Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:25015-63-8
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:25015-63-8
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:25015-63-8
Min.Order:1
Negotiable
Type:Other
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:25015-63-8
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 6 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:25015-63-8
Min.Order:1 Kilogram
FOB Price: $1000.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:25015-63-8
Min.Order:1 Kilogram
FOB Price: $9.0 / 99.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:25015-63-8
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryLIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:25015-63-8
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Products Name: 4,4,5,5-tetramethyl-1,3,2-dioxa-borolane CAS NO.: 25015-63-8 Molecular formula: C6H13BO2 Molecular weight: 127.9772 Appearance:White powder Storage:S
Cas:25015-63-8
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:25015-63-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Pinacolborane CAS:25015-63-8 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermedia
Cas:25015-63-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hebei Mojin Biotechnology Co.,Ltd
Product quality: Our company have high quality product , and also the product we have good manufacture . First of all, this product is of fine quality. Every finish should be checked by quality inspection system.And every one should be also tried
Cas:25015-63-8
Min.Order:1 Kilogram
FOB Price: $60.0 / 70.0
Type:Trading Company
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:25015-63-8
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Afine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:25015-63-8
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:25015-63-8
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Lingrui Chemical Co.,Ltd.
China cheap price 4,4,5,5-tetramethyl-1,3,2-dioxa-borolaneAppearance:Colorless transparent liquid Storage:Store in dry, dark and ventilated place. Package:according to customers' requirements Application:chemical research Transportation:By air(EMS or
Cas:25015-63-8
Min.Order:1 Gram
Negotiable
Type:Other
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality Appearance:Clear colorless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:It is used for synthesis of unsymmetrical biary
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:25015-63-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
Product Name: Pinacolborane Synonyms: Pinacolatoborane;TetraMethyl-1,3,2-dioxab;Pinacolborane, 97%, stabilized;4,4,5,5-TetraMethyl-1,3,2-dioxaborolane 97%;4,4,5,5-TetraMethyl-1,3,2-dioxaborolane solution 1.0 M in THF;Pinacolborane (4,4,5,5-Tetr
Cas:25015-63-8
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryEAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:25015-63-8
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Hangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Cas:25015-63-8
Min.Order:0
Negotiable
Type:Manufacturers
inquirySuzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:This product can be use
Synthetic route
-
-
1008-89-5
2-phenylpyridine

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
1349171-28-3
2-(2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)pyridine

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With catalyst: [Rh(OH)((CHCHC2H4)2)]2/Silica-SMAP In tetrahydrofuran for 1 h at room temp.; | A 98% B n/a |


-
-
1076-43-3
benzene-d6

-
A
-
680192-98-7
deutero-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| In benzene-d6 (N2); heated at 55°C for 90 min; not isolated, detected by NMR; | A n/a B n/a C 95% |

-
-
637-92-3
Ethyl tert-butyl ether

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
B
-
255041-56-6
2-(2-tert-butoxyethyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| (η5-pentamethylcyclopentadienyl)(η6-hexamethylbenzene)rhodium(I) In neat (no solvent) for 24 h at 150°C; | A n/a B 91% |
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium In pentane at 25℃; under 1520 Torr; for 46h; Borylation; Irradiation; | A 26 % Spectr. B 75% |
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium In neat (no solvent) Irradiation (UV/VIS); 25°C, 46 h; chromy. (SiO2; pentane or pentane/Et2O); | A n/a B 75% |
| (η5-pentamethylcyclopentadienyl)(η6-hexamethylbenzene)rhodium(I) In cyclohexane for 24 h at 150°C; | A n/a B 48% |
| bis[dichloro(pentamethylcyclopentadienyl)ruthenium(III)] In octane 150°C, 24 h; |

-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
-
10325-39-0
dichloroborane

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With triethylamine In 1,4-dioxane at 0 - 20℃; for 12h; pH=12; Solvent; pH-value; Reagent/catalyst; Concentration; Inert atmosphere; | 91% |
-
-
68654-52-4
2-(piperidin-1-yl)pyridine

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
1391930-84-9
2-(2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)piperidin-1-yl)pyridine

