-
Name
Ammonium oxalate monohydrate
- EINECS 238-135-4
- CAS No. 6009-70-7
- Article Data13
- CAS DataBase
- Density 1.5 g/cm3
- Solubility H2O: 0.1 M at 20 °C, clear, colorless
- Melting Point 133 °C (dec.)(lit.)
- Formula C2H8N2O4.H2O
- Boiling Point 365.1 °C at 760 mmHg
- Molecular Weight 142.112
- Flash Point 188.8 °C
- Transport Information UN 1759 8/PG 3
- Appearance White solid
- Safety 24/25
- Risk Codes 21/22
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms Ethanedioicacid, diammonium salt, monohydrate (9CI);Oxalic acid, diammonium salt,monohydrate (8CI);Ammonium oxalate ((NH4)2C2O4) monohydrate;Diammonium oxalate monohydrate;
- PSA 89.49000
- LogP -2.82570
Synthetic route

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| at 100℃; |

| Conditions | Yield |
|---|---|
| In water soln. of ammonia and oxalic acid in H2O slowly evapd. at room temp. overabout 15-20 d; |


| Conditions | Yield |
|---|---|
| In water neutralizing aq. oxalic acid soln. with (NH4)2CO3; evapn.; | |
| In water neutralizing aq. oxalic acid soln. with (NH4)2CO3; evapn.; |

| Conditions | Yield |
|---|---|
| With acetone In water mixt. of acetone and 3 % aq. HCN soln.; by light; | |
| With acetone In water mixt. of acetone and 3 % aq. HCN soln.; by light; |
-
-
12135-76-1
ammonium sulfide

-
B
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| In water by standing of aq. soln. of educt; | |
| In water by standing of aq. soln. of educt; |


| Conditions | Yield |
|---|---|
| In water neutralizing aq. oxalic acid soln. with NH3; evapn.; | |
| In water neutralizing aq. oxalic acid soln. with NH3; evapn.; |

| Conditions | Yield |
|---|---|
| In water with freshly pptd. Pb oxalate; | |
| In water with freshly pptd. Pb oxalate; |

-
-
7732-18-5
water

-
-
6009-70-7
ammonium oxalate monohydrate

-
-
20161-19-7, 34729-05-0
nickel(II) oxalate dihydrate

| Conditions | Yield |
|---|---|
| In water (NH4)2C2O4*H2O (48.87 g) added to 5.0 M Ni(NO3)2 (20 ml) (70-90°C); dispersed with deionized H2O; filtered; washed; dehydrated with anhyd. ethanol; dried (80-90°C); SEM; | 99.76% |


| Conditions | Yield |
|---|---|
| In water Ni(NO3)2 soln. added to supersatd. (NH4)2C2O4; SEM; | 97.98% |


-
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| In water 2 equiv. of NH4-oxalate soln. was added to aq. soln. of metal-complex; crystals were filtered, washed with EtOH, Et2O, dried in vac., elem. anal.; | 95% |
-
-
12354-84-6, 12354-85-7
bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]

-
-
6009-70-7
ammonium oxalate monohydrate

-
-
280742-17-8
[Cp*2Ir2(μ-η4-C2O4)]Cl2

| Conditions | Yield |
|---|---|
| In methanol; chloroform under N2 atm. (NH4)2C2O4*H2O was added to suspn. Ir complex in CHCl3-MeOH and stirred at 55°C for 5 h; solvent was evapd. to dryness in vacuo, residue was extd. with CH2Cl2; elem. anal.; | 91% |
| In methanol (N2); std. Schlenk technique; oxalate was added to suspn. of Ir complex in MeOH; stirred at 55°C for 5 h; evapd. (vac.); extd. (CH2Cl2); elem. anal.; | 91% |
-
-
52462-29-0
[ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2

-
-
6009-70-7
ammonium oxalate monohydrate

-
-
201336-17-6, 1185317-98-9, 201425-24-3
[(p-cymene)2Ru2(μ-η4-C2O4)]Cl2

| Conditions | Yield |
|---|---|
| In methanol; chloroform (N2); reflux (6 h); removal of solvents, taken up in CH2Cl2, filtration, evapn. to dryness; elem. anal.; | 89% |

