-
Name
CROCONIC ACID
- EINECS
- CAS No. 488-86-8
- Density 2.311 g/cm3
- Solubility soluble in water and ethanol
- Melting Point >300 °C(lit.)
- Formula C5H2O5
- Boiling Point 291.7 °C at 760 mmHg
- Molecular Weight 142.068
- Flash Point 144.4 °C
- Transport Information
- Appearance gold-coloured powder
- Safety 37/39-26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Croconicacid (6CI);4,5-Dihydroxy-4-cyclopentene-1,2,3-trione;
- PSA 91.67000
- LogP -0.96510
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: maleic anhydride With sodium hydroxide; bromine In water at 0 - 20℃; for 1.66667h; Stage #2: With hydrogenchloride In water at 0℃; Product distribution / selectivity; | 88% |
| Stage #1: maleic anhydride With water; chlorine; potassium carbonate at 0 - 50℃; for 6h; Stage #2: With hydrogenchloride at 10℃; Product distribution / selectivity; | 66% |

| Conditions | Yield |
|---|---|
| Stage #1: cyclopentene With potassium hydroxide; bromine In water; benzene at 0 - 60℃; for 5h; Stage #2: With hydrogenchloride In water; benzene at 5℃; Product distribution / selectivity; | 83% |
-
-
15849-14-6
ccyclopentane-1,2,4-trione

-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| Stage #1: ccyclopentane-1,2,4-trione With 1,3-dichloro-5,5-dimethylhydantoin In 1,2-dichloro-ethane at 0℃; for 2h; Stage #2: With sodium hydroxide In water; 1,2-dichloro-ethane at 0 - 50℃; for 4.5h; Stage #3: With hydrogenchloride In water; 1,2-dichloro-ethane at 0℃; Product distribution / selectivity; | 63% |

| Conditions | Yield |
|---|---|
| With nitric acid |

| Conditions | Yield |
|---|---|
| With oxygen In water Rate constant; Quantum yield; Irradiation; various pH; |

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; potassium carbonate |

| Conditions | Yield |
|---|---|
| Abdampfen an der Luft; |

| Conditions | Yield |
|---|---|
| Abdampfen an der Luft; |

-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| With air; water at 80℃; |

-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| With air; water at 80℃; |

-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide; potassium carbonate |
-
-
87-89-8
D-myo-inositol

-
-
7697-37-2
nitric acid

-
A
-
608-80-0
benzenehexol

-
B
-
488-86-8
croconic acid

-
C
-
118-76-3
rhodizonic acid


| Conditions | Yield |
|---|---|
| beim Aufbewahren; |
-
-
114491-15-5
triaminophloroglucinol hydrogen sulfate

-
-
488-86-8
croconic acid


-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid In water for 1h; warming; |

| Conditions | Yield |
|---|---|
| In water at 20℃; under 0 Torr; for 8h; Flow reactor; | 99.1% |

-
-
488-86-8
croconic acid

-
-
109-77-3
malononitrile

-
-
72905-24-9
1,2,3-tris(dicyanomethylene)-4-cyclopentene-4,5-diol

| Conditions | Yield |
|---|---|
| In water at 85 - 90℃; for 0.25h; | 97% |
-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol at 107℃; for 3h; Inert atmosphere; | 94.8% |

| Conditions | Yield |
|---|---|
| In propan-1-ol; toluene for 2h; Reflux; | 93% |

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol for 1h; Reflux; | 91% |
-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol at 110℃; for 3h; Dean-Stark; | 87% |
-
-
41532-84-7
2,3,3-trimethylbenzo[e]indole

-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| In water; acetone at 50℃; for 96h; Solvent; Temperature; | 86% |

| Conditions | Yield |
|---|---|
| With quinoline In ethanol at 80℃; for 24h; | 85% |

| Conditions | Yield |
|---|---|
| With quinoline In ethanol at 80℃; for 24h; | 85% |

| Conditions | Yield |
|---|---|
| In methanol at 60℃; | 85% |
-
-
488-86-8
croconic acid

-
-
100362-83-2
5-bromo-2-methyl-3-octyl-1,3-benzothiazol-3-ium iodide

| Conditions | Yield |
|---|---|
| In water; acetone at 0 - 3℃; for 48h; | 85% |

| Conditions | Yield |
|---|---|
| With NaOH In water byproducts: NaCl, NaBr; NaOH and (C4H9)4NBr added to C5O3(OH)2*3H2O in H2O, FeCl3 added dropwise; extd. (CH2Cl2), anhyd. Na2SO4 added, filtered, evapd. (vac.), crystd. (MeOH/Et2O) for days; elem. anal.; | 83% |


| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol for 1h; Reflux; | 82% |

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol for 2h; Reflux; | 81% |
-
-
54136-24-2
5-bromo-2,3,3-trimethyl-3H-indole

