-
Name
3-Chloro-1-propyne
- EINECS 210-856-9
- CAS No. 624-65-7
- Article Data34
- CAS DataBase
- Density 1.017 g/cm3
- Solubility
- Melting Point <-50 °C
- Formula C3H3Cl
- Boiling Point 57 °C at 760 mmHg
- Molecular Weight 74.5098
- Flash Point 18.9 °C
- Transport Information UN 3286 3/PG 2
- Appearance colorless flammable liquid
- Safety 16-26-36/37/39-45-27
- Risk Codes 11-23/24/25-34
-
Molecular Structure
-
Hazard Symbols
 F,
F,  T,
T,  C
C
- Synonyms Propyne,3-chloro- (6CI,8CI);1-Chloroprop-2-yne;2-Propynyl chloride;3-Chloro-1-propyne;NSC 66411;Propargyl chloride;
- PSA 0.00000
- LogP 0.85840
Synthetic route

| Conditions | Yield |
|---|---|
| With 3,5-dichlorobenzoyl chloride; potassium carbonate In N,N-dimethyl-formamide at 20℃; for 5h; Time; Reagent/catalyst; | 97.98% |

| Conditions | Yield |
|---|---|
| With thionyl chloride; tetramethylurea In toluene at 40℃; for 3.16h; | 94% |
| With benzoyl chloride; N,N-dimethyl-formamide at 0 - 20℃; for 14.25h; | 75% |
| With pyridine; bis(trichloromethyl) carbonate In tetrahydrofuran Heating; | 63% |
-
-
473-29-0
N,N-Dichlorobenzenesulfonamide

-
-
107-19-7
propargyl alcohol

-
A
-
98-10-2
benzenesulfonamide

-
B
-
80-16-0
N-chlorobenzenesulfonamide

-
C
-
624-65-7
2-propynyl chloride

| Conditions | Yield |
|---|---|
| In tetrachloromethane at 30 - 35℃; for 6h; Irradiation; | A 32% B 4% C 7 % Chromat. D 16% |


| Conditions | Yield |
|---|---|
| at 0℃; Erhitzen des erhaltenen Chlorameisensaeure-prop-2-inylesters mit Triaethylamin-hydrochlorid auf 100grad; |
-
-
4860-96-2
3-bromo-2-chloroprop-1-ene

-
A
-
10024-18-7
bromoallene

-
B
-
3223-70-9
chloroallene

-
C
-
624-65-7
2-propynyl chloride

-
D
-
106-96-7
propargyl bromide

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride; phenol Yield given; |

-
-
16400-63-8
2-bromo-3-chloro-propene

-
A
-
10024-18-7
bromoallene

-
B
-
3223-70-9
chloroallene

-
C
-
624-65-7
2-propynyl chloride

-
D
-
106-96-7
propargyl bromide

| Conditions | Yield |
|---|---|
| With sodium hydroxide; N-benzyl-N,N,N-triethylammonium chloride; phenol Yield given; |


| Conditions | Yield |
|---|---|
| With 1-chloro-1-(dimethylamino)-2-methyl-1-propene In dichloromethane 1). 0 deg C; 2.) rt., 3h; | A n/a B 99 % Spectr. |

| Conditions | Yield |
|---|---|
| With pyridine; tributyltin chloride at 50℃; Thermodynamic data; Equilibrium constant; Δ G; |

| Conditions | Yield |
|---|---|
| With pyridine; tributyltin chloride at 50℃; Thermodynamic data; Equilibrium constant; Δ G; |

| Conditions | Yield |
|---|---|
| at 661.85℃; under 125 Torr; Equilibrium constant; var. temp. and pressures; |
-
-
107-19-7
propargyl alcohol

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
624-65-7
2-propynyl chloride
-
-
624-65-7
2-propynyl chloride