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With methoxy(cyclooctadiene)rhodium(I) dimer In tetrahydrofuran at 80℃; for 15h; Inert atmosphere; Glovebox; | A 89% B n/a |
| With methoxy(cyclooctadiene)rhodium(I) dimer In hexane at 80℃; for 12h; Inert atmosphere; regioselective reaction; |

-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
-
13292-87-0
dimethylsulfide borane complex

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With 2,6-di-tert-butyl-4-methyl-phenol In dichloromethane at -10 - 0℃; Inert atmosphere; | 88.8% |
| In dichloromethane at 0 - 20℃; for 13h; Inert atmosphere; | 68% |
| In dichloromethane at 0 - 20℃; for 13h; Inert atmosphere; | 68% |
-
-
111-65-9
octane

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
66217-56-9
4,4,5,5-tetramethyl-2-octyl-[1,3,2]dioxaborolane

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With catalyst: [Ir((CHCHC2H4)2)OCH3]2/(CH3)4C12H4N2 In neat (no solvent) for 24 h at 120°C; | A 88% B n/a |
| bis[dichloro(pentamethylcyclopentadienyl)ruthenium(III)] In octane 150°C, 48 h; | A 75% B n/a |
| [bis(2-methylallyl)cycloocta-1,5-diene]ruthenium(II) In octane 150°C, 48 h; |

-
-
109-99-9
tetrahydrofuran

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With (1,5-cyclooctadiene)(methoxy)iridium(I) dimer; 3,4,7,8-Tetramethyl-o-phenanthrolin; potassium tert-butylate at 110℃; for 20h; Reagent/catalyst; Glovebox; Inert atmosphere; Sealed tube; | A n/a B 88% |

-
-
77117-48-7
perfluorooctyl-ethane

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| (η5-pentamethylcyclopentadienyl)(η6-hexamethylbenzene)rhodium(I) In neat (no solvent) for 24 h at 150°C; various product yields for various conditions; | A n/a B 84% |
| bis[dichloro(pentamethylcyclopentadienyl)ruthenium(III)] In octane 150°C, 24 h; |

-
-
1333-74-0
hydrogen

-
-
73183-34-3
bis(pinacol)diborane

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With (tBuPNN)CoCl2 In tetrahydrofuran; diethyl ether at 25℃; under 11251.1 Torr; for 9h; Solvent; Pressure; Autoclave; | 82% |

-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex In dichloromethane at 0 - 20℃; for 16h; Inert atmosphere; | 81% |
| With dimethylsulfide borane complex In dichloromethane at 0 - 20℃; for 1.58333h; Inert atmosphere; | 81% |
| Stage #1: 2,3-dimethyl-2,3-butane diol With borane-dimethyl sulfide complex In dichloromethane at 0℃; for 4h; Stage #2: In dichloromethane at 20℃; | 81.7% |
-
-
73183-34-3
bis(pinacol)diborane

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With [(tBuPNN)FeCl2]; hydrogen; sodium t-butanolate In diethyl ether under 7500.75 Torr; for 2h; Catalytic behavior; Reagent/catalyst; Solvent; Autoclave; | 76% |
| With palladium 10% on activated carbon; hydrogen In pentane at 20℃; under 750.075 Torr; for 20h; Pressure; Temperature; Time; Reagent/catalyst; Solvent; Inert atmosphere; Schlenk technique; Autoclave; | 47% |
-
-
82166-21-0
methyl cyclohexane

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium In pentane at 25℃; under 1520 Torr; for 60h; Borylation; Irradiation; | A 27 % Spectr. B 75% |
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium In neat (no solvent) Irradiation (UV/VIS); 25°C, 60 h; |


-
-
1038-95-5
Tri(p-tolyl)phosphine

-
A
-
26621-57-8
2,2'-(2,3-dimethyl-2,3-butanediyl-dioxy)bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane)

-
C
-
815597-43-4
(pentamethylcyclopentadienyl)RhH(pinacolboryl)P(p-C6H4Me)3

-
D
-
10221-56-4
bis(pinacolato)diborane

-
E
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| heating dihydrido(pentamethylcyclopentadienyl)(dimethylphenylsilyl)(pinacolatoboryl)rhodium at 100°C with 1 equiv. of P(p-tol)3; | A n/a B 75% C 5% D n/a E n/a |


