-
Name
3-Hydroxyglutaric Acid
- EINECS
- CAS No. 638-18-6
- Article Data21
- CAS DataBase
- Density 1.508 g/cm3
- Solubility
- Melting Point 94-96°C
- Formula C5H8O5
- Boiling Point 364.958 °C at 760 mmHg
- Molecular Weight 148.116
- Flash Point 188.705 °C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 3-Hydroxyglutaric acid;b-Hydroxyglutaric acid;2,4-Dideoxypentaric acid;Glutaricacid, 3-hydroxy- (7CI,8CI);
- PSA 94.83000
- LogP -0.70330
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol for 2h; | 95% |
| With potassium hydroxide In methanol |
-
-
32328-03-3
diethyl 3-hydroxyglutarate

-
-
638-18-6
diethyl 3-hydroxypentanedioate

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water for 60h; Ambient temperature; | 84% |
| With potassium hydroxide In ethanol |

| Conditions | Yield |
|---|---|
| With sodium amalgam; water; sodium carbonate Reagens 4: Kohlendioxyd; |

| Conditions | Yield |
|---|---|
| With hydrogen sulfide; mercury(II) diacetate |
-
-
127351-38-6
3-ethoxy-pentenedioic acid diethyl ester

-
-
638-18-6
diethyl 3-hydroxypentanedioate

| Conditions | Yield |
|---|---|
| With sodium amalgam; ethanol |
-
-
77-92-9
citric acid

-
A
-
638-18-6
diethyl 3-hydroxypentanedioate

-
B
-
498-24-8
Mesaconic acid

-
C
-
499-12-7
1 propene 1,2,3 tricarboxylic acid

-
D
-
97-65-4
2-methylenesuccinic acid

-
E
-
616-02-4
citraconic acid anhydride

-
F
-
2170-03-8
itaconic acid anhydride

| Conditions | Yield |
|---|---|
| With Sn/Pb solder at 150 - 210℃; |


-
-
638-18-6
diethyl 3-hydroxypentanedioate

| Conditions | Yield |
|---|---|
| With sodium carbonate |
-
-
1724-02-3
glutaconic acid

-
-
1600-27-7
mercury(II) diacetate

-
-
7732-18-5
water

-
-
638-18-6
diethyl 3-hydroxypentanedioate

| Conditions | Yield |
|---|---|
| Behandlung des Reaktionsproduktes mit H2S in alkoh. Loesung; |


-
-
638-18-6
diethyl 3-hydroxypentanedioate

| Conditions | Yield |
|---|---|
| With barium dihydroxide |

-
-
638-18-6
diethyl 3-hydroxypentanedioate

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
64-17-5
ethanol

-
-
127351-38-6
3-ethoxy-pentenedioic acid diethyl ester

-
-
638-18-6
diethyl 3-hydroxypentanedioate


-
-
638-18-6
diethyl 3-hydroxypentanedioate

| Conditions | Yield |
|---|---|
| bei Einw.von Hundemuskelbrei; |

-
-
638-18-6
diethyl 3-hydroxypentanedioate

| Conditions | Yield |
|---|---|
| With sodium carbonate |
-
-
859306-40-4
3-bromo-glutaric acid

-
A
-
625-38-7
but-3-enoic acid

-
B
-
638-18-6
diethyl 3-hydroxypentanedioate

-
-
857792-97-3
2,3-dibromo-glutaric acid

-
A
-
638-18-6
diethyl 3-hydroxypentanedioate

-
B
-
328-50-7
α-ketoglutaric acid

-
C
-
1724-02-3
glutaconic acid

| Conditions | Yield |
|---|---|
| Prod. 5:trans-Cyclopropan-dicarbonsaeure-(1.2); Prod. 6: Cyclopropen-(2)-dicarbonsaeure-(1.2); Prod. 7: Pyromellitsaeure; |

-
-
49597-05-9
propene-1,1,3,3-tetracarboxylic acid tetraethyl ester

-
A
-
64-18-6
formic acid

-
B
-
638-18-6
diethyl 3-hydroxypentanedioate

-
C
-
141-82-2
malonic acid

-
D
-
1724-02-3
glutaconic acid

-
-
49597-05-9
propene-1,1,3,3-tetracarboxylic acid tetraethyl ester

-
A
-
64-18-6
formic acid

-
B
-
638-18-6
diethyl 3-hydroxypentanedioate

-
C
-
141-82-2
malonic acid

-
D
-
1724-02-3
glutaconic acid

-
-
7647-01-0
hydrogenchloride

-
-
49597-05-9
propene-1,1,3,3-tetracarboxylic acid tetraethyl ester

-
A
-
638-18-6
diethyl 3-hydroxypentanedioate

-
B
-
2889-31-8
2-hydroxyglutaric acid

-
C
-
1724-02-3
glutaconic acid

-
-
49597-05-9
propene-1,1,3,3-tetracarboxylic acid tetraethyl ester

-
A
-
638-18-6
diethyl 3-hydroxypentanedioate

| Conditions | Yield |
|---|---|
| With barium dihydroxide |
-
-
628-48-8
glutaconic acid

-
-
1600-27-7
mercury(II) diacetate

-
-
7732-18-5
water

-
A
-
638-18-6
diethyl 3-hydroxypentanedioate

-
B
-
1724-02-3
glutaconic acid

| Conditions | Yield |
|---|---|
| das Produkt bei 100-105grad unter Gelbfaerbung und Gewichtsverlust zersetzt und liefert mit Schwefelwasserstoff in alkalischer Loesung; |

