-
Name
D-Isoascorbic acid
- EINECS 201-928-0
- CAS No. 89-65-6
- Article Data15
- CAS DataBase
- Density 1.954 g/cm3
- Solubility 0.1 g/mL in water
- Melting Point 169-172 °C (dec.)(lit.)
- Formula C6H8O6
- Boiling Point 552.7 °C at 760 mmHg
- Molecular Weight 176.126
- Flash Point 238.2 °C
- Transport Information
- Appearance Crystalline
- Safety 26-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms D-erythro-Hexonicacid, 3-keto-, g-lactone(6CI);Erythorbic acid (7CI);Araboascorbic acid;Araboascorbic acid, D-;D-(-)-Isoascorbic acid;D-Araboascorbic acid;D-Erythorbic acid;D-arabino-Ascorbic acid;E 315;Erycorbin;Glucosaccharonic acid;Isoascorbic acid;Isovitamin C;Mercate 5;Neo-Cebicure;Saccharosonic acid;Erythorbic acid;
- PSA 107.22000
- LogP -1.40740
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium cyanide; nitrogen; water anschl. mit wss. HCl unter Ausschluss von Luft; |
-
-
21063-40-1
D-arabino-[2]hexulosonic acid methyl ester

-
-
89-65-6
isoascorbic acid

| Conditions | Yield |
|---|---|
| With manganese; water | |
| With water; iron | |
| With water; sodium hydrogencarbonate |

-
-
89-65-6
isoascorbic acid

| Conditions | Yield |
|---|---|
| With ethanol; sulfuric acid anschl. mit Dicyclohexylamin und dann mit wss. NaOH; |

-
-
89-65-6
isoascorbic acid

| Conditions | Yield |
|---|---|
| With methanol; sulfuric acid; water |

-
-
89-65-6
isoascorbic acid

| Conditions | Yield |
|---|---|
| With methanol; sulfuric acid anschl. mit wss. NaHCO3; | |
| With methanol; sulfuric acid anschl. mit wss. Na2CO3; |

| Conditions | Yield |
|---|---|
| at 70℃; |


| Conditions | Yield |
|---|---|
| at 70℃; |


| Conditions | Yield |
|---|---|
| at 60℃; Rate constant; und 70grad; |


| Conditions | Yield |
|---|---|
| at 70℃; |


| Conditions | Yield |
|---|---|
| With ascorbic acid In phosphate buffer at 20℃; for 24h; pH=7.4; Product distribution; Reduction; |
-
-
77-76-9
2,2-dimethoxy-propane

-
-
89-65-6
isoascorbic acid

-
-
50271-42-6
5,6-O-isopropylidene-D-isoascorbic acid

| Conditions | Yield |
|---|---|
| With tin(ll) chloride In acetone for 0.5h; Heating; | 100% |
| With (1S)-10-camphorsulfonic acid In acetone at 20℃; for 1h; | 98% |
| With hydrogenchloride In acetone Ambient temperature; | 90% |
| With tin(ll) chloride In acetone Reflux; |

| Conditions | Yield |
|---|---|
| With Amberlite IR-120 for 2h; Heating; | 100% |
| With acetyl chloride at 40℃; for 4h; | 83% |
| With copper(II) sulfate for 48h; Ambient temperature; |
-
-
933-40-4
cycloxexanone dimethyl ketal

-
-
89-65-6
isoascorbic acid

-
-
97627-84-4
5,6-O-Cyclohexylidene-D-isoascorbic acid

| Conditions | Yield |
|---|---|
| With tin(ll) chloride In ethyl acetate for 0.5h; Heating; | 99% |
-
-
89-65-6
isoascorbic acid

-
-
74352-43-5
6-Bromo-6-deoxy-D-isoascorbic acid

| Conditions | Yield |
|---|---|
| With hydrogen bromide In acetic acid at 22℃; for 24h; | 99% |
| With hydrogen bromide; acetic acid at 20℃; for 16h; Darkness; | 84% |
| Multi-step reaction with 2 steps 1: HBr / 20 °C 2: H2O; HBr; AcOH / 5 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: hydrogen bromide 2: deacetylation View Scheme |
-
-
89-65-6
isoascorbic acid

-
-
15667-21-7
D-erythronolactone

| Conditions | Yield |
|---|---|
| Stage #1: isoascorbic acid With dihydrogen peroxide; sodium carbonate In water at 0℃; Stage #2: In water at 75℃; for 0.5h; Stage #3: With hydrogenchloride In water | 91% |
| With dihydrogen peroxide | |
| With dihydrogen peroxide; sodium carbonate In water at 0 - 40℃; for 2h; |
-
-
95-54-5
1,2-diamino-benzene

