-
Name
4-Chloro-4'-fluorobutyrophenone
- EINECS 223-395-3
- CAS No. 3874-54-2
- Article Data29
- CAS DataBase
- Density 1.183 g/cm3
- Solubility water: 0.38 g/L (20 °C)
- Melting Point 5-6 °C
- Formula C10H10ClFO
- Boiling Point 299.1 °C at 760 mmHg
- Molecular Weight 200.64
- Flash Point 134.7 °C
- Transport Information
- Appearance slightly yellow-greenish clear oil
- Safety 24/25-36-26
- Risk Codes 22-36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms Butyrophenone,4-chloro-4'-fluoro- (6CI,7CI,8CI);1-Chloro-4-(4-fluorophenyl)-4-oxobutane;3-(p-Fluorobenzoyl)propyl chloride;4-(4-Fluorophenyl)-4-oxobutyl chloride;4-Chloro-1-(4-fluorophenyl)-1-butanone;4-Chloro-1-(p-fluorophenyl)-1-oxobutane;4-p-Fluorophenyl-4-oxo-1-chlorobutane;4'-Fluoro-4-chlorobutyrophenone;p-Fluoro-4-chlorobutyrophenone;g-Chloro-p-fluorobutyrophenone;w-Chloro-p-fluorobutyrophenone;
- PSA 17.07000
- LogP 3.02740
Synthetic route
-
-
462-06-6
fluorobenzene

-
-
4635-59-0
4-Chlorobutanoyl chloride

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at 20℃; for 4h; Time; | 90% |
| With aluminium trichloride In carbon disulfide at 20℃; for 2h; | 73.2% |
| With aluminium trichloride |
-
-
3308-94-9
4-chloro-1-(4-fluorophenyl)-1,1-(ethylenedioxy)butane

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| In(OSO2CF3)3 In acetone at 100℃; for 0.0833333h; microwave irradiation; | 89% |
-
-
352-13-6
4-flourophenylmagnesium bromide

-
-
4635-59-0
4-Chlorobutanoyl chloride

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: 4-Chlorobutanoyl chloride With 1-methyl-pyrrolidin-2-one In toluene at 0℃; for 0.5h; Stage #2: 4-flourophenylmagnesium bromide In tetrahydrofuran; toluene at -10 - 0℃; for 4.25h; chemoselective reaction; | 88% |
-
-
1183278-95-6
1-(4-chlorobut-1-yn-1-yl)-4-fluorobenzene

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| With iron(III) chloride; water; silver(I) triflimide In 1,4-dioxane for 18h; regioselective reaction; | 85% |
| With water; bis(trifluoromethanesulfonyl)amide In 1,4-dioxane at 100℃; | 85% |
| With water In 1,4-dioxane at 80 - 120℃; regioselective reaction; | 85% |
-
-
339365-53-6
1-(4-fluorophenyl)cyclobutan-1-ol

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| With manganese(II) triflate; tetrabutylammonium acetate; acetic acid; magnesium chloride In acetonitrile at 25℃; for 3.5h; Inert atmosphere; Electrochemical reaction; Sealed tube; | 84% |
| With 1,10-Phenanthroline; tert-butylhypochlorite; silver trifluoromethanesulfonate In acetonitrile at 20℃; for 6h; Inert atmosphere; Schlenk technique; regioselective reaction; | 81% |
-
-
6139-84-0
4-chlorobutyraldehyde

-
-
460-00-4
1-Bromo-4-fluorobenzene

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| With Quinuclidine; 4,4'-di-tert-butyl-2,2'-bipyridine nickel(II) bromide; [Ir(C6H2F2-C5H3NCF3)2(4,4′-di-tert-butyl-2,2′-dipyridyl)]; potassium carbonate In 1,4-dioxane at 20℃; for 20h; Inert atmosphere; Sealed tube; Irradiation; | 77% |
| With palladium diacetate; 9,10-phenanthrenequinone; potassium hydrogencarbonate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In acetone at 20℃; for 24h; Reagent/catalyst; Irradiation; | 71% |
-
-
6940-76-7
1-iodo-3-chloro-propane