-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
B
-
817160-71-7
6-[2-(2,3-dimethyl-2,3-butanedioxy)hexyl]-4,4,5,5-methyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| (η5-pentamethylcyclopentadienyl)(η6-hexamethylbenzene)rhodium(I) In neat (no solvent) for 24 h at 150°C; various product yields for various conditions; | A n/a B 70% |


-
-
1038-95-5
Tri(p-tolyl)phosphine

-
A
-
26621-57-8
2,2'-(2,3-dimethyl-2,3-butanediyl-dioxy)bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane)

-
C
-
815597-43-4
(pentamethylcyclopentadienyl)RhH(pinacolboryl)P(p-C6H4Me)3

-
D
-
10221-56-4
bis(pinacolato)diborane

-
E
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| byproducts: HSiEt3; heating dihydrido(pentamethylcyclopentadienyl)(triethylsilyl)(pinacolatoboryl)rhodium at 100°C with 1 equiv. of P(p-tol)3; | A n/a B 68% C 13% D n/a E n/a |

-
-
766-09-6
1-ethyl-piperidine

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| (η5-pentamethylcyclopentadienyl)(η6-hexamethylbenzene)rhodium(I) In neat (no solvent) for 24 h at 150°C; various product yields for various conditions; | A n/a B 67% |
| bis[dichloro(pentamethylcyclopentadienyl)ruthenium(III)] In octane 150°C, 24 h; |

-
-
73183-34-3
bis(pinacol)diborane

-
-
109-66-0
pentane

-
A
-
67562-21-4
4,4,5,5-tetramethyl-2-pentyl-1,3,2-dioxaborolane

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium at 25℃; under 1520 Torr; for 56h; Borylation; Irradiation; | A 65% B 32 % Spectr. |
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium In neat (no solvent) Irradiation (UV/VIS); 25°C, 56 h; chromy. (SiO2; pentane or pentane/Et2O); | A 65% B n/a |

-
-
7226-23-5
1,3-dimethyl-3,4,5,6-tetrahydro-2(1H)-pyrimidinone

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
1391930-67-8
1-methyl-3-((4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methyl)tetrahydropyrimidin-2(1H)-one

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With methoxy(cyclooctadiene)rhodium(I) dimer In tetrahydrofuran at 70℃; for 6h; Inert atmosphere; Glovebox; | A 63% B n/a |
| With methoxy(cyclooctadiene)rhodium(I) dimer In hexane at 70℃; for 3h; Inert atmosphere; regioselective reaction; |

-
-
142-96-1
dibutyl ether

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium In pentane at 25℃; under 1520 Torr; for 45h; Borylation; Irradiation; | A 23 % Spectr. B 55% |
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium In neat (no solvent) Irradiation (UV/VIS); 25°C, 45 h; chromy. (SiO2; pentane or pentane/Et2O); | A n/a B 55% |

-
-
463-11-6
1-Fluoro-octane

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| (η5-pentamethylcyclopentadienyl)(η6-hexamethylbenzene)rhodium(I) In neat (no solvent) for 12-24 h at 140-150°C; various product yields for various conditions; | A n/a B 46% |

-
-
2294-76-0
2-pentylpyridine

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
1422450-70-1
2-(2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pentyl)pyridine

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With (1,5-cyclooctadiene)(methoxy)iridium(I) dimer at 60℃; for 15h; Reagent/catalyst; Glovebox; Inert atmosphere; | A 42% B n/a |

-
-
102-82-9
tributyl-amine

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| (η5-pentamethylcyclopentadienyl)(η6-hexamethylbenzene)rhodium(I) In neat (no solvent) for 24 h at 150°C; various product yields for various conditions; | A n/a B 33% |
| chloro(pentamethylcyclopentadienyl)ruthenium(II) tetramer In octane 150°C, 24 h; |

-
-
78-78-4
methylbutane

-
-
73183-34-3
bis(pinacol)diborane

-
A
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium In pentane at 25℃; under 1520 Torr; for 55h; Borylation; Irradiation; | A 24 % Spectr. B n/a C n/a |
| With carbon monoxide; pentamethylcyclopentadienyltricarbonylrhenium In neat (no solvent) Irradiation (UV/VIS); 25°C, 55 h; |