-
-
290333-14-1
ethyl 4-(benzyloxy)-7-bromo-1H-indole-2-carboxylate

-
-
922507-01-5
ethyl 4-(benzyloxy)-7-bromo-1-prop-2-ynyl-1H-indole-2-carboxylate

| Conditions | Yield |
|---|---|
| With potassium carbonate; sodium iodide In N,N-dimethyl-formamide at 50℃; for 24h; | 100% |
-
-
3481-02-5
cyclopropyl phenyl ketone

-
-
624-65-7
2-propynyl chloride

-
-
91909-17-0
1-cyclopropyl-1-phenyl-3-butyn-1-ol

| Conditions | Yield |
|---|---|
| With 2,4,6-trimethyl-pyridine; bis(cyclopentadienyl)titanium dichloride; manganese; chloro-trimethyl-silane In tetrahydrofuran for 6h; Barbier type propargylation; Inert atmosphere; | 100% |
-
-
616-47-7
1-methyl-1H-imidazole

-
-
624-65-7
2-propynyl chloride

-
-
98379-44-3
1-methyl-3-(2-propyn-1-yl)-imidazolium chloride

| Conditions | Yield |
|---|---|
| In toluene Reflux; | 99% |
| In benzene for 5h; Heating; |
-
-
106-44-5
p-cresol

-
-
624-65-7
2-propynyl chloride

-
A
-
6921-27-3
dipropargyl ether

-
B
-
5651-90-1
1-methyl-4-(2-propynyloxy)benzene

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra-(n-butyl)ammonium iodide | A n/a B 99% |


| Conditions | Yield |
|---|---|
| With 2,4,6-trimethyl-pyridine; bis(cyclopentadienyl)titanium dichloride; manganese; chloro-trimethyl-silane In tetrahydrofuran at 20℃; for 7h; Barbier Coupling Reaction; Inert atmosphere; chemoselective reaction; | 99% |
-
-
112-31-2
caprinaldehyde

-
-
693-54-9
Decan-2-one

-
-
624-65-7
2-propynyl chloride

-
-
74646-37-0
tridec-1-yn-4-ol

| Conditions | Yield |
|---|---|
| With 2,4,6-trimethyl-pyridine; bis(cyclopentadienyl)titanium dichloride; manganese; chloro-trimethyl-silane In tetrahydrofuran at 20℃; for 7h; Barbier Coupling Reaction; Inert atmosphere; chemoselective reaction; | 99% |


| Conditions | Yield |
|---|---|
| In toluene Reflux; | 99% |

| Conditions | Yield |
|---|---|
| With piperazine; hydrogen In ethanol at 80℃; under 4500.45 Torr; for 24h; | 99% |
| With piperazine; hydrogen In ethanol at 100℃; under 4500.45 Torr; for 15h; Green chemistry; | |
| With hydrogen In ethanol at 100℃; under 4500.45 Torr; for 24h; chemoselective reaction; | 87 %Chromat. |
-
-
39987-25-2, 62491-02-5
dimethyl iminodiacetate hydrochloride

-
-
624-65-7
2-propynyl chloride

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 80℃; for 5h; Inert atmosphere; | 99% |
-
-
39512-49-7
4-(4-chlorophenyl)-4-hydroxypiperidine

-
-
624-65-7
2-propynyl chloride

-
-
43141-21-5
N-propargyl-4-(4'-chlorophenyl)-4-piperidinol

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide Ambient temperature; | 98% |
-
-
6118-22-5
3-vinyloxy-propan-1-ol

-
-
624-65-7
2-propynyl chloride

-
-
6188-75-6
3-(3-vinyloxy-propoxy)-propyne

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tributyl-amine In water at 50 - 60℃; for 3h; | 98% |
-
-
118017-13-3
trans-benzoic acid 4-oxo-but-2-enyl ester

-
-
624-65-7
2-propynyl chloride

-
-
213545-10-9
rac-1-benzoyloxy-7-chlorohept-2(E)-en-5-yn-4-ol

| Conditions | Yield |
|---|---|
| Stage #1: 2-propynyl chloride With n-butyllithium In diethyl ether; hexane at -78℃; for 0.166667h; Metallation; Stage #2: trans-benzoic acid 4-oxo-but-2-enyl ester In diethyl ether at -78℃; for 0.5h; Condensation; | 98% |
-
-
624-65-7
2-propynyl chloride

