-
Name
4-Hydroxy-3-nitrobenzaldehyde
- EINECS 221-141-6
- CAS No. 3011-34-5
- Article Data60
- CAS DataBase
- Density 1.5 g/cm3
- Solubility
- Melting Point 139-144 °C
- Formula C7H5NO4
- Boiling Point 275 °C at 760 mmHg
- Molecular Weight 167.121
- Flash Point 124.3 °C
- Transport Information
- Appearance Yellow-brown to brown powder
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Benzaldehyde, 4-hydroxy-3-nitro-;NSC 138267;AC1L2BHN;4-Formyl-2-nitrophenol;4-Formyl-2-nitro-phenolate;
- PSA 83.12000
- LogP 1.63610
Synthetic route

| Conditions | Yield |
|---|---|
| With dinitrogen tetraoxide; ferric nitrate In acetone for 2.5h; Ambient temperature; | 100% |
| With magnesium(II) nitrate hexahydrate; AMA at 20℃; for 0.583333h; Neat (no solvent); regioselective reaction; | 98% |
| With tetrachlorosilane; silica gel; sodium nitrite In dichloromethane at 20℃; for 1.5h; | 97% |
-
-
41833-13-0
4-hydroxymethyl-2-nitrophenol

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With manganese(II) nitrate hexahydrate; copper(II) nitrate trihydrate; acetic acid In α,α,α-trifluorotoluene at 50℃; for 2h; In air; chemoselective reaction; | 91% |
| With potassium hydroxide; permanganate(VII) ion at 90 - 100℃; |

| Conditions | Yield |
|---|---|
| With oxygen; cobalt(II) diacetate tetrahydrate; sodium hydroxide In ethylene glycol at 80℃; under 760.051 Torr; for 8h; | 87% |
| With oxygen; sodium hydroxide In 2-methoxy-ethanol at 80℃; for 7h; chemoselective reaction; |
-
-
123-08-0
4-hydroxy-benzaldehyde

-
A
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
B
-
90151-04-5
4-hydroxy-2-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With Nitrogen dioxide under 225.02 Torr; for 12h; | A 77% B 17% |
-
-
42564-51-2
4-fluoro-3-nitrobenzaldehyde

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With 4a,9a-dihydro-9,9-dimethyl-4,5-bis(diphenylphosphino)-xanthene; 4-methoxyphenylboronic acid; tetrabutylammomium bromide; palladium diacetate; caesium carbonate In 1,2-dimethoxyethane at 70℃; for 48h; Suzuki-Miyaura Coupling; Inert atmosphere; | 75% |
| With sodium hydroxide; N,N-dimethyl-formamide anschliessendes Behandeln mit Saeure; |
-
-
501-36-0
(E)-5-[2-4-(hydroxyphenyl)ethenyl]-1,3-benzenediol

-
C
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
D
-
123-08-0
4-hydroxy-benzaldehyde

| Conditions | Yield |
|---|---|
| With phosphate buffer; sodium nitrite In methanol at 20℃; for 3h; pH=3.0; Further byproducts given; | A 4% B 2% C 1% D 4% |

-
-
163596-75-6
4-bromo-3-nitrobenzaldehyde

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With sodium carbonate |
-
-
51818-98-5
4-acetamido-3-nitrobenzaldehyde

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydroxide |
-
-
127-08-2
potassium acetate

-
-
16588-34-4
4-chloro-3-nitro-benzaldehyde

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| at 150℃; | |
| at 150℃; |
-
-
16588-34-4
4-chloro-3-nitro-benzaldehyde

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| With potassium acetate at 140 - 150℃; | |
| With sodium carbonate at 140 - 150℃; |

| Conditions | Yield |
|---|---|
| With hexamethylenetetramine; acetic acid anschliessendes Erhitzen mit wss.HCl; |
-
-
699-06-9, 60221-52-5, 60221-53-6
4-hydroxybenzaldehyde oxime

-
A
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
B
-
26879-83-4
4-hydroxy-3-nitro-benzaldehyde-oxime

| Conditions | Yield |
|---|---|
| With nitric acid; acetic acid |
-
-
16588-34-4
4-chloro-3-nitro-benzaldehyde

