-
Name
Benzilic acid
- EINECS 200-993-2
- CAS No. 76-93-7
- Article Data164
- CAS DataBase
- Density 1.279 g/cm3
- Solubility 1.41 g/L (25 ºC)
- Melting Point 149-151 °C(lit.)
- Formula C14H12O3
- Boiling Point 409 °C at 760 mmHg
- Molecular Weight 228.247
- Flash Point 215.3 °C
- Transport Information
- Appearance white powder
- Safety 36/37/39-26
- Risk Codes 36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 2-Hydroxy-2,2-diphenylaceticacid;Diphenylhydroxyacetic acid;a,a-Diphenyl-a-hydroxyacetic acid;a-Hydroxy-2,2-diphenylacetic acid;a-Hydroxydiphenylacetic acid;
- PSA 57.53000
- LogP 2.00710
Synthetic route

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In acetonitrile Electrochemical reaction; Irradiation; | 98% |
| With Tetrapropylammonium chloride In acetonitrile at 20℃; under 760.051 Torr; for 10h; Electrochemical reaction; | 90% |
| With potassium iodide In N,N-dimethyl-formamide Hg pool cathode, platinum plate anode, constant current of 2.5 nA/cm2; | 86% |
| With tetrabutylammomium bromide In N,N-dimethyl-formamide at 20℃; electrochemical reaction; | 75% |
| With tetrabutylammonium halide In N,N-dimethyl-formamide Ambient temperature; electrolysis, Mg anode; | 70% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water for 0.5h; Heating; | 97% |
| With water; sodium hydroxide In ethanol for 0.5h; Reflux; | 97% |
| Stage #1: benzil With N-benzyl-trimethylammonium hydroxide at 40℃; for 4h; Stage #2: With hydrogenchloride In water pH=3; | 92% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol at 65℃; | 92% |
-
-
13249-75-7
(Z)-1,2,4-triphenyl-2-butene-1,4-dione

-
-
119-53-9
2-hydroxy-2-phenylacetophenone

-
A
-
4441-01-4
1,2,4-triphenylbutane-1,4-dione

-
B
-
76-93-7
Benzilic acid

-
C
-
134-81-6
benzil

| Conditions | Yield |
|---|---|
| With sodium hydroxide In dimethyl sulfoxide for 2h; Mechanism; Ambient temperature; other reaction conditions, other substrates; | A 91% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With C19H32N6O4(2+)*2Br(1-); sodium hydroxide In acetonitrile for 1.83333h; Catalytic behavior; Reagent/catalyst; | 90% |
| Stage #1: benzaldehyde With oxygen; 1-butyl-3-methylimidazolium Tetrafluoroborate; potassium hydroxide at 60℃; for 0.416667h; Neat (no solvent); Microwave irradiation; Stage #2: With hydrogenchloride In water at 20℃; | 74% |
-
-
18181-80-1
bromopropylate

-
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| With methanol; gold; hydrogen; caesium carbonate at 100℃; under 3800.26 Torr; for 96h; | 87% |
-
-
62217-32-7
2-diphenylmethylene-1,3-benzodithiole

-
A
-
6461-76-3
1,2-benzenedisulfonyl dichloride

-
B
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| With chlorine In water; tert-butyl alcohol at 0 - 5℃; for 2h; Yields of byproduct given; | A n/a B 85% |
| With chlorine In water; tert-butyl alcohol at 0 - 5℃; for 2h; Yield given; | A n/a B 85% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; benzene | 75% |
-
-
492-70-6, 655-48-1, 2325-10-2, 38270-73-4, 52340-78-0, 579-43-1
1,2-diphenyl-1,2-ethanediol

-
A
-
76-93-7
Benzilic acid

-
B
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1,2-diphenyl-1,2-ethanediol With oxygen; sodium t-butanolate In tetrahydrofuran at 20℃; under 760.051 Torr; for 3h; Stage #2: With hydrogenchloride In tetrahydrofuran; water pH=1; Reagent/catalyst; Solvent; chemoselective reaction; | A 10% B 74% |
-
-
451-40-1
phenyl benzyl ketone

-
A
-
106-47-8
4-chloro-aniline

-
B
-
76-93-7
Benzilic acid

-
C
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With nitrobenzene In potassium hydroxide; tert-butyl alcohol for 0.5h; Heating; | A 71% B 58% C 24% |


| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 100℃; for 1h; Temperature; Sealed tube; | 70% |
| With potassium hydroxide | |
| With potassium hydroxide at 100℃; |
-
-
119-61-9
benzophenone