-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol at 110℃; for 3h; Dean-Stark; | 80% |

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol for 1h; Inert atmosphere; Reflux; | 79.9% |
-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| With calcium acetate In water at 180℃; for 2h; | 79% |

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol for 1h; Inert atmosphere; Reflux; | 79% |
-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol Reflux; | 78% |
-
-
488-86-8
croconic acid

-
-
95-54-5
1,2-diamino-benzene

-
-
23774-23-4
2,3-dihydroxy-1H-cyclopenta[b]quinoxalin-1-one

| Conditions | Yield |
|---|---|
| With acetic acid at 40℃; for 2h; Inert atmosphere; | 78% |

| Conditions | Yield |
|---|---|
| In butan-1-ol; benzene Heating; | 76% |
-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| With pyridine In methanol for 3h; Condensation; Heating; | 75% |

-
-
488-86-8
croconic acid

-
-
1005495-54-4
C83H108N2O9

| Conditions | Yield |
|---|---|
| With quinoline In ethanol at 80℃; for 24h; | 75% |

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol for 1h; Inert atmosphere; Reflux; | 74% |

| Conditions | Yield |
|---|---|
| With NaOH In water NaOH and P(C6H5)4Br added to C5O3(OH)2*3H2O in H2O, FeCl3 added dropwise; extd. (CH2Cl2), anhyd. Na2SO4 added, filtered, evapd. (vac.), crystd. (MeOH/Et2O); elem. anal.; | 72% |

-
-
488-86-8
croconic acid

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol at 80℃; for 1h; | 71% |

| Conditions | Yield |
|---|---|
| In toluene; butan-1-ol Heating; | 70% |

| Conditions | Yield |
|---|---|
| In butan-1-ol; benzene Heating; | 70% |
Croconic acid History
In 1825, Croconic acid and potassium croconate dihydrate were discovered by Leopold Gmelin, who named the compounds from Greek meaning "saffron" or "egg yolk". In 1964, the structure of ammonium croconate was determined by Baenziger et al. . The structure of K2C5O5 . 2H2O was determined by J. D. Dunitz in 2001.
Croconic acid Specification
The Croconic acid, with the CAS registry number 488-86-8, is also known as UNII-6HGY35UXAA. This chemical's molecular formula is C5H2O5 and molecular weight is 142.07. Its IUPAC name is called 4,5-dihydroxycyclopent-4-ene-1,2,3-trione. Croconic acid also forms esters such as dimethyl croconate.
Physical properties of Croconic acid: (1)ACD/LogP: -2.23; (2)ACD/LogD (pH 5.5): -6.72; (3)ACD/LogD (pH 7.4): -6.72; (4)ACD/BCF (pH 5.5): 1; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 1; (7)ACD/KOC (pH 7.4): 1; (8)#H bond acceptors: 5; (9)#H bond donors: 2; (10)#Freely Rotating Bonds: 2; (11)Index of Refraction: 1.798; (12)Molar Refractivity: 26.22 cm3; (13)Molar Volume: 61.4 cm3; (14)Surface Tension: 197.1 dyne/cm; (15)Density: 2.311 g/cm3; (16)Flash Point: 144.4 °C; (17)Enthalpy of Vaporization: 61.63 kJ/mol; (18)Boiling Point: 291.7 °C at 760 mmHg; (19)Vapour Pressure: 0.000206 mmHg at 25°C.
Uses of Croconic acid: it can be used to produce 1,3-bis-(2-dimethylamino-5-thienyl)croconine with dimethyl-thiophen-2-yl-amine by heating. This reaction will need solvents butan-1-ol and toluene. The yield is about 55%.
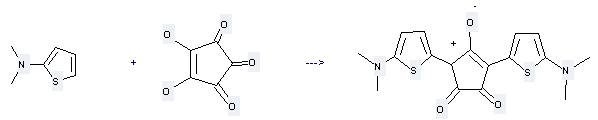
When you are using this chemical, please be cautious about it as the following:
This chemical may cause inflammation to the skin or other mucous membranes. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. Whenever you will contact it, please wear suitable protective gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: C1(=C(C(=O)C(=O)C1=O)O)O
(2)InChI: InChI=1S/C5H2O5/c6-1-2(7)4(9)5(10)3(1)8/h6-7H
(3)InChIKey: RBSLJAJQOVYTRQ-UHFFFAOYSA-N
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View