-
-
73183-34-3
bis(pinacol)diborane

-
-
71-43-2
benzene

-
A
-
24388-23-6
2-phenyl-4,4,5,5-tetramethyl-1,3,2-dioxoborole

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With carbon monoxide; [C5H4MeMn(CO)3] In pentane at 25℃; under 1520 Torr; for 36h; Borylation; Irradiation; | A 76 % Spectr. B 23 % Spectr. |
| With carbon monoxide; methylcyclopentadienyl manganese(I) tricarbonyl In neat (no solvent) Irradiation (UV/VIS); 25°C, 36 h; |

-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
A
-
26621-57-8
2,2'-(2,3-dimethyl-2,3-butanediyl-dioxy)bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane)

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With borane-ammonia complex In various solvent(s) at 20 - 25℃; for 0.5h; |
-
-
590-35-2
2,2-dimethylpentane

-
-
73183-34-3
bis(pinacol)diborane

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| bis[dichloro(pentamethylcyclopentadienyl)ruthenium(III)] In octane 150°C, 24 h; |

-
-
255831-64-2, 255041-57-7
[Re(CO)2(BO2C2(CH3)4)2(C5(CH3)5)]

-
-
109-66-0
pentane

-
A
-
67562-21-4
4,4,5,5-tetramethyl-2-pentyl-1,3,2-dioxaborolane

-
B
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| In neat (no solvent) Irradiation (UV/VIS); |


-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With P(CH2CH3)3 Kinetics; react. with excess of PEt3 at 100°C; half-life 2 h; NMR; |

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With P(CH2CH3)3 Kinetics; react. with excess of PEt3 at 100°C; half-life 4.5 h; NMR; |
-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
71-43-2
benzene

-
-
24388-23-6
2-phenyl-4,4,5,5-tetramethyl-1,3,2-dioxoborole

| Conditions | Yield |
|---|---|
| [Ir(COD)(1,3-dicyclohexylimidazolidin-2-ylidene)2]CF3CO2 at 40℃; for 10h; | 100% |
| (η4-1,5-cyclooctadiene)bis(1,3-dimethylimidazolin-2-ylidene)iridium(I) trifluoracetate In benzene (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(C3H2N2Me2)2(CF3CO2) in benzene; stirring and heating at 40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
| (η4-1,5-cyclooctadiene)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)iridium(I) trifluoroacetate In benzene (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)2(CF3CO2) in benzene; stirring and heating at 40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
-
-
108-86-1
bromobenzene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
24388-23-6
2-phenyl-4,4,5,5-tetramethyl-1,3,2-dioxoborole

| Conditions | Yield |
|---|---|
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium acetate In 1,4-dioxane at 85℃; for 6h; Inert atmosphere; | 100% |
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride In methanol at 20℃; for 8h; | 92% |
| Stage #1: bromobenzene; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane With bis(trifluoromethane)sulfonimide lithium In tetrahydrofuran at 20℃; Electrochemical reaction; Stage #2: With sulfuric acid at 0℃; pH=5; Further stages.; | 78% |
-
-
536-74-3
phenylacetylene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
83947-56-2
4,4,5,5-tetramethyl-2-((E)-styryl)-[1,3,2]dioxaborolane

| Conditions | Yield |
|---|---|
| With C39H22BF10OP In dichloromethane-d2 at 20℃; for 12h; Catalytic behavior; Reagent/catalyst; | 100% |
| With tris(2,4,6-trifluorophenyl)borane triethylphosphine oxide In dichloromethane at 60℃; for 6h; Catalytic behavior; Reagent/catalyst; Solvent; Inert atmosphere; Sealed tube; | 99% |
| Stage #1: 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane With C32H41CuN2O2S In toluene at 20℃; for 0.25h; Schlenk technique; Inert atmosphere; Stage #2: phenylacetylene In toluene at 20℃; for 2h; Catalytic behavior; Reagent/catalyst; Schlenk technique; Inert atmosphere; stereoselective reaction; | 98% |
-
-
513-35-9
2-methyl-but-2-ene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With sodium triethylborohydride at 23℃; for 18h; Catalytic behavior; Inert atmosphere; | 100% |
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride In dichloromethane at 20℃; for 24h; | 36% |
| With sodium triethylborohydride In tetrahydrofuran at 23℃; for 18h; Reagent/catalyst; Inert atmosphere; Glovebox; | 100 %Spectr. |
-
-
111-66-0
oct-1-ene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
66217-56-9
4,4,5,5-tetramethyl-2-octyl-[1,3,2]dioxaborolane