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde
-
-
16588-34-4
4-chloro-3-nitro-benzaldehyde

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| at 140 - 150℃; | |
| at 140 - 150℃; |
-
-
7697-37-2
nitric acid

-
-
64-19-7
acetic acid

-
-
699-06-9, 60221-52-5, 60221-53-6
4-hydroxybenzaldehyde oxime

-
A
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde


| Conditions | Yield |
|---|---|
| man verduennt mit Wasser.Diazotization; |

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With water; nitric acid Diazotization; |
-
-
41833-13-0
4-hydroxymethyl-2-nitrophenol

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| at 90 - 100℃; |

-
-
699-06-9, 60221-52-5, 60221-53-6
4-hydroxybenzaldehyde oxime

-
A
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With nitric acid; acetic acid |
-
-
51818-98-5
4-acetamido-3-nitrobenzaldehyde

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde
-
-
22068-49-1
4-(1,3-dithiolan-2-yl)phenol

-
A
-
100-02-7
4-nitro-phenol

-
B
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
C
-
123-08-0
4-hydroxy-benzaldehyde

| Conditions | Yield |
|---|---|
| With bismuth(III) nitrate; water In benzene at 20℃; for 8h; Product distribution; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: HNO3; water; H2SO4 2: DMF; aq. NaOH solution / anschliessendes Behandeln mit Saeure View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: concentrated sulfuric acid; nitric 2: natrium carbonate View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: concentrated hydrochloric acid 2: permanganate; diluted KOH-solution / 90 - 100 °C View Scheme | |
| Multi-step reaction with 2 steps 1: formaldehyde; concentrated hydrochloric acid / Erhitzen des Reaktionsprodukts mit Wasser 2: permanganate; diluted KOH-solution / 90 - 100 °C View Scheme |
-
-
1375797-26-4
C38H42N6O10

-
A
-
288-13-1
NH-pyrazole

-
B
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
C
-
1375797-17-3
N,N-bis(2-hydroxyphenylmethyl)-N-(2-pyridylmethyl)ethane-1,2-diamine

| Conditions | Yield |
|---|---|
| With water; 2-hydroxyethanethiol; magnesium chloride; β-galactosidase at 37℃; for 3h; pH=7.2; Kinetics; aq. phosphate buffer; Enzymatic reaction; |

-
-
1375797-28-6
C41H43N7O10

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
C
-
1375797-17-3
N,N-bis(2-hydroxyphenylmethyl)-N-(2-pyridylmethyl)ethane-1,2-diamine

| Conditions | Yield |
|---|---|
| With water; 2-hydroxyethanethiol; magnesium chloride; β-galactosidase at 37℃; for 3h; pH=7.2; Kinetics; aq. phosphate buffer; Enzymatic reaction; |

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
106-95-6
allyl bromide

-
-
125872-98-2
3-nitro-4-(2-propenyloxy)benzaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 60℃; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 50℃; for 1h; | 99% |
| With potassium carbonate; potassium iodide In acetonitrile for 20h; Inert atmosphere; Reflux; | 97% |
| With potassium carbonate In acetonitrile at 60℃; for 72h; Inert atmosphere; | 92% |
-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
75-03-6
ethyl iodide

-
-
132390-61-5
4-ethoxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 20 - 70℃; | 99% |
| With potassium carbonate In acetonitrile for 15h; Heating; | 82% |
| With potassium carbonate In N,N-dimethyl-formamide |
-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
107-21-1
ethylene glycol

-
-
697306-05-1
4-(1,3-dioxolan-2-yl)-2-nitrophenyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| Stage #1: trifluoromethylsulfonic anhydride; 4-hydroxy-3-nitrobenzaldehyde With triethylamine In dichloromethane at 0℃; for 0.666667h; Stage #2: ethylene glycol With methanesulfonic acid In toluene for 14h; Heating / reflux; | 99% |

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
41833-13-0
4-hydroxymethyl-2-nitrophenol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 0℃; for 0.5h; | 98% |
| With sodium tetrahydroborate In ethanol at 0 - 20℃; for 3h; | 97% |
| With sodium tetrahydroborate In methanol at 0℃; for 1h; Inert atmosphere; | 80% |
-
-
13057-17-5
bromethyl methyl ether