-
-
124-38-9
carbon dioxide

-
A
-
91-01-0
1,1-Diphenylmethanol

-
B
-
117-34-0
2,2-diphenylacetic acid

-
C
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| With ytterbium In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide under 760 Torr; for 0.833333h; Ambient temperature; | A 12% B 4% C 61% |
| With ytterbium 1.) THF, HMPA, RT, 2.) RT, 1 atm, 30 min; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
64-18-6
formic acid

-
-
7475-56-1
chloro(diphenyl)acetic acid

-
A
-
133217-20-6
(formyloxy)diphenylacetic acid

-
B
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| In dichloromethane at 80℃; for 1h; | A 61% B 25% C n/a |

-
-
119-61-9
benzophenone

-
-
124-38-9
carbon dioxide

-
-
121-44-8
triethylamine

-
A
-
91-01-0
1,1-Diphenylmethanol

-
B
-
52183-00-3
1,1-diphenylpropane-1,2-diol

-
C
-
464-72-2
tetraphenylethane-1,2-diol

-
D
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| With tetraethylammonium chloride; poly(p-phenylene) In N,N-dimethyl-formamide for 24h; Carboxylation; reduction; dimerization; Irradiation; | A 4% B 3% C 49% D 27% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; water; tetra-(n-butyl)ammonium iodide In acetonitrile at 90℃; for 12h; | 45% |
-
-
119-61-9
benzophenone

-
-
124-38-9
carbon dioxide

-
A
-
117-34-0
2,2-diphenylacetic acid

-
B
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| Stage #1: benzophenone With LiVH2 In tetrahydrofuran at 20℃; Stage #2: carbon dioxide In tetrahydrofuran Further stages.; | A 9% B 28% |

-
-
119-61-9
benzophenone

-
-
124-38-9
carbon dioxide

-
A
-
91-01-0
1,1-Diphenylmethanol

-
B
-
464-72-2
tetraphenylethane-1,2-diol

-
C
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| With cadmium(II) sulphide; triethylamine In N,N-dimethyl-formamide at 20℃; for 0.5h; Irradiation; | A 26% B 23% C 11% |
| With triethylamine; lithium chloride; poly(p-phenylene) In N,N-dimethyl-formamide for 24h; Irradiation; | A 8 % Chromat. B 25% C 5% |
| With tetraethylammonium chloride; triethylamine; poly(p-phenylene) In N,N-dimethyl-formamide for 24h; Irradiation; | A 10% B 10% C 34 % Chromat. |

-
-
7494-95-3
bromo-diphenyl-acetic acid

-
A
-
2571-41-7
azido-diphenyl-acetic acid

-
B
-
525-06-4
diphenyl ketene

-
C
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| With sodium azide In N,N-dimethyl-formamide | A 25% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With diethyl ether; carbon dioxide; sodium Zersetzung mit Wasser; |
-
-
623-11-0
1-methyl-4-nitrosobenzene

-
-
427-47-4
fluoro-diphenyl-acetic acid ethyl ester

-
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| at 20℃; |

| Conditions | Yield |
|---|---|
| With ammonia; sodium |

| Conditions | Yield |
|---|---|
| With potassium permanganate at 0℃; |

| Conditions | Yield |
|---|---|
| With nitric acid |
-
-
4198-95-2
α-phenylimino-deoxybenzoin

-
A
-
1922-78-7
anilino-diphenyl-acetic acid

-
B
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| at 170 - 180℃; in der Kalischmelze; | |
| at 170 - 180℃; bei der Kalischmelze; |

| Conditions | Yield |
|---|---|
| With sodium carbonate |
-
-
10049-90-8
N,N-diethyl-α-hydroxy-α-phenylbenzeneacetamide

-
A
-
76-93-7
Benzilic acid

-
B
-
109-89-7
diethylamine

| Conditions | Yield |
|---|---|
| in der Alkalischmelze; |

| Conditions | Yield |
|---|---|
| With water | |
| With barium dihydroxide |

-
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| With ethanol |

| Conditions | Yield |
|---|---|
| bei der Verseifung; |

| Conditions | Yield |
|---|---|
| With pyridine chromium peroxide In dichloromethane for 0.3h; Product distribution; Ambient temperature; effect of various chromium(VI) based oxidants; | 100% |
| In acetic acid for 0.25h; Heating; | 100% |
| With sodium hydroxide; copper(III) periodate for 0.05h; Heating; oxidative decarboxylation of α-hydroxy acids; var. α-hydroxy acids, also Cu(III) tellurate as oxidant; | 95% |

| Conditions | Yield |
|---|---|
| With phosphonic Acid; methanesulfonic acid; sodium iodide In water at 95℃; for 24h; Inert atmosphere; | 99.3% |
| With triethylsilane; perchloric acid In dichloromethane; water at 40℃; for 20h; | 94% |
| With phosphorus; iodine weiteres Reagens: wss. Phosphorsaeure; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; acetonitrile at 80 - 85℃; for 16 - 18h; | 99% |
| With thionyl chloride at 0 - 75℃; for 12.0833h; Schlenk technique; Inert atmosphere; Reflux; | 91% |
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| With sulfuric acid In acetic acid for 18h; Ambient temperature; | 97% |
-
-
148661-29-4
2,2-dihydroxy-6-methyl-1,3,6,2-dioxazagermocane