-
-
100-63-0
phenylhydrazine

-
-
89-65-6
isoascorbic acid

-
-
68667-54-9
3-(1-phenylhydrazono-D-erythro-2,3,4-trihydroxybut-1-yl)quinoxalin-2-one

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol; water for 0.0666667h; microwave irradiation; | 86% |

-
-
89-65-6
isoascorbic acid

-
-
26301-79-1
D-mannono-1,4-lactone

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In methanol; water under 2585.74 Torr; | 82% |
| With hydrogen; palladium on activated charcoal In water at 50℃; under 2585.7 Torr; for 6h; | 71% |
| With hydrogen; palladium on activated charcoal In water under 3102.9 Torr; for 288h; Ambient temperature; | 51% |
-
-
25636-49-1
3,3-dimethoxypentane

-
-
89-65-6
isoascorbic acid

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In N,N-dimethyl-formamide at 20℃; for 24h; Inert atmosphere; | 79% |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase In acetonitrile at 50℃; Enzymatic reaction; | 78.5% |
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| Stage #1: 1,1-dimethoxy-1-(4-bromophenyl)methane; isoascorbic acid With trifluoroacetic acid In N,N-dimethyl-formamide at 20℃; for 120h; Inert atmosphere; Stage #2: With dihydrogen peroxide; potassium carbonate In water at 0 - 20℃; for 16h; Stage #3: ethyl iodide In acetonitrile for 20h; Reflux; | 75% |


| Conditions | Yield |
|---|---|
| Stage #1: isoascorbic acid With dihydrogen peroxide; sodium carbonate In water at 0 - 75℃; for 1h; Inert atmosphere; Stage #2: benzoyl chloride With pyridine at 0 - 20℃; for 12h; Inert atmosphere; | 69% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; sodium carbonate 1) 17 deg C, 5 min; 2) 40 deg C, 30 min; | A n/a B 67.5% |

| Conditions | Yield |
|---|---|
| In water Ambient temperature; | 65% |

-
-
89-65-6
isoascorbic acid

-
-
7152-74-1
(-)-(3R-cis)-mono(tetrahydro-4-hydroxy-2-oxo-3-furanyl)ester 2-(4-methyl-phenyl)hidrazide of ethanedioic acid

| Conditions | Yield |
|---|---|
| at 0℃; | 55% |

-
-
89-65-6
isoascorbic acid

| Conditions | Yield |
|---|---|
| With immobilized lipase from Candida Antarctica In acetone at 45℃; for 16h; Molecular sieve; | 49% |
-
-
1334953-93-3
6-O-[(+)-L-ascorbic acid]-vinyl dodecanedioate

-
-
89-65-6
isoascorbic acid

| Conditions | Yield |
|---|---|
| With immobilized lipase from Candida Antarctica In acetone at 45℃; for 16h; Molecular sieve; | 47% |

| Conditions | Yield |
|---|---|
| In water Ambient temperature; | 39% |
-
-
1576-86-9, 1576-87-0, 764-39-6
2-pentenal

-
-
89-65-6
isoascorbic acid

| Conditions | Yield |
|---|---|
| In water Ambient temperature; | 20% |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
89-65-6
isoascorbic acid

-
-
13443-57-7, 139164-18-4
D-erythro-2,4,5,6-tetrahydro-3-methoxy-hex-2c-enoic acid-4-lactone

| Conditions | Yield |
|---|---|
| With methanol; diethyl ether at -10℃; |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
89-65-6
isoascorbic acid

-
-
40613-68-1
D-erythro-4,5,6-trihydroxy-2,3-dimethoxy-hex-2c-enoic acid-4-lactone

| Conditions | Yield |
|---|---|
| With methanol; diethyl ether |

| Conditions | Yield |
|---|---|
| With sulfuric acid | |
| With Novozym 435 In neat (no solvent) for 72h; Inert atmosphere; Enzymatic reaction; |
-
-
57-10-3
1-hexadecylcarboxylic acid

-
-
89-65-6
isoascorbic acid

-
-
47635-02-9
6-O-palmitoyl-L-ascorbic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
57-11-4
stearic acid

-
-
89-65-6
isoascorbic acid

-
-
71623-59-1
D-erythro-2,3,4,5-tetrahydroxy-6-stearoyloxy-hex-2c-enoic acid-4-lactone

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
119-26-6
(2,4-dinitro-phenyl)-hydrazine

