / 20 °C View Scheme |
-
-
124-38-9
carbon dioxide

-
-
98527-70-9
tris(3-bromo phenyl)boroxine

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: tris(3-bromo phenyl)boroxine With iodine; magnesium In tetrahydrofuran at 40 - 70℃; Inert atmosphere; Stage #2: carbon dioxide In tetrahydrofuran at -20℃; | 110 g |
-
-
1899-24-7
5-bromo-2-furancarboxaldehyde

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
304884-54-6
5-(3-carboxyphenyl)furane-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetrakis(triphenylphosphine) palladium(0) In ethanol; toluene at 90℃; for 7h; Suzuki-Miyaura coupling; | 100% |
| With C11H8Cl4N2O3Pd; potassium carbonate In methanol; water; N,N-dimethyl-formamide at 100℃; for 0.0833333h; Suzuki Coupling; | 98% |
| With ethylenediaminetetraacetic acid; tetrabutylammomium bromide; potassium carbonate; palladium dichloride at 50℃; for 0.166667h; Suzuki-Miyaura reaction; | 92% |
-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
586-78-7
para-nitrophenyl bromide

-
-
729-01-1
4'-nitrobiphenyl-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetrakis(triphenylphosphine) palladium(0) In ethanol; water; toluene for 48h; Suzuki Coupling; Heating / reflux; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene for 48h; Suzuki Coupling; Reflux; Inert atmosphere; | 96.1% |
| Stage #1: 3-Carboxyphenylboronic acid; para-nitrophenyl bromide With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene for 48h; Suzuki coupling; Inert atmosphere; Reflux; Stage #2: With hydrogenchloride In water pH=3; | 89% |

| Conditions | Yield |
|---|---|
| With diisopropyl-carbodiimide In methanol at 0 - 20℃; for 24.5h; | 100% |
| With diisopropyl-carbodiimide In methanol at 0 - 20℃; for 24.5h; |
-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
74-11-3
para-chlorobenzoic acid

| Conditions | Yield |
|---|---|
| With sodium 2'‐(dicyclohexylphosphaneyl)‐2,6‐diisopropyl‐[1,1'‐biphenyl]‐3‐sulfonate; palladium diacetate; potassium carbonate In water at 50℃; for 8h; Suzuki-Miyaura coupling; | 99% |
-
-
1010413-06-5
1,1-dimethylethyl 1-[(5-bromo-2-methylphenyl)methyl]-3-[4-(1,1-dimethylethyl)phenyl]-1H-indole-2-carboxylate

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
1010413-07-6
3'-({2-{[(1,1-dimethylethyl)oxy]carbonyl}-3-[4-(1,1-dimethylethyl)phenyl]-1H-indol-1-yl}methyl)-4'-methyl-3-biphenylcarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1,1-dimethylethyl 1-[(5-bromo-2-methylphenyl)methyl]-3-[4-(1,1-dimethylethyl)phenyl]-1H-indole-2-carboxylate; 3-Carboxyphenylboronic acid With sodium hydrogencarbonate; palladium 10% on activated carbon In water; N,N-dimethyl-formamide at 90℃; for 36h; Stage #2: With hydrogenchloride In water; N,N-dimethyl-formamide | 99% |
-
-
5367-28-2
5-chloro-2-nitrotoluene

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
1303573-22-9
3'-methyl-4'-nitrobiphenyl-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate; triphenylphosphine In ethanol; water; toluene at 100℃; for 24h; Suzuki coupling; Inert atmosphere; Sealed vessel; | 99% |
-
-
1355152-77-0
6-bromo-1-methyl-3-trityl-1H-imidazo[4,5-b]pyridin-2(3H)-one

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
1355152-95-2
3-(1-methyl-2-oxo-3-trityl-2,3-dihydro-1H-imidazo[4,5-b]pyridin-6-yl)benzoic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); tricyclohexylphosphine In 1,4-dioxane at 100℃; for 1h; Microwave irradiation; | 99% |
-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium hydrogencarbonate In water; acetonitrile at 100℃; Suzuki Coupling; Inert atmosphere; | 98% |
-
-
67-56-1
methanol

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
99769-19-4
[3-(methoxycarbonyl)phenyl]boronic acid

| Conditions | Yield |
|---|---|
| With thionyl chloride at 55 - 65℃; Autoclave; Industrial scale; | 97.1% |
| With sulfuric acid at 80℃; for 20h; Temperature; |

| Conditions | Yield |
|---|---|
| With ammonium bicarbonate In water at 20℃; for 2h; Schlenk technique; | 97% |