-
-
6009-70-7
ammonium oxalate monohydrate

-
-
958667-73-7
[(η5-pentamethylcyclopentadienyl)2Rh2(μ-oxalato-κO)Cl2]

| Conditions | Yield |
|---|---|
| In methanol; chloroform byproducts: ammonium chloride; (NH4)2C2O4*H2O added to CHCl3-MeOH soln. of Rh complex; refluxed for 6 h; solvent removed under vac.; dissolved in CH2Cl2; filtered; filtrate evapd. to dryness; elem. anal.; | 88% |

-
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| In water satd. aq. soln. of NH4-oxalate was added to aq. soln. of metal-complex; crystals were filtered, washed with EtOH, Et2O, dried in vac., elem. anal.; | 86% |

-
-
6009-70-7
ammonium oxalate monohydrate

-
-
958667-73-7
[Cp*2Rh2(μ-η2-η2-C2O4)]Cl2

| Conditions | Yield |
|---|---|
| In methanol; chloroform under N2 atm. (NH4)2C2O4*H2O was added to suspn. Rh complex in CHCl3-MeOH and stirred at 55°C for 5 h; solvent was evapd. to dryness in vacuo, residue was extd. with CH2Cl2; elem. anal.; | 86% |
-
-
52462-29-0
[ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2

-
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| With AgSO4 In water (N2); addn. of AgSO4 to the Ru complex in H2O, stirring (room temp., 3 h), filtration, addn. of (NH4)2C2O4*H2O, adjustment ot pH to 7-8, heatingto 70°C (1-2 h); evapn. to dryness, addn. of CH2Cl2, filtration, evapn., recrystn. (MeOH/Et2O); elem. anal.; | 84% |

-
-
7722-84-1
dihydrogen peroxide

-
-
6153-56-6
oxalic acid dihydrate

-
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| In water ammonium heptamolybdate tetrahydrate dissolved in soln. of oxalic acid and ammonium oxalate, treated with excess of soln. of H2O2 (35 wt,-%), stirred for 2 h; EtOH added, ppt. filtered off, washed with EtOH and air-dried; elem. anal.; | 84% |

-
-
12354-84-6, 12354-85-7
bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]

-
-
6009-70-7
ammonium oxalate monohydrate

-
-
280742-17-8
[(η5-C5Me5)IrCl(μ-η2-η2-oxalato)ClIr(η5-C5Me5)]

| Conditions | Yield |
|---|---|
| In methanol; chloroform byproducts: ammonium chloride; (NH4)2C2O4*H2O added to CHCl3-MeOH soln. of Ir complex; refluxed for 6 h; solvent removed under vac.; dissolved in CH2Cl2; filtered; filtrate evapd. to dryness; elem. anal.; | 81% |
-
-
20023-19-2
[Co(tris(2-aminoethyl)amine)Cl2]Cl

-
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| In water slow addition of soln. of oxalate to soln. of Co complex, heating (10 min), addn. of NaClO4; filtration, pptn. (2 weeks); elem. anal.; | 77% |


| Conditions | Yield |
|---|---|
| In water warm aq. soln. of (NH4)2ox (11 mmol) added to aq. soln. of Co compd. (10mmol) at 60°C, stirred for 30 min; pptd. on storage overnight, ppt. filtered off, washed (H2O, EtOH, ether), elem. anal.; | 73% |


-
-
1093853-11-2
N-(1H-pyrrol-2-ylmethylene)-2-(pyridin-2-yl)ethanamine

-
-
6009-70-7
ammonium oxalate monohydrate

-
-
1190210-76-4
[Cu2(N-(1H-pyrrol-2-ylmethylene)-2-pyridine-ethaneamine)2(oxalate)]

| Conditions | Yield |
|---|---|
| With Et3N In methanol; water MeOH soln. of Et3N (0.3 mmol) added to meOH soln. of ligand (0.3 mmol), soln. added to meOH soln. of Cu compd. (0.3 mmol), aq. ammonium oxalate (0.15 mmol) added, mixt. stirred for 6 h; ppt. filtered off, washed (MeOH), dried (vac.), elem. anal.; | 71% |


| Conditions | Yield |
|---|---|
| With Et3N In water High Pressure; mixt. of La2O3, (NH4)2C2O4*H2O, C6H5O3N and H2O in molar ratio of 1:1:1:290 adjusted to pH of 4.5 (Et3N) at room temp.; treated in autoclave at 170°C for 5 d; cooled to room temp. at a rate of 5°C/h; elem. anal., detd. by XRD; | 68% |


| Conditions | Yield |
|---|---|
| With Et3N In water High Pressure; mixt. of Dy2O3, (NH4)2C2O4*H2O, C6H5O3N and H2O in molar ratio of 1:1:1:290 adjusted to pH of 4.6 (Et3N) at room temp.; treated in autoclave at 180°C for 2 d; cooled to room temp. at a rate of 2°C/h; elem. anal., detd. by XRD; | 67% |