-
-
150-76-5
4-methoxy-phenol

-
-
17061-86-8
1-methoxy-4-(2-propynyloxy)benzene

| Conditions | Yield |
|---|---|
| Stage #1: 4-methoxy-phenol With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; Stage #2: 2-propynyl chloride In N,N-dimethyl-formamide at 0 - 20℃; | 98% |
| With potassium carbonate In N,N-dimethyl-formamide at 50℃; for 16h; | 93% |
-
-
75108-70-2
trans-PdEt2(PMe2Ph)2

-
-
624-65-7
2-propynyl chloride

-
-
1428586-22-4
trans-[ClPd(η1-allenyl)(PMe2Ph)2]

| Conditions | Yield |
|---|---|
| Stage #1: trans-PdEt2(PMe2Ph)2 With styrene In tetrahydrofuran at 55℃; for 1h; Schlenk technique; Inert atmosphere; Stage #2: 2-propynyl chloride In tetrahydrofuran at 20℃; for 3h; Inert atmosphere; Schlenk technique; | 98% |
-
-
624-65-7
2-propynyl chloride

| Conditions | Yield |
|---|---|
| With tellurium tetrachloride In benzene at 80℃; for 4h; stereospecific reaction; | 98% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.333333h; Kinetics; Inert atmosphere; regioselective reaction; | 98% |

-
-
624-65-7
2-propynyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: (S)-N-(2-formyl-benzyl-4-chlorophenyl)-1-benzyl-pyrrolidine-2-carboxamide-glycine nickel complex With sodium hydroxide In water; N,N-dimethyl-formamide at -10℃; for 0.5h; Stage #2: 2-propynyl chloride In water; N,N-dimethyl-formamide Solvent; Temperature; enantioselective reaction; | 97.8% |
| With sodium hydroxide In water; N,N-dimethyl-formamide at -10℃; for 1.5h; Solvent; | 93.87 %Chromat. |
-
-
624-65-7
2-propynyl chloride

-
-
210217-98-4
2-Fluoro-2-phenyl-2-phenylthioacetonitrile

| Conditions | Yield |
|---|---|
| With Triethylgermyl-natrium In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide 1.) -60 deg C, 0.25 h, 2.) -80 deg C, 0.5 h; | 97% |
-
-
29654-55-5
5-(hydroxymethyl)benzene-1,3-diol

-
-
624-65-7
2-propynyl chloride

-
-
176038-84-9
3,5-dipropargyloxybenzyl alcohol

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In acetone Heating / reflux; | 97% |
| With Ki; potassium carbonate In acetone | 97% |
| With potassium carbonate; potassium iodide In acetone Heating / reflux; | 97% |
| With potassium carbonate; potassium iodide In acetone Heating; | 88% |
-
-
79-77-6
4-(2,6,6-trimethyl-1-cyclohexen-1-yl)-3-buten-2-one

-
-
624-65-7
2-propynyl chloride

-
-
14168-34-4
(E)-3-Methyl-1-(2,6,6-trimethyl-cyclohex-1-enyl)-hex-1-en-5-yn-3-ol

| Conditions | Yield |
|---|---|
| With 2,4,6-trimethyl-pyridine; bis(cyclopentadienyl)titanium dichloride; manganese; chloro-trimethyl-silane In tetrahydrofuran for 6h; Barbier type propargylation; Inert atmosphere; | 97% |
-
-
624-65-7
2-propynyl chloride

-
-
300662-21-9
N-acetyl-5-methoxy-2,4,6-tribromotryptamine

| Conditions | Yield |
|---|---|
| Stage #1: N-acetyl-5-methoxy-2,4,6-tribromotryptamine With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 0.0833333h; Stage #2: 2-propynyl chloride In N,N-dimethyl-formamide at 20℃; for 4h; | 97% |

| Conditions | Yield |
|---|---|
| With oxygen; 2-hydroxyethanethiol In water at 20℃; for 24h; optical yield given as %de; stereoselective reaction; | A 5 %Chromat. B 97% |