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); caesium carbonate In 1,4-dioxane; water at 90℃; for 4h; | 96% |
-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
126-30-7
2,2-Dimethyl-1,3-propanediol

| Conditions | Yield |
|---|---|
| In chloroform at 25℃; for 12h; | 96% |
| In diethyl ether at 20℃; Inert atmosphere; Molecular sieve; |
-
-
695-96-5
2-bromo-4-chlorophenol

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
376592-57-3
5'-chloro-2'-hydroxybiphenyl-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate In water at 50℃; for 4h; Inert atmosphere; | 95% |
| With palladium diacetate; sodium carbonate In water at 50℃; for 19h; Inert atmosphere; |
-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In water; acetonitrile at 160℃; for 1h; Inert atmosphere; Microwave irradiation; | 95% |
-
-
625-92-3
3,5-dibromopyridine

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
1429436-06-5
3,3’-(pyridine-3,5-diyl)dibenzoic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In acetonitrile at 90℃; for 36h; Suzuki Coupling; Inert atmosphere; | 93% |
-
-
698-67-9
p-bromobenzamide

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium carbonate In water at 100℃; for 4h; Suzuki Coupling; | 93% |
-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
269409-73-6
3-carboxyphenylboronic acid pinacol ester

| Conditions | Yield |
|---|---|
| In toluene for 12h; Reflux; Inert atmosphere; | 92% |
| In ethyl acetate for 4h; | 91% |
| With 1H-imidazole; iron(III) chloride In water; acetonitrile at 20℃; for 0.5h; | 21.5% |
| In ethyl acetate for 4h; Inert atmosphere; |
-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
99-06-9
3-Carboxyphenol

| Conditions | Yield |
|---|---|
| With copper(II) ferrite; water; sodium hydroxide at 40℃; for 24h; Green chemistry; | 92% |
| With oxygen; triethylamine In water; acetonitrile at 25℃; for 8h; UV-irradiation; | 86% |
| With oxygen; N-ethyl-N,N-diisopropylamine; [5,6]fullerene-C70 In chloroform; toluene at 20℃; for 12h; Irradiation; | 66% |
-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,4-dioxane; water at 75 - 80℃; for 2h; Suzuki Coupling; Inert atmosphere; | 92% |
-
-
13073-25-1
2-bromo-6-nitro-phenol

-
-
25487-66-5
3-Carboxyphenylboronic acid

- 2'-hydroxy-3'-nitro-biphenyl-3-carboxylic acid
-
376591-95-6
2'-hydroxy-3'-nitro-biphenyl-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; potassium carbonate In methanol; water for 6h; Inert atmosphere; Reflux; | 92% |
-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
4595-60-2
2-bromopyrimidine

-
-
579476-26-9
3-(pyrimidin-2-yl)benzoic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In acetonitrile at 90℃; for 4h; Substitution; Suzuki reaction; | 91% |
-
-
4487-59-6
2-bromo-5-nitropyridine

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
864075-95-6
3-(5-nitropyridin-2-yl)benzoic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate; tetrakis(triphenylphosphine) palladium(0) In ethanol; water; toluene for 48h; Suzuki Coupling; Heating / reflux; | 91% |
-
-
695-96-5
2-bromo-4-chlorophenol

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromo-4-chlorophenol With potassium hydroxide In 1,2-dimethoxyethane; water Inert atmosphere; Stage #2: 3-Carboxyphenylboronic acid With 5%-palladium/activated carbon; potassium carbonate In 1,2-dimethoxyethane; water at 18 - 40℃; Inert atmosphere; | 90.7% |
-
-
885066-04-6
(6-chloro-2-methoxy-pyrimidin-4-yl)-[2-(4-methoxy-phenyl)-ethyl]-amine

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: (6-chloro-2-methoxy-pyrimidin-4-yl)-[2-(4-methoxy-phenyl)-ethyl]-amine; 3-Carboxyphenylboronic acid With caesium carbonate In 1,2-dimethoxyethane; water for 0.0833333h; Stage #2: tetrakis(triphenylphosphine) palladium(0) In 1,2-dimethoxyethane; water at 90℃; for 17h; | 90.4% |
| With caesium carbonate; tetrakis(triphenylphosphine) palladium(0) In 1,2-dimethoxyethane; water at 90℃; for 17.0833h; | 90.4% |
-
-
20191-75-7
1-bromo-2-naphthylamine