-
-
76-93-7
Benzilic acid

-
-
149236-31-7
Ge{(OCH2CH2)2NCH3}{OC(C6H5)2C(O)O}

| Conditions | Yield |
|---|---|
| In ethanol; xylene addn. of Ge-compd. (abs. alcohol) to refluxing mixt. of org. ligand in EtOH and xylene over a period of 2 h, further refluxing for 1 h; concn. of soln., crystn. on standing; collecting, washing (benzene), drying in vac. over P2O5; elem. anal.; | 96% |

| Conditions | Yield |
|---|---|
| With acetyl chloride In dichloromethane | 95% |
| With phosphorus pentachloride; benzene | |
| With phosphorus trichloride |
-
-
712356-53-1
(1α, 5α, 6α)-6N-(3-azabicyclo [3.1.0] hexyl-3-benzyl)-2-chloro acetamide

-
-
76-93-7
Benzilic acid

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In xylene for 3h; | 95% |
-
-
143794-20-1
dihydroxo(η3-2,2'-iminodiethoxo)germanium(IV)

-
-
76-93-7
Benzilic acid

-
-
148661-56-7
Ge{(OCH2CH2)2NH}{OC(C6H5)2C(O)O}

| Conditions | Yield |
|---|---|
| In ethanol; xylene addn. of Ge-compd. (abs. alcohol) to refluxing mixt. of org. ligand in EtOH and xylene over a period of 2 h, further refluxing for 1 h; concn. of soln., crystn. on standing; collecting, washing (benzene), drying in vac. over P2O5; elem. anal.; | 95% |
-
-
102-79-4
n-butyldiethanolamine

-
-
76-93-7
Benzilic acid

-
-
162441-06-7
(α-hydroxydiphenylaceto-O,O')(N-n-butyliminodiethanolato-N,O,O')germanium(IV)

| Conditions | Yield |
|---|---|
| In ethanol; water; xylene refluxing (2 h); solvent removal, crystn. on cooling, filtn., drying (vac.); elem. anal.; | 95% |

-
-
1201903-03-8
Ν,Ν',Ν''-[boroxin-2,4,6-triyltris[[(1R)-3-methylbutane-1,1-diyl]imino(2-oxoethane-2,1-diyl)]]tris(2,5-dichlorobenzamide)

-
-
76-93-7
Benzilic acid

-
-
1201902-93-3
2,5-dichloro-N-(2-{[(1R)-3-methyl-1-(5-oxo-4,4-diphenyl-1,3,2-dioxaborolan-2-yl)butyl]amino}-2-oxoethyl)benzamide

| Conditions | Yield |
|---|---|
| In ethyl acetate at 25 - 60℃; | 95% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene at 85 - 90℃; | 93% |
| With phosphorus pentaoxide | |
| at 155 - 165℃; under 15 Torr; |

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; benzotriazol-1-ol; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; | 93% |
-
-
58859-46-4
ethyl 4-aminopiperidine-1-carboxylate

-
-
76-93-7
Benzilic acid

-
-
160099-10-5
N-(1-carboethoxy-4-piperidyl)-2-hydroxy-2,2-diphenylacetamide

| Conditions | Yield |
|---|---|
| With 1,1'-carbonyldiimidazole In dichloromethane for 0.75h; Ambient temperature; | 92.5% |
-
-
76-93-7
Benzilic acid

-
-
6295-06-3
butyl glyoxalate

-
-
78733-54-7
2-(carbobutoxy)-5,5-diphenyl-1,3-dioxolan-4-one

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 92.2% |

| Conditions | Yield |
|---|---|
| With triethylamine In benzene byproducts: Et3NHCl; elem. anal.; | 92% |
| With triethylamine In benzene (inert atm.); room temp. 2h; filtn., evapn., sublimation (195°C/760 mm Hg); | 67% |

-
-
76-93-7
Benzilic acid

-
-
110-97-4
diisopropanolamine

-
-
162441-07-8
(α-hydroxydiphenylaceto-O,O')(iminodiisopropanolato-N,O,O')germanium(IV)

| Conditions | Yield |
|---|---|
| In ethanol; water; xylene refluxing (2 h); solvent removal, crystn. on cooling, filtn., drying (vac.); elem. anal.; | 92% |


| Conditions | Yield |
|---|---|
| With sulfuric acid at 40℃; for 3h; | 92% |


| Conditions | Yield |
|---|---|
| With sulfuric acid; acetic acid at 20℃; for 5h; Ritter reaction; | 91% |
-
-
54856-83-6
3-dimethylamino-2,2-dimethyl-2H-azirine