-
-
1765-93-1
4-fluoroboronic acid

-
-
13939-06-5, 199620-15-0
molybdenum hexacarbonyl

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: 1-iodo-3-chloro-propane; 4-fluoroboronic acid With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In water; benzene at 20℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Irradiation; Stage #2: molybdenum hexacarbonyl With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 70℃; Suzuki-Miyaura Coupling; Inert atmosphere; | 68% |

-
-
766-98-3
1-ethynyl-4-fluorobenzene

-
-
19263-25-3
3-chloropropionyl peroxide

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| With iron(III) trifluoromethanesulfonate; trifluorormethanesulfonic acid In tetrahydrofuran at 70 - 80℃; for 12.25h; Inert atmosphere; Schlenk technique; | 60% |
-
-
347-93-3
3-chloro-4'-fluoropropiophenone

-
-
3613-34-1
diethyl 4-chloromethyl-1,4-dihydro-2,6-dimethylpyridine-3,5-dicarboxylate

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| With manganese; di-tert-butyl peroxide; α,α,α-trifluorotoluene at 100℃; for 48h; Inert atmosphere; | 51% |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: iodine; magnesium / tetrahydrofuran / Inert atmosphere; Reflux; Sealed tube 1.2: 0 - 20 °C / Inert atmosphere; Sealed tube 2.1: acetic acid; manganese(II) triflate; magnesium chloride; tetrabutylammonium acetate / acetonitrile / 3.5 h / 25 °C / Inert atmosphere; Electrochemical reaction; Sealed tube View Scheme |
-
-
106-45-6
para-thiocresol

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
132427-90-8
1-(4-fluorophenyl)-4-(p-tolylthio)butan-1-one

| Conditions | Yield |
|---|---|
| 100% | |
| With triethylamine In tetrahydrofuran for 16h; Ambient temperature; | 99% |
-
-
593-56-6
N-methoxylamine hydrochloride

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
1352950-38-9
4-chloro-1-(4-fluorophenyl)butan-1-one O-methyl oxime

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 60℃; for 5h; | 100% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran | 99% |
| With triethylamine In tetrahydrofuran | 99% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran | 99% |
-
-
107-21-1
ethylene glycol

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
3308-94-9
4-chloro-1-(4-fluorophenyl)-1,1-(ethylenedioxy)butane

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In water; toluene for 16h; Inert atmosphere; Reflux; | 98% |
| With toluene-4-sulfonic acid In benzene for 18h; Heating; | 93% |
| With toluene-4-sulfonic acid In benzene for 15h; Heating; | 90% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; sodium iodide In acetonitrile for 22h; Reflux; | 96% |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
51787-87-2
(+/-)-4-chloro-1-(4-fluorophenyl)butan-1-ol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol at 0℃; for 7h; | 95% |
| With sodium tetrahydroborate In ethanol at 0 - 20℃; for 3h; | 94% |
| With sodium tetrahydroborate In ethanol at 0 - 20℃; for 12h; | 93% |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
40862-32-6
1-iodo-3-(p-fluorophenylcarbonyl)propane

| Conditions | Yield |
|---|---|
| With sodium iodide In butanone for 3h; Heating; | 95% |
| With sodium iodide In acetone for 24h; Reflux; | 92% |
| With sodium iodide In acetone for 16h; Reflux; | 77% |
| In sodium iodide; butanone | |
| With sodium iodide In [(2)H6]acetone for 8h; Reflux; |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
20651-65-4
4-(n-butyl)fluorobenzene

| Conditions | Yield |
|---|---|
| With nickel In tetrahydrofuran for 15h; Heating; | 95% |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
154440-76-3
C10H10FN3O

| Conditions | Yield |
|---|---|
| With sodium azide In N,N-dimethyl-formamide | 95% |
| With sodium azide; potassium iodide In N,N-dimethyl-formamide at 90℃; |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
343338-28-3
(S)-2-methylpropane-2-sulfinamide