| Conditions | Yield |
|---|---|
| With sodium triethylborohydride at 23℃; for 3h; Catalytic behavior; Reagent/catalyst; Inert atmosphere; | 100% |
| Wilkinson's catalyst In dichloromethane 25°C, 10 min; | 99% |
| With chlorocarbonylbis(triphenylphosphine)rhodium(I) In dichloromethane at 0 - 20℃; for 1.83333h; Inert atmosphere; | 99% |
-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
98-83-9
isopropenylbenzene

-
-
280559-30-0
2-(2-phenyl-2-methylethyl)-4,4,5,5-tetramethyl-[1,3,2]-dioxaborolane

| Conditions | Yield |
|---|---|
| With sodium triethylborohydride at 23℃; Catalytic behavior; Inert atmosphere; | 100% |
| With C39H48FeN2Si In benzene-d6 at 60℃; for 3h; Reagent/catalyst; Glovebox; Inert atmosphere; Schlenk technique; | 98% |
| With bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; 1,4-di(diphenylphosphino)-butane In tetrahydrofuran at 20 - 40℃; for 16h; Inert atmosphere; | 96% |

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With triethylamine; bis-triphenylphosphine-palladium(II) chloride In 1,2-dichloro-ethane for 18h; Heating; | 100% |
-
-
108-86-1
bromobenzene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
68716-49-4
p-bromophenylboronic acid pinacol ester

| Conditions | Yield |
|---|---|
| [Ir(COD)(1,3-dicyclohexylimidazolidin-2-ylidene)2]CF3CO2 at 40℃; for 10h; | 100% |
| (η4-1,5-cyclooctadiene)bis(1,3-dimethylimidazolin-2-ylidene)iridium(I) trifluoracetate In further solvent(s) byproducts: H2; (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(C3H2N2Me2)2CF3CO2 in bromobenzene; stirring and heating at 40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
| (η4-1,5-cyclooctadiene)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)iridium(I) trifluoroacetate In further solvent(s) (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)2CF3CO2 in bromobenzene; stirring and heating at 40°C for 12h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
-
-
591-50-4
iodobenzene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
73852-88-7
2-(4-iodophenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| [Ir(COD)(1,3-dicyclohexylimidazolidin-2-ylidene)2]CF3CO2 at 40℃; for 10h; | 100% |
| (η4-1,5-cyclooctadiene)bis(1,3-dimethylimidazolin-2-ylidene)iridium(I) trifluoracetate In further solvent(s) byproducts: H2; (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(C3H2N2Me2)2CF3CO2 in IC6H5; stirring and heating at 40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
| (η4-1,5-cyclooctadiene)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)iridium(I) trifluoroacetate In further solvent(s) (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)2CF3CO2 in iodobenzene; stirring and heating at 40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
-
-
108-90-7
chlorobenzene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
195062-61-4
4-chlorophenylboronic acid pinacol ester

| Conditions | Yield |
|---|---|
| [Ir(COD)(1,3-dicyclohexylimidazolidin-2-ylidene)2]CF3CO2 at 40℃; for 10h; | 100% |
| (η4-1,5-cyclooctadiene)bis(1,3-dimethylimidazolin-2-ylidene)iridium(I) trifluoracetate In chlorobenzene byproducts: H2; (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(C3H2N2Me2)2(CF3CO2) in chlorobenzene; stirring and heating at 40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
| (η4-1,5-cyclooctadiene)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)iridium(I) trifluoroacetate In chlorobenzene (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)2(CF3CO2) in chlorobenzene; stirring and heating at 40°C for12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
-
-
108-88-3
toluene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
195062-57-8
p-tolylboronic pinacol ester

