-
Name
Cyclododecanone
- EINECS 212-595-6
- CAS No. 830-13-7
- Article Data250
- CAS DataBase
- Density 0.87 g/cm3
- Solubility Insoluble in water
- Melting Point 59-61 °C(lit.)
- Formula C12H22O
- Boiling Point 261.3 °C at 760 mmHg
- Molecular Weight 182.306
- Flash Point 108.8 °C
- Transport Information UN 3077 9/PG 3
- Appearance colourless to white adhering crystals
- Safety 60-61
- Risk Codes 51/53
-
Molecular Structure
-
Hazard Symbols
 N
N
- Synonyms NSC 77116;Cyclododecan-1-one;
- PSA 17.07000
- LogP 3.86020
Synthetic route

| Conditions | Yield |
|---|---|
| With bis-trimethylsilanyl peroxide; dipyridinium dichromate In dichloromethane at 25℃; | 100% |
| With tert.-butylhydroperoxide; dimesityl diselenide In benzene for 4h; Heating; | 100% |
| With bis-trimethylsilanyl peroxide; dipyridinium dichromate In dichloromethane for 0.5h; | 100% |
-
-
65763-18-0
cis,trans-4,8-cyclododecadien-1-one

-
-
65763-17-9
trans,cis-4,8-cyclododecadien-1-one

-
-
110307-32-9
trans,trans-cyclododeca-4,8-dienone

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With hydrogen; 5%-palladium/activated carbon In methanol at 60 - 70℃; under 7500.75 Torr; for 24h; | 99.5% |
| With hydrogen; palladium 10% on activated carbon at 50℃; under 3750.38 Torr; for 10h; | 98% |

-
-
70015-94-0
1,4-oxathiaspiro<4.11>hexadecane

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With eosin Y disodium salt In acetonitrile at 20℃; for 3h; Irradiation; | 99% |
| With perchloric acid; dihydrogen peroxide; ammonium bromide; molybdic acid In dichloromethane at 0 - 5℃; for 4h; | 96% |
| Stage #1: 1,4-oxathiaspiro<4.11>hexadecane With acetyl chloride; sodium nitrite In dichloromethane at 0 - 5℃; for 0.25h; Stage #2: With water In dichloromethane at 0 - 20℃; for 0.166667h; | 95% |

| Conditions | Yield |
|---|---|
| With Oxone; potassium bromide In acetonitrile at 0 - 30℃; for 24h; Darkness; Green chemistry; | 99% |
| With silica gel; Nitrogen dioxide In tetrachloromethane for 0.5h; Ambient temperature; | 88% |
| With silica gel; Nitrogen dioxide In tetrachloromethane for 0.5h; Product distribution; Mechanism; Ambient temperature; var. ethers; | 88% |
-
-
1616490-83-5
C19H29BrO

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With Oxone; potassium bromide In acetonitrile at 0 - 30℃; for 24h; Darkness; Green chemistry; | 99% |
-
-
42858-38-8
(E)/(Z)-cyclododec-2-enone

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With enone reductase from Sporidiobolus salmonicolor TPU 2001; NADH In aq. phosphate buffer at 30℃; for 1h; pH=7; Kinetics; Enzymatic reaction; | 98.8% |
-
-
42858-38-8
(E)-cyclododec-2-enone

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With triethyl borane; triphenylstannane In hexane; benzene at 25℃; for 6h; | 98% |
| With triethyl borane; triphenylstannane In hexane; benzene at 25℃; for 6h; other substrates; | 98% |
-
-
24459-40-3
2,12-dibromocyclododecanone

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; sodium iodide In acetonitrile for 12h; Ambient temperature; | 98% |
-
-
14990-47-7
cyclododecanone semicarbazone

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| Amberlyst 15 In water; acetone at 80℃; for 15h; | 98% |
-
-
85895-36-9
1,1-bis(phenylthio)cyclododecanone

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid; trifluoroacetic acid In dichloromethane for 1h; Ambient temperature; | 98% |

| Conditions | Yield |
|---|---|
| With polyethylene glycol-400; lithium bromide at 194℃; for 4h; | 98% |
| With Pd/ZrO2 at 220 - 230℃; | 98% |
| In nitrogen; toluene | 94% |

| Conditions | Yield |
|---|---|
| With iodine In acetone at 56℃; for 5h; | 97% |
| palladium (II) ion In acetone for 1h; Ambient temperature; | 96 % Chromat. |
-
-
160036-85-1
{(E)-3-[1-(2-Methoxy-ethoxymethoxy)-cyclododecyl]-propenyl}-trimethyl-silane

-
A
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78℃; for 0.5h; | A 97% B 2% |
-
-
60719-91-7
1,5-dithiaspiro<5.11>heptadecane