-
-
1218989-31-1
(S,E)-N-(4-chloro-1-(4-fluorophenyl)butylidene)-2-methylpropane-2-sulfinamide

| Conditions | Yield |
|---|---|
| With titanium (IV) ethoxide In tetrahydrofuran at 65℃; for 48h; Inert atmosphere; | 94% |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: 4-(4-fluorophenyl)-4-oxo-n-butyl chloride With C32H33FeN3O2Si; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In toluene at -40 - 20℃; for 2h; Schlenk technique; Glovebox; Stage #2: With potassium carbonate In methanol; toluene at 20℃; for 12h; Schlenk technique; Glovebox; enantioselective reaction; | 94% |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
1021-25-6
1-Phenyl-1,3,8-triaza-spiro[4.5]decan-4-one

-
-
749-02-0
spiperone

| Conditions | Yield |
|---|---|
| With diisopropylamine In N,N-dimethyl-formamide at 80℃; for 17h; Inert atmosphere; | 93% |
| With triethylamine; potassium iodide In acetonitrile for 20h; Heating; | 74% |
| With triethylamine; potassium iodide In acetonitrile for 20h; Inert atmosphere; Reflux; | 63% |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
10387-40-3
potassium thioacetate

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 0 - 20℃; for 10h; Inert atmosphere; | 92% |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
126640-11-7
(1R)-(+)-4-chloro-1-(4-fluorophenyl)butan-1-ol

| Conditions | Yield |
|---|---|
| With B-chlorodiisopinocampheylborane In diethyl ether at -25℃; for 16h; | 90% |
| With C22H27Cl2CoN3O; sodium triethylborohydride; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; enantioselective reaction; | 87% |
| Stage #1: 4-(4-fluorophenyl)-4-oxo-n-butyl chloride With Triethoxysilane; (S,E)-(+)-2,6-diisopropyl-N-(2-((2-(4-phenyl-4,5-dihydrooxazol-2-yl)phenyl)amino)benzylidene)aniline; sodium triethylborohydride; cobalt(II) chloride In tetrahydrofuran; dichloromethane at 20℃; for 15h; Schlenk technique; Inert atmosphere; Stage #2: With potassium carbonate In tetrahydrofuran; methanol; dichloromethane at 20℃; for 2h; Schlenk technique; Inert atmosphere; enantioselective reaction; | 75% |
| Multi-step reaction with 2 steps 1: 81 percent / NaBH4 / methanol / 4 °C View Scheme | |
| With B-chlorodiisopinocampheylborane In tetrahydrofuran; diethyl ether; 2,2'-iminobis[ethanol] at 25℃; |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
126640-10-6
(1S)-(-)-4-chloro-1-(4-fluorophenyl)butan-1-ol

| Conditions | Yield |
|---|---|
| With (-)-diisopinocamphenylborane chloride In diethyl ether at -25℃; for 16h; | 90% |
| Multi-step reaction with 2 steps 1: 81 percent / NaBH4 / methanol / 4 °C View Scheme | |
| With (-)-diisopinocamphenylborane chloride In tetrahydrofuran; diethyl ether; 2,2'-iminobis[ethanol] at 25℃; |
-
-
459-56-3
4-fluorobenzylic alcohol

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
868699-44-9
cyclopropyl-[4-(4-fluoro-benzyloxy)-phenyl]-methanone

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium hydride In tetrahydrofuran at 30℃; for 18h; | 90% |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: 4-(4-fluorophenyl)-4-oxo-n-butyl chloride With (S)-2,2',6,6'-tetramethoxy-4,4'-bis(diphenylphosphino)-3,3'-bipyridine; phenylsilane; copper(II) acetate monohydrate In toluene at -20 - 20℃; for 48h; Stage #2: With hydrogenchloride In water; toluene Reagent/catalyst; enantioselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With methyllithium lithium bromide In tetrahydrofuran; diethyl ether at -78℃; for 0.0833333h; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide at 140℃; | 90% |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
54540-58-8
1-(4-chlorobutyl)-4-fluorobenzene

| Conditions | Yield |
|---|---|
| With triethylsilane; trifluoroacetic acid at 0℃; for 2h; | 89% |
| With hydrogen; palladium on activated charcoal In ethanol | |
| With hydrogenchloride; mercury dichloride; zinc In toluene for 5h; Reflux; | |
| With triethylsilane In trifluoroacetic acid |
-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
352-13-6
4-flourophenylmagnesium bromide