| Conditions | Yield |
|---|---|
| [Ir(COD)(1,3-dicyclohexylimidazolidin-2-ylidene)2]CF3CO2 at 40℃; for 10h; | 100% |
| (η4-1,5-cyclooctadiene)bis(1,3-dimethylimidazolin-2-ylidene)iridium(I) trifluoracetate In toluene byproducts: H2; (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(C3H2N2Me2)2CF3CO2 in toluene; stirring and heating at40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
| (η4-1,5-cyclooctadiene)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)iridium(I) trifluoroacetate In toluene (N2); using Schlenk techniques; dissolving of 2 mmol pinacolborane and 1.5 mol% of Ir(COD)(1,1'-dimethyl-3,3'-o-xylylene-diimidazolin-2,2'-diylidene)2(CF3CO2) in toluene; stirring and heating at 40°C for 12 h; monitoring by GC-MS; removal of solvent under vac. at room temp.; chromy. over silica gel, eluting with CH2Cl2; | 100% |
-
-
353235-94-6
N-(t-butoxycarbonyl)-N-(2-(4-iodophenyl)ethyl)-N-(methylsulfonyl)amine

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
353235-95-7
N-(t-butoxycarbonyl)-N-(methylsulfonyl)-N-(2-(4-(4,4,5,5-tetramethyl(1,3,2-dioxaborolan-2-yl))phenyl)ethyl)amine

| Conditions | Yield |
|---|---|
| With triethylamine; (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride In acetonitrile at 75℃; for 4h; Inert atmosphere; | 100% |
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; triethylamine In dichloromethane; acetonitrile at 70 - 74℃; for 8h; | 81% |
| With triethylamine; 1,1'-bis(diphenylphosphino) ferrocene-dichloropalladium (II)-CH2Cl2 complex In acetonitrile at 70 - 74℃; for 8h; | 60.3% |
-
-
757977-94-9
[4-(1-benzenesulfonyl-3-iodo-1H-pyrrolo[2,3-b]pyridin-5-yl)-phenyl]-dimethyl-amine

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With triethylamine; dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2 In 1,4-dioxane at 120℃; for 0.5h; Microwaves; | 100% |
-
-
153993-80-7
[3-(4-bromophenyl)propoxy](tert-butyl)dimethylsilane

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
936023-12-0
2-{4-[3-(tert-butyl-dimethyl-silanyloxy)-propyl]-phenyl}-4,4,5,5-tetramethyl-[1,3,2]dioxaborolane

| Conditions | Yield |
|---|---|
| With triethylamine; bis-triphenylphosphine-palladium(II) chloride In 1,4-dioxane at 100℃; for 16h; | 100% |
-
-
1015076-51-3
tert-butyl (((2S,3S,5R,6R)-2-ethynyl-5,6-dimethoxy-5,6-dimethyl-1,4-dioxan-2-yl)methoxy)dimethylsilane

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1015076-52-4
tert-butyl (((2S,3S,5R,6R)-5,6-dimethoxy-5,6-dimethyl-3-((E)-2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)vinyl)-1,4-dioxan-2-yl)methoxy)dimethylsilane

| Conditions | Yield |
|---|---|
| at 100℃; for 24h; | 100% |
| at 100 - 110℃; | 100% |
-
-
100-52-7
benzaldehyde

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
95843-98-4
2-(benzyloxy)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With C27H44AlN3 In neat (no solvent) at 20℃; for 0.333333h; Inert atmosphere; Glovebox; | 100% |
| With C28H26FeN2S4 In benzene-d6 at 20℃; for 0.5h; | 99% |
| With sodium hydroxide; 1,3,5-trimethyl-benzene In chloroform at 20℃; for 0.25h; Reagent/catalyst; Solvent; Schlenk technique; Inert atmosphere; | 99% |
-
-
98-86-2
acetophenone