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With iron(III)-acetylacetonate; dihydrogen peroxide; sodium iodide In water; ethyl acetate at 20℃; for 2.8h; | 97% |
| Stage #1: 1,5-dithiaspiro<5.11>heptadecane With trichloroisocyanuric acid; silica gel at 20℃; for 0.05h; Stage #2: With water at 20℃; | 89% |
| With dihydrogen peroxide; tantalum pentachloride; sodium iodide In water; ethyl acetate at 20℃; for 5h; | 89% |

| Conditions | Yield |
|---|---|
| With lithium iodide at 140℃; for 3h; | 96% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; potassium carbonate 1.) MeOH, 0 deg C, H2O 2.) 8h, room temp.; | 96% |
| With potassium permanganate; sodium hydride; magnesium sulfate; tert-butyl alcohol 1.) pentane, 20 min, 2.) water, 10 min; Yield given. Multistep reaction; |
-
-
31236-94-9
2-bromocyclododecan-1-one

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With tetrahydrofuran; chromium chloride; lithium aluminium tetrahydride In tetrahydrofuran for 3.5h; Ambient temperature; | 96% |
| With DMBI In 1,4-dioxane for 52h; Heating; | 95% |
-
-
88358-52-5
2-(tert-Butyl-dimethyl-silanyloxy)-3-methyl-1-oxa-4-thia-2-aza-spiro[4.11]hexadecane

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran | 96% |
-
-
1207858-28-3
C18H34N2O

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide; dihydrogen peroxide In methanol at 20℃; pH=7; aq. phosphate buffer; | 96% |
-
-
104151-45-3
2-(1-Hydroxy-cyclododecyl)-3-methyl-benzothiazol-3-ium; iodide

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In hexane at 60℃; for 2h; | 95% |
-
-
115663-67-7
1,1-Bis-ethylsulfanyl-cyclododecane

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid; trifluoroacetic acid In dichloromethane for 1h; Ambient temperature; | 95% |
| Stage #1: 1,1-Bis-ethylsulfanyl-cyclododecane With acetyl chloride; sodium nitrite In dichloromethane at 0 - 5℃; for 0.25h; Stage #2: With water In dichloromethane at 0 - 20℃; for 0.5h; | 78% |
| With cetyl(trimethyl)-ammonium tribromide In dichloromethane at 0 - 5℃; for 0.25h; | 75% |
-
-
78804-10-1
cyclododecanone dimethylhydrazone

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With sodium perborate; sodium hydroxide; potassium dihydrogenphosphate; water In tert-butyl alcohol at 60℃; for 12h; | 95% |

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate; Montmorillonite K10; silver nitrate In hexane at 50℃; for 2.5h; | 95% |
| With silica gel; chromic acid In dichloromethane | 93% |
| With L-alanin; silica gel; chlorochromic acid In dichloromethane at 20℃; for 5h; | 90% |
-
-
121758-10-9
(phenylmethoxy)cyclododecane

-
A
-
100-52-7
benzaldehyde

-
B
-
830-13-7
cyclododecanone

-
C
-
65-85-0
benzoic acid

| Conditions | Yield |
|---|---|
| With Oxone; potassium bromide In acetonitrile at 0 - 30℃; for 24.5h; Solvent; Temperature; Reagent/catalyst; Darkness; Green chemistry; | A 21% B 95% C 33% |

-
-
42858-37-7
2-(phenylseleno)cyclododecan-1-one

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; nickel dichloride In tetrahydrofuran; methanol at 0℃; for 0.25h; | 93% |

| Conditions | Yield |
|---|---|
| Envirocat EPZG In water; acetone at 80℃; for 3h; | 93% |
| With 1-methyl-1H-imidazole; oxygen; pivalaldehyde; (NMe4)2[Ni(Me2opba)]*4H2O In fluorobenzene at 20℃; for 24h; Oxidation; | 82% |
-
-
69381-33-5
α-iodocyclododecanone

-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With diphosphorus tetraiodide In dichloromethane at 25℃; for 1h; | 92% |

| Conditions | Yield |
|---|---|
| Amberlyst 15 In water; acetone at 80℃; for 5h; | 92% |

| Conditions | Yield |
|---|---|
| With calcium chloride In 1,4-dioxane; water at 19.85 - 179.85℃; under 7575.61 Torr; for 0.666667h; | 92% |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; sodium cyanoborohydride In tetrahydrofuran at 65℃; for 11h; | 100% |
| With aluminum oxide; sodium tetrahydroborate In hexane at 60℃; for 3h; | 98% |
| With tricarbonyl(η4-1,3-bis(trimethylsilyl)-4,5,6,7-tetrahydro-2H-inden-2-one)iron; hydrogen; potassium carbonate In water; isopropyl alcohol at 100℃; under 22502.3 Torr; for 17h; Inert atmosphere; | 98% |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
830-13-7
cyclododecanone