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; palladium diacetate; sodium carbonate; potassium carbonate; disodium ethylenediamine tetraacetic acid In water for 16h; Reflux; Inert atmosphere; | 90.4% |
-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
1761-61-1
5-bromosalicyclaldehyde

| Conditions | Yield |
|---|---|
| With palladium bis[bis(diphenylphosphino)ferrocene] dichloride; caesium carbonate In water; N,N-dimethyl-formamide at 100℃; for 4h; | 90.2% |
-
-
626-55-1
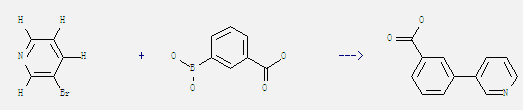
3-Bromopyridine

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
4385-77-7
3-(pyridine-3-yl)benzoic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In acetonitrile at 90℃; for 16h; Substitution; Suzuki reaction; | 90% |
| With potassium carbonate; bis-triphenylphosphine-palladium(II) chloride In water; acetonitrile Reflux; | 83% |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium carbonate In 1,2-dimethoxyethane; water at 100℃; for 4h; Suzuki Coupling; Inert atmosphere; | 70% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In acetonitrile at 90℃; for 36h; Suzuki Coupling; Inert atmosphere; | 22% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In acetonitrile at 90℃; for 14h; |

-
-
25487-66-5
3-Carboxyphenylboronic acid

-
-
1192471-48-9
cyclopentyl N-[3-(dihydroxyboryl)benzoyl]-2-methylalaninate

| Conditions | Yield |
|---|---|
| Stage #1: 3-Carboxyphenylboronic acid With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 0℃; for 0.333333h; Stage #2: cyclopentyl 2-amino-2-methylpropanoate hydrochloride In dichloromethane at 20℃; for 4h; | 90% |
-
-
22726-00-7
3-bromobenzamide

-
-
25487-66-5
3-Carboxyphenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium carbonate In water at 100℃; for 4h; Suzuki Coupling; | 90% |
3-Carboxyphenylboronic acid Specification
The IUPAC name of m-Carboxyphenylboronic acid is 3-boronobenzoic acid. With the CAS registry number 25487-66-5, it is also named as Benzeneboronic acid, m-carboxy-. The product's categories are Blocks; Boronic Acids; Carboxes; Acids and Derivatives; Boron, Nitrile, Thio & TM-Cpds; Substituted Boronic Acids; Boronic Acids & Esters; Phenyls & Phenyl-Het; Aryl; Organoborons; B (Classes of Boron Compounds); Boronic Acids and Derivatives. It is off-white crystalline powder which should be stored at the temperature of 0-6°C.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 0.62; (2)# of Rule of 5 Violations: 0; (3)ACD/BCF (pH 5.5): 1; (4)ACD/BCF (pH 7.4): 1; (5)ACD/KOC (pH 5.5): 1; (6)ACD/KOC (pH 7.4): 1; (7)#H bond acceptors: 4; (8)#H bond donors: 3; (9)#Freely Rotating Bonds: 4; (10)Polar Surface Area: 77.76 Å2; (11)Index of Refraction: 1.585; (12)Molar Refractivity: 39.564 cm3; (13)Molar Volume: 118.001 cm3; (14)Polarizability: 15.685×10-24 cm3; (15)Surface Tension: 62.794 dyne/cm; (16)Density: 1.406 g/cm3; (17)Flash Point: 216.585 °C; (18)Enthalpy of Vaporization: 72.797 kJ/mol; (19)Boiling Point: 434.512 °C at 760 mmHg; (20)Vapour Pressure: 0 mmHg at 25°C.
Uses of m-Carboxyphenylboronic acid: It can react with 3-bromo-pyridine to get 3-pyridin-3-yl-benzoic acid. This reaction which is a kind of Substitution//Suzuki reaction needs reagent aq. Na2CO3, catalytic agent Pd(PPh3)4 and solvent acetonitrile at temperature of 90 °C. The reaction time is 16 hours. The yield is 90%.
When you are using this chemical, please be cautious about it as the following:
It is not onlt harmful by inhalation, in contact with skin and if swallowed, but also irritating to eyes, respiratory system and skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If you want to contact this product, you must wear suitable protective clothing, gloves and eye/face protection.
People can use the following data to convert to the molecule structure.
1. SMILES:B(c1cccc(c1)C(=O)O)(O)O
2. InChI:InChI=1/C7H7BO4/c9-7(10)5-2-1-3-6(4-5)8(11)12/h1-4,11-12H,(H,9,10)
3. InChIKey:DBVFWZMQJQMJCB-UHFFFAOYAT
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View