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1143018-72-7
4,4,5,5-tetramethyl-2-(1-phenylethoxy)-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With Zr6(μ3-O)4(μ3-OMgMe)4(3'-nitro-[1,1':4',1'':4'',1'''-tetraphenyl]-4,4'''-dicarboxylate)6 In hexane at 23℃; for 24h; Catalytic behavior; Kinetics; Mechanism; Concentration; Solvent; Time; Inert atmosphere; Glovebox; Schlenk technique; | 100% |
| With 6Zr(4+)*8O(2-)*6C26H15NO6(2-)*4Mg(2+)*4CH3(1-) In hexane at 23℃; for 24h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Glovebox; | 100% |
| With Nd(Cp)3 In tetrahydrofuran at 20℃; for 1h; Reagent/catalyst; Inert atmosphere; | 99% |
-
-
1187734-91-3
[5,10-bis(3,5-di-tert-butylphenyl)-15,20-dibromoporphyrinato]zinc(II)

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1187734-92-4
5,10-bis[3,5-di(tert-butyl)phenyl]-15,20-bis(4,4,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)porphyrinato-N(21),N(22),N(23),N(24)zinc(II)

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; water; triethylamine In 1,2-dichloro-ethane for 1.5h; Suzuki-Miyaura cross-coupling; Inert atmosphere; Reflux; | 100% |
-
-
215313-78-3
[5,15-dibromo-10,20-bis-(3,5-di-tert-butyphenyl)porphyrinato]zinc(II)

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1039027-73-0
5,15-bis[3,5-di(tert-butyl)phenyl]-10,20-bis(4,4,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)porphyrinato-N(21),N(22),N(23),N(24)zinc(II)

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; water; triethylamine In 1,2-dichloro-ethane for 1.5h; Suzuki-Miyaura cross-coupling; Inert atmosphere; Reflux; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; triethylamine In tetrahydrofuran; toluene at 60℃; Inert atmosphere; | 74% |
| With pyridine; triethylamine; bis-triphenylphosphine-palladium(II) chloride In toluene for 1h; Inert atmosphere; Reflux; |
-
-
107-00-6
but-1-yne

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1301680-12-5
2-[(1E)-but-1-en-1-yl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With zirconocene dichloride; lithium triethylborohydride; triethylamine In tetrahydrofuran at -78 - 20℃; Inert atmosphere; | 100% |
| Stage #1: but-1-yne; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane at -78℃; for 0.5h; Inert atmosphere; Stage #2: With bis(pentamethylcyclopentadienyl)zirconium(IV) dichloride; lithium triethylborohydride; triethylamine In tetrahydrofuran at -78 - 20℃; for 20h; | 65% |
| With bis(cyclohexanyl)borane In neat (no solvent) at -78 - 0℃; for 6h; Glovebox; Inert atmosphere; enantioselective reaction; |
-
-
85807-84-7
tert-butyl(5-hexenyloxy)dimethylsilane

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
863208-10-0
tert-butyldimethyl ((6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)hexyl)oxy)silane

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; 1,2-bis-(diphenylphosphino)ethane In dichloromethane at 0 - 20℃; Inert atmosphere; | 100% |
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; 1,2-bis-(diphenylphosphino)ethane In dichloromethane at 20℃; for 6h; | 96% |
-
-
1458-98-6
2-methyl-3-bromo-1-propene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
167773-10-6
4,4,5,5-tetramethyl-2-(2-methylallyl)-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With magnesium In tetrahydrofuran at 25℃; for 2.08333h; Inert atmosphere; | 100% |
| With magnesium In tetrahydrofuran at 25℃; for 2.5h; | 69% |
| With magnesium In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; Schlenk technique; | 65% |
-
-
1357587-50-8
N-[3-(4-bromo-1-isopropyl-3H-pyrazol-3-yl)-2-fluorophenyl]-2,5-difluoro-N-methoxymethylbenzenesulfonamide

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1357587-72-4
2,5-difluoro-N-{2-fluoro-3-[1-isopropyl-4-(4,4,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)-1H-pyrazol-3-yl]phenyl}-N-methoxymethylbenzenesulfonamide

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; triethylamine; bis(acetonitrile)palladium(II) dichloride In toluene at 90℃; for 1h; Microwave irradiation; Inert atmosphere; | 100% |

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| In benzene-d6 at 0℃; for 0.0166667h; Inert atmosphere; | 100% |
-
-
119-61-9
benzophenone