-
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| In methanol under N2 atm.; (NH4)2C2O4*H2O added to soln. of Ir complex in MeOH, stirred at 55°C for 5 h; | 65% |
-
-
23814-12-2
1h-benzotriazole-5-carboxylic acid

-
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| In water; acetonitrile at 140℃; for 72h; Autoclave; | 56% |


| Conditions | Yield |
|---|---|
| With Et3N In water High Pressure; mixt. of Tb2O3, (NH4)2C2O4*H2O, C6H5O3N and H2O in molar ratio of 1:1.5:1:290 adjusted to pH of 4.0 (Et3N) at room temp.; treated in autoclave at 170°C for 3 d; cooled to room temp. at a rate of 2.5°C/h; elem. anal., detd. by XRD; | 54% |


| Conditions | Yield |
|---|---|
| With Et3N In water High Pressure; mixt. of Sm2O3, (NH4)2C2O4*H2O, C6H5O3N and H2O in molar ratio of 1:1.5:1:290 adjusted to pH of 4.0 (Et3N) at room temp.; treated in autoclave at 170°C for 3 d; cooled to room temp. at a rate of 2.5°C/h; elem. anal., detd. by XRD; | 52% |

-
-
23814-12-2
1h-benzotriazole-5-carboxylic acid

-
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| In water; acetonitrile at 140℃; for 72h; Autoclave; | 46% |


| Conditions | Yield |
|---|---|
| With Et3N In water High Pressure; mixt. of Pr2O3, (NH4)2C2O4*H2O, C6H5O3N and H2O in molar ratio of 1:1:1:290 adjusted to pH of 4.5 (Et3N) at room temp.; treated in autoclave at 170°C for 5 d; cooled to room temp. at a rate of 5°C/h; elem. anal., detd. by XRD; | 42% |


| Conditions | Yield |
|---|---|
| With Et3N In water High Pressure; mixt. of Er2O3, (NH4)2C2O4*H2O, C6H5O3N and H2O in molar ratio of 1:1:1:290 adjusted to pH of 4.6 (Et3N) at room temp.; treated in autoclave at 180°C for 2 d; cooled to room temp. at a rate of 2°C/h; elem. anal., detd. by XRD; | 41% |


| Conditions | Yield |
|---|---|
| With Et3N In water High Pressure; mixt. of Gd2O3, (NH4)2C2O4*H2O, C6H5O3N and H2O in molar ratio of 1:1.5:1:290 adjusted to pH of 4.0 (Et3N) at room temp.; treated in autoclave at 170°C for 3 d; cooled to room temp. at a rate of 2.5°C/h; elem. anal., detd. by XRD; | 41% |


| Conditions | Yield |
|---|---|
| With Et3N In water High Pressure; mixt. of Nd2O3, (NH4)2C2O4*H2O, C6H5O3N and H2O in molar ratio of 1:1:1:290 adjusted to pH of 4.5 (Et3N) at room temp.; treated in autoclave at 170°C for 5 d; cooled to room temp. at a rate of 5°C/h; elem. anal., detd. by XRD; | 41% |


| Conditions | Yield |
|---|---|
| With HNO3 In sodium hydroxide; water aq. NaOH; High Pressure; dicarboxylic acid and (NH4)2C2O4 (1:2) in aq.NaOH mixed with aq. soln. of Er salt (2 equiv.), pH adjusted to 5 (HNO3), stirred for 8 min (air), heated in an autoclave at 160°C for 72 h; cooled to room temp. over 12 h, elem. anal.; | 36% |


| Conditions | Yield |
|---|---|
| In water (NH4)2C2O4*H2O was added to aq. soln. FeCl3, gently warmed for 10 min, soln. was allowed to stand at room temp. for several days; crystals were filtered and washed with EtOH and Et2O; elem. anal.; | A n/a B 30% |