-
-
50361-51-8
1-trimethylsilyloxy-1-cyclododecanecarbonitrile

| Conditions | Yield |
|---|---|
| With zinc(II) iodide | 100% |
| With lithium perchlorate; tetraethylammonium perchlorate In dichloromethane Ambient temperature; electrolysis: 0.28 F/mol; | 95% |
| With n-butyllithium In tetrahydrofuran; hexane for 2h; Ambient temperature; | 90.6% |
| With zinc(II) iodide |

| Conditions | Yield |
|---|---|
| With N-methylanilinium trifluoroacetate In 1,4-dioxane at 100℃; for 4h; | 100% |
| With N-methylanilinium trifluoroacetate In 1,4-dioxane for 4.5h; Heating; | 48% |
-
-
50635-71-7, 50635-72-8, 31350-93-3
1-chloroethyl p-tolyl sulfoxide

-
-
830-13-7
cyclododecanone

-
-
159763-15-2
1-<1-chloro-1-(p-tolylsulfinyl)ethyl>-1-cyclododecanol

| Conditions | Yield |
|---|---|
| at -70℃; | 100% |
| With n-butyllithium; diisopropylamine In tetrahydrofuran at -65℃; for 0.416667h; | 85% |

| Conditions | Yield |
|---|---|
| With pyridine; hydroxylamine hydrochloride In ethanol at 90 - 100℃; for 2h; | 100% |
| With hydroxylamine hydrochloride; sodium acetate In ethanol; water Reflux; | 99% |
| With ammonium chloride; Amberlyst A-21 In ethanol for 1h; Ambient temperature; | 96% |
-
-
830-13-7
cyclododecanone

-
-
35951-28-1
2-chlorocyclododecanone

| Conditions | Yield |
|---|---|
| Stage #1: cyclododecanone With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: With methyl chlorosulfate In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; | 100% |
| With p-toluenesulfonyl chloride; lithium diisopropyl amide In tetrahydrofuran -78 deg C to room t.; | 83% |
| With [bis(acetoxy)iodo]benzene; iron(III) chloride hexahydrate In acetic acid at 20℃; for 18h; Inert atmosphere; | 79% |
-
-
830-13-7
cyclododecanone

-
-
1576-35-8
toluene-4-sulfonic acid hydrazide

-
-
3552-02-1
N'-cyclododecylidene-4-methylbenzenesulfonohydrazide

| Conditions | Yield |
|---|---|
| In ethanol at 100℃; for 1h; | 100% |
| In ethanol at 100℃; for 1.66667h; Inert atmosphere; | 100% |
| In methanol at 20℃; Schlenk technique; | 100% |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; N-ethyl-N,N-diisopropylamine In dichloromethane at -78℃; for 2h; | 100% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol for 0.5h; Reflux; | 100% |
| With acetic acid In ethanol for 24h; Reflux; |
-
-
830-13-7
cyclododecanone

| Conditions | Yield |
|---|---|
| With sodium hydroxide; trimethyl orthoformate In 2-methoxy-ethanol | 99.8% |

| Conditions | Yield |
|---|---|
| With manganese; chloro-trimethyl-silane; lead(II) chloride In tetrahydrofuran at 25℃; for 0.5h; Product distribution; further times, reagent ratios; | 99% |
| With manganese; chloro-trimethyl-silane; lead(II) chloride In tetrahydrofuran at 25℃; for 0.5h; | 99% |
| Stage #1: allyl bromide With indium; aluminium In tetrahydrofuran at 25℃; for 0.5h; Stage #2: cyclododecanone In tetrahydrofuran at 10℃; for 0.5h; Further stages.; | 98% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 99% |
| With toluene-4-sulfonic acid In toluene Reflux; | 99% |
| With boron trifluoride diacetate for 0.25h; | 95% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| With trimethyl orthoformate at 20℃; for 1h; | 99% |
-
-
830-13-7
cyclododecanone

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With La(Cp)3 In tetrahydrofuran at 20℃; for 2h; Reagent/catalyst; Inert atmosphere; | 99% |
| With [RhCl2(p-cymene)]2 In benzene at 60℃; for 15h; Inert atmosphere; Glovebox; chemoselective reaction; | |
| With tris(cyclopentadienyl)lanthanum(III) In tetrahydrofuran at 25℃; for 1h; Inert atmosphere; Schlenk technique; Glovebox; | |
| With tris(η5-methylcyclopentadienyl)lanthanum(III) In tetrahydrofuran at 20℃; for 0.166667h; Catalytic behavior; Schlenk technique; Glovebox; Inert atmosphere; chemoselective reaction; | 99 %Spectr. |
| With La(Cp)3 In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; |
-
-
2942-58-7
diethyl cyanophosphonate

-
-
2408-36-8
lithium cyanide

-
-
830-13-7
cyclododecanone

-
-
88151-69-3
1-cyanocyclododecyl diethylphosphate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 0.0833333h; | 99% |