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1373393-16-8
2-[(benzhydryl)oxy]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With C32H45F3N2O4SZn In tetrahydrofuran at 25℃; for 0.0833333h; Catalytic behavior; Reagent/catalyst; Inert atmosphere; | 100% |
| With C37H76N6Si6Th In benzene-d6 at 25℃; for 0.166667h; Catalytic behavior; Glovebox; Sealed tube; Schlenk technique; chemoselective reaction; | 100% |
| With C26H60AlLiN4 In benzene-d6 at 20℃; for 3h; Reagent/catalyst; Inert atmosphere; | 99% |
-
-
1378429-52-7
6-(N-(2-(benzyloxymethyl)allyl)-2-methylpropylsulfonamido)-5-cyclopropyl-2-(4-fluorophenyl)-N-methylbenzofuran-3-carboxamide

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1378429-53-8
6-(N-(3-(benzyloxy)-2-((4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methyl)propyl)-2-methylpropylsulfonamido)-5-cyclopropyl-2-(4-fluorophenyl)-N-methylbenzofuran-3-carboxamide

| Conditions | Yield |
|---|---|
| carbonylbis(triphenylphosphine)rhodium(1)chloride In tetrahydrofuran for 3h; Inert atmosphere; | 100% |
-
-
590-86-3
isovaleraldehyde

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
94845-30-4
2-(isopentyloxy)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With [HC(CMeN(2,6-(isopropyl)2C6H3)2)]Al(hydride)(triflate) In benzene-d6 at 20℃; for 1h; Inert atmosphere; Sealed tube; | 100% |
| With catalyst:C4H9Mg(2,6-iPr2C6H3NCCH3)CH In benzene-d6 pinacolborane reacted with aldehyde in C6D6 at 25°C catalized by nBuMg(2,6-iPrC6H3NCCH3)2CH 0.1-0.5 mol% for <0.5 h; | |
| With 2-H-1,3-di-tert-butyl-2,3-dihydro-1H-1,3,2-diazaphosphole In [D3]acetonitrile at 20℃; for 9h; Catalytic behavior; Inert atmosphere; Sealed tube; | 88 %Spectr. |
-
-
1380090-10-7
tert-butyl (1-(4-bromo-3-methylisoxazol-5-yl)cyclopropyl)carbamate

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1380090-11-8
tert-butyl (1-(3-methyl-4-(4,4,5,5-tetramethyl1,3,2-dioxaborolan-2-yl)isoxazol-5-yl)cyclopropyl)carbamate

| Conditions | Yield |
|---|---|
| With triethylamine; dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; dichloro bis(acetonitrile) palladium(II) In 1,4-dioxane at 100℃; for 1h; Inert atmosphere; | 100% |
-
-
134-84-9
4-Methylbenzophenone

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1416718-99-4
2-(1-(4-methylphenyl)-1-phenylmethoxy)pinacolborane

| Conditions | Yield |
|---|---|
| With C37H76N6Si6Th In benzene-d6 at 25℃; for 0.166667h; Catalytic behavior; Glovebox; Sealed tube; Schlenk technique; chemoselective reaction; | 100% |
| With titanocene bis(catecholborane) In toluene; pentane at 20℃; for 2h; Inert atmosphere; Schlenk technique; Glovebox; | 69% |
| With tris[N,N-bis(trimethylsilyl)amide]lanthanum(III) In benzene-d6 at 25℃; for 0.25h; | > 99 %Spectr. |
| With C48H57HfN3 In benzene-d6 at 25℃; for 0.25h; Reagent/catalyst; Glovebox; | 99 %Spectr. |
-
-
1428881-45-1
7-bromo-3-cyclopropylbenzo[d]isoxazole

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
1428881-46-2
3-cyclopropyl-7-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzo[d]isoxazole

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; dichloro bis(acetonitrile) palladium(II); triethylamine In 1,4-dioxane at 110℃; for 19h; Inert atmosphere; Sealed tube; | 100% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
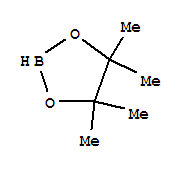
 F,
F, Xi
Xi