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
105183-11-7
3-nitro-(4-methoxymethoxy)benzaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 0℃; for 0.5h; | 98% |
-
-
14384-45-3
N,N'-dihydroxy-2,3-dimethylbutane-2,3-diamine

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
1169932-83-5
2-(4-hydroxy-3-nitrophenyl)-4,4,5,5-tetramethylimidazolidine-1,3-diol

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 3h; | 98% |
-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
3082-95-9, 72692-06-9, 73464-50-3
2,3,4-Tri-O-acetyl-β-D-glucopyranuronic acid methylester

-
-
148579-93-5
(2S,3R,4S,5S,6S)-2-(4-formyl-2-nitrophenoxy)-6-(methoxycarbonyl)tetrahydro-2H-pyran-3,4,5-triyl triacetate

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,4-Tri-O-acetyl-β-D-glucopyranuronic acid methylester With hydrogen bromide; acetic acid In dichloromethane at 0 - 20℃; Stage #2: 4-hydroxy-3-nitrobenzaldehyde With silver(l) oxide In acetonitrile at 20℃; Darkness; | 98% |
-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
95-54-5
1,2-diamino-benzene

-
-
111664-46-1
4-(1-(4-hydroxy-3-nitrobenzyl)-1H-benzo[d]imidazol-2-yl)-2-nitrophenol

| Conditions | Yield |
|---|---|
| at 30℃; for 0.366667h; Catalytic behavior; Mechanism; Reagent/catalyst; Temperature; Time; Inert atmosphere; Green chemistry; chemoselective reaction; | 97% |
-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
21085-72-3
1-bromo-2,3,4-tri-O-acetyl-α-D-glucuronic acid methyl ester

-
-
148579-93-5
(2S,3R,4S,5S,6S)-2-(4-formyl-2-nitrophenoxy)-6-(methoxycarbonyl)tetrahydro-2H-pyran-3,4,5-triyl triacetate

| Conditions | Yield |
|---|---|
| With silver(l) oxide In acetonitrile for 4h; Koenigs-Knorr reaction; | 96% |
| With silver(l) oxide In acetonitrile for 4h; Koenigs-Knorr reaction; | 96% |
| With silver(l) oxide In acetonitrile at 20℃; for 12h; Koenigs-Knorr Glycosidation; Schlenk technique; Inert atmosphere; Molecular sieve; Darkness; | 96% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 30h; Inert atmosphere; | 96% |
| With potassium carbonate In acetonitrile Alkylation; |

| Conditions | Yield |
|---|---|
| In ethanol for 5h; Heating; | 96% |

-
-
4971-56-6
tetrahydrofuran-2,4-dione

-
-
580-15-4
6-aminoquinoline

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
1049682-10-1
11-(4-hydroxy-3-nitrophenyl)-8,11-dihydrofuro[3,4-b][4,7]phenanthrolin-10(7H)-one

| Conditions | Yield |
|---|---|
| In water at 120℃; for 0.116667h; microwave irradiation; | 96% |


| Conditions | Yield |
|---|---|
| potassium hydrogensulfate In acetic acid at 80℃; for 1.5h; | 95% |

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
100-39-0
benzyl bromide

-
-
22955-07-3
4-phenylmethoxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide | 95% |
| With potassium carbonate In N,N-dimethyl-formamide | 95% |
| With potassium carbonate In N,N-dimethyl-formamide | 95% |
-
-
580-15-4
6-aminoquinoline

-
-
1076-38-6
4-hydroxy[1]benzopyran-2-one

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
1038883-16-7
14-(4-hydroxy-3-nitrophenyl)-7H-chromeno[3,4-b][4,7]phenanthrolin-13(14H)-one

| Conditions | Yield |
|---|---|
| In water at 140℃; for 0.0833333h; microwave irradiation; | 95% |

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
126-81-8
dimedone

-
-
100-19-6
(4-nitrophenyl)ethanone

-
-
1107620-89-2
7,8-dihydro-4-(4-hydroxy-3-nitrophenyl)-7,7-dimethyl-2-(4-nitrophenyl)quinolin-5(1H,4H,6H)-one

| Conditions | Yield |
|---|---|
| With ammonium acetate at 100℃; for 0.15h; Microwave irradiation; | 95% |