-
-
7722-64-7
potassium permanganate

-
-
7732-18-5
water

-
-
333-18-6
1,2-diaminoethane dihydrochloride

-
-
6009-70-7
ammonium oxalate monohydrate

-
-
7440-48-4
cobalt

| Conditions | Yield |
|---|---|
| With CH3OH; air In methanol mixt. of Co powder, KMnO4, C2H4(NH2)2(HCl)2, (NH4)2C2O4*H2O and methanolheated at 50-60°C in air with stirring for 8 h; ppt. filtered off, washed with 2-propanol and dried under vac. at room temp.; recrystn. from H2O; elem. anal.; | 30% |

-
-
7722-64-7
potassium permanganate

-
-
7732-18-5
water

-
-
333-18-6
1,2-diaminoethane dihydrochloride

-
-
6009-70-7
ammonium oxalate monohydrate

-
-
7440-48-4
cobalt

| Conditions | Yield |
|---|---|
| With CH3OH; air In N,N-dimethyl-formamide mixt. of Co powder, KMnO4, C2H4(NH2)2(HCl)2, (NH4)2C2O4*H2O and DMF/methanol (20/0.1) heated at 50-60°C in air with stirring for 8 h; ppt. filtered off, washed with 2-propanol and dried under vac. at room temp.; elem. anal.; | 27% |


-
-
7722-84-1
dihydrogen peroxide

-
-
6153-56-6
oxalic acid dihydrate

-
-
6009-70-7
ammonium oxalate monohydrate

| Conditions | Yield |
|---|---|
| In water ammonium heptamolybdate tetrahydrate dissolved in soln. of oxalic acid and ammonium oxalate, treated with soln. of H2O2 (35 wt.-%); EtOH added, stored at 5°C for 1 week, ppt. filtered off, washed with EtOH and air-dried ((NH4)2(MoO2(O2)(ox))*H2O), slow evapn. of filtrate yields (NH4)2(Mo2O2(O2)2(OH)2(ox)2); elem. anal.; | A 16% B n/a |

Ammonium oxalate monohydrate Specification
The Ethanedioic acid,ammonium salt, hydrate (1:2:1), with CAS registry number 6009-70-7, belongs to the following product category: Industrial/Fine Chemicals. It has the systematic name of ethanedioic acid diammoniate hydrate. This chemical is a kind of white solid. And it should be stored at the room temperature.
Physical properties of Ethanedioic acid,ammonium salt, hydrate (1:2:1): (1)ACD/LogP: -1.19; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -5.7; (4)ACD/LogD (pH 7.4): -5.94; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 4; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 52.6 Å2.
Preparation: this chemical can be prepared by Ammonia and ammonium carbonate. This two chemical should add into oxalic acid, so they can happen the neutralization reaction. After concentration, it can be the Ethanedioic acid,ammonium salt, hydrate (1:2:1).
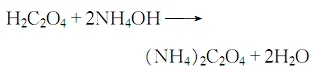
When you are using this chemical, please be cautious about it as the following:
The Ethanedioic acid,ammonium salt, hydrate (1:2:1) is harmful in contact with skin and if swallowed. When use this chemical, avoid contact with skin and eyes.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)C(=O)O.O.N.N
(2)InChI: InChI=1/C2H2O4.2H3N.H2O/c3-1(4)2(5)6;;;/h(H,3,4)(H,5,6);2*1H3;1H2
(3)InChIKey: MSMNVXKYCPHLLN-UHFFFAOYAR
(4)Std. InChI: InChI=1S/C2H2O4.2H3N.H2O/c3-1(4)2(5)6;;;/h(H,3,4)(H,5,6);2*1H3;1H2
(5)Std. InChIKey: MSMNVXKYCPHLLN-UHFFFAOYSA-N
Related Products
- Ammonium (-)-3-bromo-8-camphorsulfonate
- Ammonium (S)-5-((4-amino-4-carboxy-1-oxobutyl)amino)-2-nitrobenzoate
- Ammonium 1-pyrrolidinedithiocarbamate
- Ammonium 2,4-dichlorophenoxyacetate
- Ammonium 2-hydroxyethanesulphonate
- Ammonium 4-nitrobenzoate dihydrate
- Ammonium acetate
- Ammonium adipate
- Ammonium alginate
- Ammonium azide
- 6009-98-9
- 601-01-4
- 60103-17-5
- 601-03-6
- 60104-30-5
- 60-10-6
- 6010-91-9
- 6011-14-9
- 60111-51-5
- 60111-54-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View