-
-
89-65-6
isoascorbic acid

-
-
35923-30-9
D-erythro-2,3-bis-(2,4-dinitro-phenylhydrazono)-4,5,6-trihydroxy-hexanoic acid-4-lactone

| Conditions | Yield |
|---|---|
| With hydrogenchloride; iodine |

| Conditions | Yield |
|---|---|
| With H(1+)*Fe(CN)6(3-)=HFe(CN)6(2-) In water at 20℃; Rate constant; Mechanism; further oxidants: Fe(3+)aq., Fe(OH)(2+)aq.; | |
| With ammonium molybdate; water; dihydrogen peroxide | |
| With p-benzoquinone In ethanol for 90h; Ambient temperature; | |
| With iodine In ethanol; water |
-
-
67-56-1
methanol

-
-
89-65-6
isoascorbic acid

-
A
-
75795-84-5, 75829-07-1, 78512-72-8, 116499-89-9
(3S,3aS,6R,6aR)-3,3a,6-Trihydroxy-3-methoxy-tetrahydro-furo[3,2-b]furan-2-one

-
B
-
75795-84-5, 75829-07-1, 78512-72-8, 116499-89-9
(3R,3aS,6R,6aR)-3,3a,6-Trihydroxy-3-methoxy-tetrahydro-furo[3,2-b]furan-2-one

| Conditions | Yield |
|---|---|
| With p-benzoquinone 1.) DMF, room temp.; Multistep reaction; |


| Conditions | Yield |
|---|---|
| In dimethylsulfoxide-d6 for 2h; Product distribution; other time and solvent; | A 48 % Spectr. B 31 % Spectr. C 21 % Spectr. |
| In perdeuteriopyridine for 0.05h; | A 45 % Spectr. B 45 % Spectr. C 10 % Spectr. |

D-Isoascorbic acid Chemical Properties
Molecular Structure of D-Isoascorbic acid (CAS NO.89-65-6):
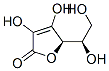
IUPAC Name: 2-(1,2-dihydroxyethyl)-4,5-dihydroxyfuran-3-one
Empirical Formula: C6H8O6
Molecular Weight: 176.1241
EINECS: 201-928-0
Melting point: 169-172 °C (dec.)(lit.)
Boiling Point: 552.7 °C at 760 mmHg
Flash Point: 238.2 °C
Index of Refraction: 1.711
Molar Refractivity: 35.26 cm3
Molar Volume: 90.1 cm3
Polarizability: 13.97 10 -24 cm3
Surface Tension: 140.5 dyne/cm
Density: 1.954 g/cm3
Enthalpy of Vaporization: 95.78 kJ/mol
Vapour Pressure: 1.62E-14 mmHg at 25°C
alpha: -17.25 o (c=10, H2O 25 oC)
Storage temp: Store at 0-5°C
Water Solubility: 1g/10mL
Appearance: Crystalline
Merck: 14,5126
Stability: Stable. Combustible. Incompatible with chemically active metals, aluminium, zinc, copper, magnesium, strong bases, strong oxidizing agents.
Product Categories: Food and Feed Additive; Sugar Acids; Vitamin Derivatives; Biochemistry;Sugars; Vitamins; Water Ttreatment Chemicals
D-Isoascorbic acid Toxicity Data With Reference
| 1. | mma-sat 50 mg/plate | FCTOD7 Food and Chemical Toxicology. 22 (1984),623. | ||
| 2. | dnd-uns:lyms 5 mmol/L | JNSVA5 Journal of Nutritional Science and Vitaminology. 21 (1975),237. |
D-Isoascorbic acid Consensus Reports
Reported in EPA TSCA Inventory.
D-Isoascorbic acid Safety Profile
Mutation data reported. When heated to decomposition it emits acrid smoke and irritating vapors.
Hazard Codes of D-Isoascorbic acid (CAS NO.89-65-6): Xi
Xi
Risk Statements:
36/37/38: Irritating to eyes, respiratory system and skin
Safety Statements:
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
24/25: Avoid contact with skin and eyes
WGK Germany: 2
RTECS: KF3015000
D-Isoascorbic acid Specification
D-Isoascorbic acid , with CAS number of 89-65-6, can be called D-erythro-Hexonicacid, 3-keto-, g-lactone(6CI) ; Araboascorbic acid ; Araboascorbic acid, D- ; D-(-)-Isoascorbic acid ; D-Araboascorbic acid ; D-Erythorbic acid ; Glucosaccharonic acid ; Saccharosonic acid .
Related Products
- D-Isoascorbic acid
- 89659-66-5
- 89660-14-0
- 89660-20-8
- 896658-52-9
- 896720-20-0
- 896722-53-5
- 89672-77-5
- 896731-82-1
- 896740-06-0
- 89677-46-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View