-
-
51787-80-5
δ-Hydroxy-δ-bis(p-fluorophenyl)butyl chloride

| Conditions | Yield |
|---|---|
| In diethyl ether Heating; | 88.4% |
-
-
40807-61-2
4-hydroxy-4-phenylpiperidin

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
3109-12-4
1-(4-fluorophenyl)-4-(4-hydroxy-4-phenylpiperidin-1-yl)butan-1-one

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; potassium iodide In toluene for 24h; Reflux; | 88% |
| With potassium iodate; potassium carbonate In toluene Inert atmosphere; | 27% |
| With toluene; potassium iodide |
-
-
39512-49-7
4-(4-chlorophenyl)-4-hydroxypiperidine

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
52-86-8
haloperidol

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In acetonitrile at 75℃; for 24h; Temperature; | 88% |
| With potassium iodide In toluene for 45h; Reflux; | 85% |
| With potassium iodide In toluene at 130℃; for 45h; Sealed tube; | 79% |
-
-
41885-98-7
1-(4-chlorophenyl)-1,4-diazepane

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In 1,2-dimethoxyethane for 18h; Heating; | 87% |
-
-
3240-34-4
[bis(acetoxy)iodo]benzene

-
-
3874-54-2
4-(4-fluorophenyl)-4-oxo-n-butyl chloride

-
-
1394244-16-6
C12H12ClFO3

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium hydroxide In 1,4-dioxane at 20℃; for 1h; | 87% |
4-chloro-4'-fluorobutyrophenone Chemical Properties
The MF of 4-chloro-4'-fluorobutyrophenone(3874-54-2): C10H10ClFO
The MW of 4-chloro-4'-fluorobutyrophenone(3874-54-2): 200.64
EINECS: 223-395-3
mp: 5-6 °C
bp: 130-132 °C (0.97513 mmHg)
density: 1.22 g/mL at 25 °C(lit.)
refractive index: n20/D 1.5255(lit.)
Fp: >230 °F
Water Solubility: 0.38 g/L (20 °C)
Appearance: Yellow to green liquid.
Incompatibilities: Oxidizing agents.
Stability: Stable under normal temperatures and pressures.
Decomposition: Hydrogen chloride, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, hydrogen fluoride.
Categories: ketone;Fluorobenzene;C10;Carbonyl Compounds;Ketones
Synonyms: GAMMA-CHLORO-4-FLUOROBUTYROPHENONE;GAMMA-CHLORO-P-FLUOROBUTYROPHENONE;G-CHLORO-P-FLUOROBUTYROPHENONE;AKOS BBS-00006481;4-CHLORO-1-(4-FLUOROPHENYL)-1-BUTANONE;4-CHLORO-1-(4-FLUOROPHENYL)-1-OXOBUTANE;4-CHLORO-4'-FLUOROBUTYROPHENONE;4-CHLORO-4-FLUOROBUTYROPHENONE
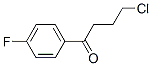
The Structure of 4-chloro-4'-fluorobutyrophenone(3874-54-2):
4-chloro-4'-fluorobutyrophenone Safety Profile
 Xn,Xi
Xn,Xi Risk Statements: 22-36/37/38-20/21/22
22: Harmful if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
20/21/22: Harmful by inhalation, in contact with skin and if swallowed
Safety Statements: 24/25-36-26
24/25: Avoid contact with skin and eyes
36: Wear suitable protective clothing
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
WGK Germany: 3
Hazard Note: Irritant
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View