-
-
67-56-1
methanol

-
-
542-05-2
acetonedicarboxylic acid

-
A
-
638-18-6
diethyl 3-hydroxypentanedioate

-
B
-
109462-19-3
Monomethyl 3-Hydroxypentanedioic Acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide; sodium tetrahydroborate for 0.333333h; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 54 percent / NaBH4 2: 95 percent / KOH / methanol / 2 h View Scheme | |
| Multi-step reaction with 2 steps 1: NaBH4 / methanol 2: KOH / methanol View Scheme |
-
-
7250-55-7
dimethyl 3-hydroxypentanedioate

-
A
-
638-18-6
diethyl 3-hydroxypentanedioate

-
B
-
1724-02-3
glutaconic acid

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl 3-hydroxypentanedioate With potassium hydroxide In methanol at 0 - 20℃; Stage #2: With hydrogenchloride In water |
-
-
638-18-6
diethyl 3-hydroxypentanedioate

-
-
75-36-5
acetyl chloride

-
-
98546-48-6
3-acetoxypentanedioic anhydride

| Conditions | Yield |
|---|---|
| 92% |
-
-
6066-82-6
1-hydroxy-pyrrolidine-2,5-dione

-
-
638-18-6
diethyl 3-hydroxypentanedioate

-
-
197906-35-7
3-Hydroxy-pentanedioic acid bis-(2,5-dioxo-pyrrolidin-1-yl) ester

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In 1,4-dioxane for 24h; Ambient temperature; | 92% |
-
-
638-18-6
diethyl 3-hydroxypentanedioate

-
-
118511-81-2
1-(4-piperidinyl)-1H-indole

-
-
1093881-98-1
C31H36N4O3

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In ethanol at 25℃; for 3h; | 76% |
-
-
638-18-6
diethyl 3-hydroxypentanedioate

-
-
152294-14-9
dihydro-4-hydroxy-2H-pyran-2,6(3H)-dione

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In tetrahydrofuran at 22℃; for 1h; | 54% |

| Conditions | Yield |
|---|---|
| With sulfuric acid | |
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| Destillation im Vakuum; |

| Conditions | Yield |
|---|---|
| With hydrogen iodide at 180℃; unter Druck; |

| Conditions | Yield |
|---|---|
| Destillation im Vakuum; |

| Conditions | Yield |
|---|---|
| Destillation im Vakuum; | |
| bei der Destillation im luftverduennten Raum; |

| Conditions | Yield |
|---|---|
| Destillation im Vakuum; | |
| With sodium hydroxide | |
| With sulfuric acid | |
| bei der Destillation im luftverduennten Raum; |
-
-
638-18-6
diethyl 3-hydroxypentanedioate

-
-
859306-40-4
3-bromo-glutaric acid

| Conditions | Yield |
|---|---|
| With hydrogen bromide at 100℃; | |
| With hydrogen bromide |

| Conditions | Yield |
|---|---|
| at 120℃; |

| Conditions | Yield |
|---|---|
| at 180℃; |
3-Hydroxypentanedioic acid Specification
The Pentanedioic acid,3-hydroxy-, with the CAS registry number 638-18-6, is also known as 2,4-Dideoxypentaric acid. It belongs to the product category of Aliphatics. This chemical's molecular formula is C5H8O5 and molecular weight is 148.04. What's more, both its IUPAC name and systematic name are the same which is called 3-Hydroxypentanedioic acid.
Physical properties about Pentanedioic acid,3-hydroxy- are: (1)ACD/LogP: -1.422; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -3.88; (4)ACD/LogD (pH 7.4): -6.15; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 1.00; (8)ACD/KOC (pH 7.4): 1.00; (9)#H bond acceptors: 5; (10)#H bond donors: 3; (11)#Freely Rotating Bonds: 5; (12)Polar Surface Area: 94.83 Å2; (13)Index of Refraction: 1.52; (14)Molar Refractivity: 29.836 cm3; (15)Molar Volume: 98.203 cm3; (16)Polarizability: 11.828×10-24cm3; (17)Surface Tension: 74.778 dyne/cm; (18)Density: 1.508 g/cm3; (19)Flash Point: 188.705 °C; (20)Enthalpy of Vaporization: 70.737 kJ/mol; (21)Boiling Point: 364.958 °C at 760 mmHg; (22)Vapour Pressure: 0 mmHg at 25 °C.
Preparation of Pentanedioic acid,3-hydroxy-: this chemical can be prepared by 3-hydroxy-glutaric acid dimethyl ester. This reaction needs reagent KOH and solvent methanol. The reaction time is 2 hours. The yield is 95 %.

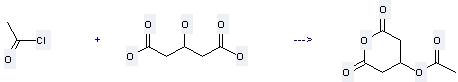
Uses of Pentanedioic acid,3-hydroxy-: it is used to produce other chemicals. For example, it can react with acetyl chloride to get 3-acetoxy-pentanedioic acid anhydride. The yield is 92 %.
You can still convert the following datas into molecular structure:
(1) SMILES: O=C(O)CC(O)CC(=O)O
(2) InChI: InChI=1S/C5H8O5/c6-3(1-4(7)8)2-5(9)10/h3,6H,1-2H2,(H,7,8)(H,9,10)
(3) InChIKey: ZQHYXNSQOIDNTL-UHFFFAOYSA-N
Related Products
- 3-Hydroxypentanedioic acid
- 6381-92-6
- 6382-01-0
- 638203-32-4
- 6382-06-5
- 638207-62-2
- 638-21-1
- 638217-08-0
- 63823-26-7
- 638-23-3
- 638-25-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View