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
126-81-8
dimedone

-
-
135-19-3
β-naphthol

-
-
1254077-26-3
12-(4-hydroxy-3-nitrophenyl)-9,9-dimethyl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-one

| Conditions | Yield |
|---|---|
| With silica supported perchloric acid at 80℃; for 1h; neat (no solvent); | 95% |

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
543-24-8
N-acetylglycine

-
-
346633-49-6
4-(4-Hydroxy-3-nitrobenzylidene)-2-methyloxazol-5(4H)-one

| Conditions | Yield |
|---|---|
| With sodium acetate In acetic anhydride for 4h; Inert atmosphere; Reflux; | 95% |

| Conditions | Yield |
|---|---|
| In methanol for 2h; Reflux; | 95% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In water at 20℃; for 0.583333h; | 95% |


| Conditions | Yield |
|---|---|
| With ethanolpyridole based semi-dendrimer supported on silica-coated magnetic Fe3O4 nanoparticles In neat (no solvent) for 0.25h; | 95% |
-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
3068-32-4
1-bromo-1-deoxy-2,3,4,6-tetra-O-acetyl-a-D-galactopyranoside

-
-
121358-46-1, 77667-26-6
(2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-(4-formyl-2-nitrophenoxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate

| Conditions | Yield |
|---|---|
| With silver(l) oxide In acetonitrile | 94% |
| With silver(l) oxide In acetonitrile for 4h; Inert atmosphere; | 83% |
| With silver(l) oxide In acetonitrile at 20℃; for 4h; Inert atmosphere; | 83% |
-
-
6919-98-8
2,3,4-tri-O-acetyl-1-bromo-1-deoxy-β-D-glucopyranuronic acid methy ester

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| silver(l) oxide In acetonitrile at 23℃; for 16h; | 94% |

| Conditions | Yield |
|---|---|
| With sodium azide; trifluorormethanesulfonic acid In acetonitrile at 20℃; for 0.0333333h; Schmidt reaction; | 94% |
| With trifluorormethanesulfonic acid; trimethylsilylazide In acetonitrile at 20℃; for 0.75h; Schmidt Reaction; Sealed tube; Inert atmosphere; | 82% |
| With hydroxylamine hydrochloride; sodium formate In formic acid; water |
-
-
4341-24-6
5-methylcyclohexan-1,3-dione

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
18940-21-1
5,5-dimethyl-3-(phenylamino)cyclohex-2-en-1-one

-
-
1026246-66-1
C28H28N2O5

| Conditions | Yield |
|---|---|
| With acetic acid at 120℃; for 0.0333333h; microwave irradiation; | 94% |

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
106-96-7
propargyl bromide

-
-
1313026-26-4
rac-4-(1-hydroxybut-3-yn-1-yl)-2-nitrophenol

| Conditions | Yield |
|---|---|
| Stage #1: propargyl bromide With aluminium; mercury dichloride In tetrahydrofuran; toluene at 65℃; for 0.5h; Inert atmosphere; Stage #2: 4-hydroxy-3-nitrobenzaldehyde In tetrahydrofuran; toluene Inert atmosphere; | 94% |
| Stage #1: propargyl bromide With aluminium; mercury dichloride In tetrahydrofuran; toluene for 6h; Reflux; Stage #2: 4-hydroxy-3-nitrobenzaldehyde In tetrahydrofuran; toluene at 0℃; for 0.5h; | 94% |
| Stage #1: propargyl bromide With aluminium; mercury dichloride In tetrahydrofuran; toluene for 6h; Reflux; Stage #2: 4-hydroxy-3-nitrobenzaldehyde In tetrahydrofuran; toluene at 0℃; for 0.5h; | 45% |
| Stage #1: propargyl bromide With aluminium; mercury dichloride In tetrahydrofuran for 6h; Reflux; Stage #2: 4-hydroxy-3-nitrobenzaldehyde In tetrahydrofuran at 0℃; for 30h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With ammonium acetate Hantzsch Dihydropyridine Synthesis; Reflux; | 94% |
-
-
1445-69-8
2,3-dihydrophthalazine-1,4-dione