-
-
76-93-7
Benzilic acid

-
-
111492-15-0
2-(2-Hydroxy-2,2-diphenylacetamido)-N,N,2-trimethylpropionamid

| Conditions | Yield |
|---|---|
| In acetonitrile Ambient temperature; | 90.5% |
-
-
76-93-7
Benzilic acid

-
-
74-95-3
1,2-dibromomethane

-
-
111008-65-2
Hydroxy-diphenyl-acetic acid 2-hydroxy-2,2-diphenyl-acetoxymethyl ester

| Conditions | Yield |
|---|---|
| With anion exchange resin for 5h; Heating; | 90% |

| Conditions | Yield |
|---|---|
| With sulfuric acid Reflux; regioselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With thionyl chloride for 10h; Product distribution / selectivity; Reflux; | 90% |

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In methanol for 0.25h; Microwave irradiation; Reflux; | 90% |

| Conditions | Yield |
|---|---|
| In benzene byproducts: ethanol; 10 h reflux, molar ratio of educts=1:2; ethanol recovered as azeotrope, elem. anal.; | 88% |

| Conditions | Yield |
|---|---|
| at 60℃; for 2h; Reflux; | 87% |

Benzilic acid Consensus Reports
Reported in EPA TSCA Inventory.
Benzilic acid Specification
The Benzilic acid with CAS registry number of 76-93-7 is also called Benzeneacetic acid,a-hydroxy-a-phenyl-. Its EINECS registry number is 200-993-2. The IUPAC name is 2-hydroxy-2,2-diphenylacetic acid. In addition, the molecular formula is C14H12O3 and the molecular weight is 228.24. It is a kind of a white crystalline aromatic acid and soluble in many primary alcohols. In addition, it belongs to the classes of Pharmaceutical Intermediates; Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts; Organics.
Physical properties about this chemical are: (1)ACD/LogP: 3.03; (2)ACD/LogD (pH 5.5): 0.14; (3)ACD/LogD (pH 7.4): -0.69; (4)ACD/BCF (pH 5.5): 1; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 1.37; (7)ACD/KOC (pH 7.4): 1; (8)#H bond acceptors: 3; (9)#H bond donors: 2; (10)#Freely Rotating Bonds: 4; (11)Polar Surface Area: 35.53 Å2; (12)Index of Refraction: 1.622; (13)Molar Refractivity: 62.87 cm3; (14)Molar Volume: 178.3 cm3; (15)Polarizability: 24.92 ×10-24cm3; (16)Surface Tension: 56.3 dyne/cm; (17)Density: 1.279 g/cm3; (18)Flash Point: 215.3 °C; (19)Enthalpy of Vaporization: 69.71 kJ/mol; (20)Boiling Point: 409 °C at 760 mmHg; (21)Vapour Pressure: 2.01E-07 mmHg at 25°C.
Preparation of Benzilic acid: It can be prepared by heating mixture of benzil, alcohol and potassium hydroxide. And it can be prepared by diphenylethanedione. This reaction will need reagent NaOH and solvents ethanol and H2O. The reaction time is 30 minutes by heating at reaction. The yield is about 97%.
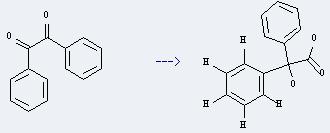
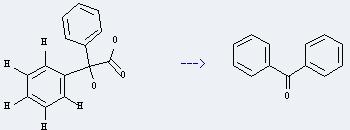
Uses of Benzilic acid: it can be used to determine the zirconium. And it can be used to get benzophenone. This reaction will need reagents sodium metavanadate and 70percent aq.HClO4. The reaction time is 10 minutes by heating. The yield is about 80%.
When you are using this chemical, please be cautious about it as the following:
This chemical is harmful by inhalation, in contact with skin and if swallowed. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. Whenever you will contact it, please wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)C(O)(c1ccccc1)c2ccccc2
(2)InChI: InChI=1/C14H12O3/c15-13(16)14(17,11-7-3-1-4-8-11)12-9-5-2-6-10-12/h1-10,17H,(H,15,16)
(3)InChIKey: UKXSKSHDVLQNKG-UHFFFAOYAV
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 2gm/kg (2000mg/kg) | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 116, Pg. 154, 1958. | |
| mouse | LD50 | subcutaneous | 1300mg/kg (1300mg/kg) | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 116, Pg. 154, 1958. |
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View