-
-
75-77-4
chloro-trimethyl-silane

-
-
830-13-7
cyclododecanone

-
-
55314-44-8
[((E)-Cyclododec-1-enyl)oxy]-trimethyl-silane

| Conditions | Yield |
|---|---|
| With triethylamine; sodium iodide In acetonitrile; pentane at 20℃; for 12h; | 98% |
| With magnesium In N,N-dimethyl-formamide at 15 - 25℃; | 80% |
| With triethylamine In N,N-dimethyl-formamide at 120℃; |
-
-
830-13-7
cyclododecanone

-
-
65-45-2
salicylamide

-
-
77773-97-8
Spiro<2H-1,3-benzoxazine-2,1'-cyclododecan>-4(3H)-one

| Conditions | Yield |
|---|---|
| 98% |
-
-
830-13-7
cyclododecanone

-
-
109-94-4
formic acid ethyl ester

-
-
949-07-5
(Z)-2-(hydroxymethylidene)cyclododecanone

| Conditions | Yield |
|---|---|
| Stage #1: formic acid ethyl ester With potassium tert-butylate In tetrahydrofuran at 0℃; Stage #2: cyclododecanone In tetrahydrofuran at 0 - 20℃; | 98% |
| With sodium methylate In diethyl ether for 12h; Ambient temperature; |
-
-
830-13-7
cyclododecanone

-
-
60223-10-1
1,1-dichlorocyclododecane

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride In benzene at 0 - 20℃; for 28h; | 98% |
Cyclododecanone Specification
This product is an organic compound with the formula C12H22O. The systematic name of this chemical is Cyclododecanone. It belongs to the product categories of C11 to C12; Carbonyl Compounds; Ketones. Its EINECS number is 212-595-6. With the CAS registry number 830-13-7, it is also named as Cyclododecan-1-one. In addition, the molecular weight is 225.60. It is stable at common pressure and temperature, and it should be sealed and stored in a cool and dry place. Moreover, it should be protected from strong oxidants. It is used as organic synthetic raw material, especially the intermediate of Nylon-12.
Physical properties of Cyclododecanone are: (1)ACD/LogP: 4.15; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 4.15; (4)ACD/LogD (pH 7.4): 4.15; (5)ACD/BCF (pH 5.5): 836.29; (6)ACD/BCF (pH 7.4): 836.29; (7)ACD/KOC (pH 5.5): 4299.57; (8)ACD/KOC (pH 7.4): 4299.57; (9)#H bond acceptors: 1; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 17.07 Å2; (13)Index of Refraction: 1.442; (14)Molar Refractivity: 55.47 cm3; (15)Molar Volume: 209.3 cm3; (16)Polarizability: 21.99×10-24cm3; (17)Surface Tension: 29 dyne/cm; (18)Density: 0.87 g/cm3; (19)Flash Point: 108.8 °C; (20)Enthalpy of Vaporization: 49.9 kJ/mol; (21)Boiling Point: 261.3 °C at 760 mmHg; (22)Vapour Pressure: 0.0116 mmHg at 25°C.
Preparation of Cyclododecanone: this chemical can be prepared by cyclododecanol at the ambient temperature. This reaction will need reagents oxygen, acetaldehyde. This reaction will also need catalysts RuCl3·nH2O, Co(OAc)2·4H2O. The yield is about 97%.

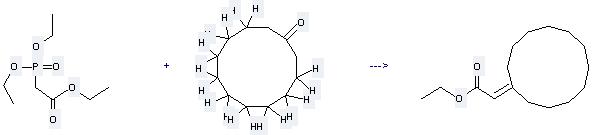
Uses of Cyclododecanone: it can be used to produce cyclododecylideneacetic acid ethyl ester at the temperature of 20 °C. It will need reagent NaH and solvent tetrahydrofuran with the reaction time of 8 hours. The yield is about 96%.
When you are using this chemical, please be cautious about it as the following:
It is toxic to aquatic organisms as it may cause long-term adverse effects in the aquatic environment. This material and its container must be disposed of as hazardous waste. You should avoid releasing it to the environment just refering to special instructions/safety data sheet.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C1CCCCCCCCCCC1
(2)Std. InChI: InChI=1S/C12H22O/c13-12-10-8-6-4-2-1-3-5-7-9-11-12/h1-11H2
(3)Std. InChIKey: SXVPOSFURRDKBO-UHFFFAOYSA-N
Related Products
- Cyclododecanone
- Cyclododecanone,2,12-dibromo-
- Cyclododecanone,2-nitro-
- 83014-44-2
- 83015-26-3
- 83015-28-5
- 83015-88-7
- 83016-76-6
- 83020-74-0
- 83023-58-9
- 83024-92-4
- 83027-13-8
- 83027-36-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View