-
-
60885-31-6
trans-diethylbis(Et3phosphine)palladium(II)

-
-
624-65-7
2-propynyl chloride

-
-
1428586-21-3
trans-[ClPd(η1-allenyl)(PEt3)2]

| Conditions | Yield |
|---|---|
| Stage #1: trans-diethylbis(Et3phosphine)palladium(II) With styrene In tetrahydrofuran at 55℃; for 1h; Schlenk technique; Inert atmosphere; Stage #2: 2-propynyl chloride In tetrahydrofuran at 20℃; for 3h; Inert atmosphere; Schlenk technique; | 97% |

-
-
624-65-7
2-propynyl chloride

| Conditions | Yield |
|---|---|
| In acetone at 20℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 60℃; for 48.5h; | 97% |
| With sodium hydrogencarbonate In ethanol at 25℃; for 3h; |

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetone at 110℃; for 2h; Microwave irradiation; | 97% |
-
-
19099-93-5
benzyl 4-oxo-1-piperidinecarboxylate

-
-
624-65-7
2-propynyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: 2-propynyl chloride With n-butyllithium In tetrahydrofuran; hexane at -78℃; Inert atmosphere; Stage #2: benzyl 4-oxo-1-piperidinecarboxylate In tetrahydrofuran; hexane at -78℃; for 6h; Inert atmosphere; | 97% |
-
-
764-48-7
2-hydroxyethyl vinyl ether

-
-
624-65-7
2-propynyl chloride

-
-
14846-79-8
ethylene glycol vinyl propargyl diether

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tributyl-amine In water at 50 - 60℃; for 3h; Product distribution; other catalyst; | 96.8% |
| With sodium hydroxide; tributyl-amine In water at 50 - 60℃; for 3h; | 96.8% |
-
-
107-21-1
ethylene glycol

-
-
624-65-7
2-propynyl chloride

-
-
40842-04-4
α,ω-bis(O-propargyl)ethylene glycol

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 29 - 32℃; for 2h; | 96% |
3-Chloropropyne Chemical Properties
IUPAC Name: 3-Chloroprop-1-yne
Molecular Formula: C3H3Cl
Molecular Weight: 74.508920 g/mol
Appearance: Colorless flammable liquid
Melting Point: <-50°C
Boiling Point: 58 °C(lit.)
Flash Point: 7 °F
Insolubility: Water, glycerol
Density: 1.03 g/mL at 25 °C(lit.)
Structure of 3-Chloropropyne (CAS NO.624-65-7):
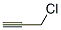
Categories of 3-Chloropropyne (CAS NO.624-65-7): Aliphatics;Halides;Acetylenes;Functionalized Acetylenes
3-Chloropropyne Uses
3-Chloropropyne (CAS NO.624-65-7) is engineering plastics modifier. It is used as intermediate in organic synthesis, and is used in the manufacture of Excellent drop-ning and soil fumigant.
3-Chloropropyne Safety Profile
Hazard Codes: F  ,T
,T  ,C
,C 
Risk Statements: 11-23/24/25-34
11:Highly Flammable
23/24/25:Toxic by inhalation, in contact with skin and if swallowed
34:Causes burns
Safety Statements: 16-26-36/37/39-45-27
16:Keep away from sources of ignition-Nosmoking
26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39:Wear suitable protective clothing, gloves and eye/face protection
45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
27:Take off immediately all contaminated clothing
A pressure-sensitive explosive. Reacts explosively with ammonia in a closed container. When heated to decomposition it emits toxic fumes of Cl−. See also ACETYLENE COMPOUNDS and ALKYNES; CHLORINATED HYDROCARBONS, ALIPHATIC; and EXPLOSIVES, HIGH; EXPLOSIVES, LOW; EXPLOSIVES, PERMITTED.
3-Chloropropyne Specification
3-Chloropropyne , its cas register number is 624-65-7. It also can be called 3-Chloro-1-propyne ; and Propargyl chloride .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View