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
109-77-3
malononitrile

| Conditions | Yield |
|---|---|
| With piperazine immobilized on shell of NiFe2O4-TiO2 core-shell nanoparticles via propylsilyl linkage at 80℃; for 1h; | 94% |
| With N,N,2,2,6,6-hexamethyl-N-(3-(trimethoxysilyl)propyl)piperidin-4-ammonium iodide grafted onto TiO2-coated NiFe2O4 nanocatalyst at 80℃; for 0.583333h; Green chemistry; | 93% |

-
-
3011-34-5
4-hydroxy-3-nitrobenzaldehyde

-
-
74-88-4
methyl iodide

-
-
31680-08-7
4-methoxy-3-nitrobenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 93.7% |
| With potassium carbonate | |
| With sodium carbonate In N-methyl-acetamide |
4-Hydroxy-3-nitrobenzaldehyde Specification
The 4-Hydroxy-3-nitrobenzaldehyde with CAS registry number of 3011-34-5 is also known as 4-Formyl-2-nitro-phenolate. The IUPAC name and product name are the same. It belongs to product categories of Alcohols and Derivatives; Carbonyl Compounds; Aromatic Aldehydes & Derivatives (substituted); Aldehydes; C7; Carbonyl Compounds. Its EINECS registry number is 221-141-6. In addition, the formula is C7H5NO4 and the molecular weight is 167.12. This chemical is a yellow-brown to brown powder and should be stored in sealed containers in cool and dry place.
Physical properties about 4-Hydroxy-3-nitrobenzaldehyde are: (1)ACD/LogP: 2.00; (2)ACD/LogD (pH 5.5): 1.38; (3)ACD/LogD (pH 7.4): -0.34; (4)ACD/BCF (pH 5.5): 4.63; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 69.27; (7)ACD/KOC (pH 7.4): 1.34; (8)#H bond acceptors: 5; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 3; (11)Index of Refraction: 1.666; (12)Molar Refractivity: 41.43 cm3; (13)Molar Volume: 111.3 cm3; (14)Surface Tension: 70.5 dyne/cm; (15)Density: 1.5 g/cm3; (16)Flash Point: 124.3 °C; (17)Enthalpy of Vaporization: 53.4 kJ/mol; (18)Boiling Point: 275 °C at 760 mmHg; (19)Vapour Pressure: 0.00312 mmHg at 25 °C.
Preparation of 4-Hydroxy-3-nitrobenzaldehyde. Dropping 24 g p-hydroxybenzaldehyde into acetic acid under stirring for 1 hour. Then the reaction mixture is cooled, filtrated and the filter cake is washed by ice water. At last, product is obtained by recrystallization of ethanol and the yield is about 61%.
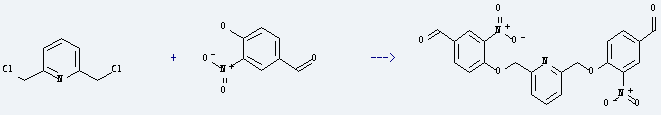
Uses of 4-Hydroxy-3-nitrobenzaldehyde: it is used as intermediate of medicine, pesticide and dye. It is also used to produce 2,6-di-(2'-nitro-4'-formylphenoxymethyl)pyridine by reaction with 2,6-bis-chloromethyl-pyridine. The reaction occurs with reagent K2CO3 and solvent dimethylformamide at 90-95 °C. The yield is about 70.5%.
When you are using this chemical, please be cautious about it. As a chemical, it is irritating to eyes, respiratory system and skin. During using it, wear suitable protective clothing. Avoid contact with skin and eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: C1=CC(=C(C=C1C=O)[N+](=O)[O-])O
2. InChI: InChI=1S/C7H5NO4/c9-4-5-1-2-7(10)6(3-5)8(11)12/h1-4,10H
3. InChIKey: YTHJCZRFJGXPTL-UHFFFAOYSA-N
Related Products
- 4-Hydroxy-3-nitrobenzaldehyde
- 30115-75-4
- 301180-04-1
- 3011-89-0
- 301-19-9
- 301206-84-8
- 30121-98-3
- 301221-56-7
- 301222-66-2
- 301224-40-8
- 301225-58-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View