-
Name
Dihydrotachysterol
- EINECS 200-672-7
- CAS No. 67-96-9
- Article Data16
- CAS DataBase
- Density 1.003 g/cm3
- Solubility
- Melting Point 116-120°C
- Formula C28H46O
- Boiling Point 499.5 °C at 760 mmHg
- Molecular Weight 398.673
- Flash Point 217.6 °C
- Transport Information
- Appearance
- Safety 36
- Risk Codes 22
-
Molecular Structure
-
Hazard Symbols

- Synonyms 9,10-Secoergosta-5,7,22-trien-3-ol,(3b,5E,7E,10a,22E)- (9CI);Tachysterol,dihydro- (7CI,8CI);Tachysterol2, dihydro- (6CI);24-Methyl-9,10-secocholesta-5,7,22-trien-3b-ol;9,10-Secoergosta-5,7,22-trien-3b-ol;A.T. 10;A.T. 10 (steroid);Anti-tetany substance 10;Antitanil;Calcamine;DHT2;Dichystrolum;Dihydral;
- PSA 20.23000
- LogP 7.72090
Synthetic route
-
-
51744-66-2
3β-hydroxy-9,10-secoergosta-5(E),7(E),10(19),22(E)-tetraene

-
A
-
65377-91-5
(3S,5E,7E,10R)-9,10-seco-ergosta-5,7,22t-trien-3-ol

-
B
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| With bis(cyclopentadienyl)titanium dichloride; lithium aluminium tetrahydride In tetrahydrofuran for 2.5h; | A 6.5% B 86% |
| With bis(cyclopentadienyl)titanium dichloride In tetrahydrofuran; toluene for 1.5h; Ambient temperature; | A 38% B 57% |
-
-
142980-94-7, 143060-77-9
dihydrotachysterol2 tert-butyldimethylsilyl ether

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran for 1h; Ambient temperature; | 86% |

| Conditions | Yield |
|---|---|
| With sodium; butan-1-ol | |
| With pentan-1-ol; sodium | |
| With sodium; glycerol; xylene | |
| With sodium; ethylene glycol; xylene | |
| Multi-step reaction with 2 steps 1: 58 percent / iodine / diethyl ether / 2.5 h 2: 86 percent / LiAlH4, Cp2TiCl2 / tetrahydrofuran / 2.5 h View Scheme |
-
-
67-96-9, 807-27-2, 7665-90-9, 34611-22-8, 62777-58-6, 65377-86-8, 65377-91-5, 116559-85-4, 116559-86-5, 143060-73-5
(3aR)-3ar-Methyl-3c-((1R:4R)-1.4.5-trimethyl-hexen-(2t)-yl)-7-[2-((2S)-5t-hydroxy-2r-methyl-cyclohexyliden-(seqcis))-aethyliden-(seqtrans)]-(7atH)-hexahydro-indan

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| ueber mehrere Stufen; |

| Conditions | Yield |
|---|---|
| With ammonia; lithium | |
| With sodium; diethylamine | |
| With sodium; N-methylaniline |

| Conditions | Yield |
|---|---|
| With ethanol; sodium | |
| With ethanol; sodium Isolierung als O-Allophanoyl-Derivat; |
-
-
104396-98-7
toxisterol2 R

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| ueber mehrere Stufen; |
-
-
56-23-5
tetrachloromethane

-
-
50-14-6
Calciferol

-
-
107-21-1
ethylene glycol

-
A
-
67-96-9
Dihydrotachysterol2

-
B
-
67-96-9, 807-27-2, 7665-90-9, 34611-22-8, 62777-58-6, 65377-86-8, 65377-91-5, 116559-85-4, 116559-86-5, 143060-73-5
(3aR)-3ar-Methyl-3c-((1R:4R)-1.4.5-trimethyl-hexen-(2t)-yl)-7-[2-((2S)-5t-hydroxy-2r-methyl-cyclohexyliden-(seqcis))-aethyliden-(seqtrans)]-(7atH)-hexahydro-indan

-
C
-
104396-98-7
toxisterol2 R

-
-
56-23-5
tetrachloromethane

-
-
50-14-6
Calciferol

-
-
56-81-5
glycerol

-
A
-
67-96-9
Dihydrotachysterol2

-
B
-
67-96-9, 807-27-2, 7665-90-9, 34611-22-8, 62777-58-6, 65377-86-8, 65377-91-5, 116559-85-4, 116559-86-5, 143060-73-5
(3aR)-3ar-Methyl-3c-((1R:4R)-1.4.5-trimethyl-hexen-(2t)-yl)-7-[2-((2S)-5t-hydroxy-2r-methyl-cyclohexyliden-(seqcis))-aethyliden-(seqtrans)]-(7atH)-hexahydro-indan

-
C
-
104396-98-7
toxisterol2 R

-
-
50-14-6
Calciferol

-
-
71-36-3
butan-1-ol

-
A
-
67-96-9
Dihydrotachysterol2

-
B
-
67-96-9, 807-27-2, 7665-90-9, 34611-22-8, 62777-58-6, 65377-86-8, 65377-91-5, 116559-85-4, 116559-86-5, 143060-73-5
(3aR)-3ar-Methyl-3c-((1R:4R)-1.4.5-trimethyl-hexen-(2t)-yl)-7-[2-((2S)-5t-hydroxy-2r-methyl-cyclohexyliden-(seqcis))-aethyliden-(seqtrans)]-(7atH)-hexahydro-indan

-
C
-
104396-98-7
toxisterol2 R


-
A
-
65377-91-5
(3S,5E,7E,10R)-9,10-seco-ergosta-5,7,22t-trien-3-ol

-
B
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| With Wilkinson's catalyst; benzene Hydrogenation; |

-
A
-
67-96-9
Dihydrotachysterol2

-
B
-
104396-98-7
toxisterol2 R

| Conditions | Yield |
|---|---|
| With propan-1-ol; sodium | |
| With propan-1-ol; sodium Isolierung als O-Allophanoyl-Derivat; |


-
-
142980-92-5
(2E)-2-<(2'S,5'S)-5'-<(tert-butyldimethylsilyl)oxy>-2'-methylcyclohexylidene>ethanol

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 2: 85 percent / n-butyl lithium / hexane; tetrahydrofuran / a) -78 deg C, 30 min, b) -70 deg C, 2 h, c) to r.t., 2 h 3: 86 percent / tetra-n-butylammonium fluoride / tetrahydrofuran / 1 h / Ambient temperature View Scheme |
-
-
142980-91-4
(2E)-ethyl <(2'S,5'S)-5'-<(tert-butyldimethylsilyl)oxy>-2'-methylcyclohexylidene>ethanoate

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 99 percent / diisobutylaluminium hydride / toluene / 1 h / -78 °C 3: 85 percent / n-butyl lithium / hexane; tetrahydrofuran / a) -78 deg C, 30 min, b) -70 deg C, 2 h, c) to r.t., 2 h 4: 86 percent / tetra-n-butylammonium fluoride / tetrahydrofuran / 1 h / Ambient temperature View Scheme |
-
-
141277-81-8
(2E)-<2-<(2'S,5'S)-5'-<(tert-butyldimethylsilyl)oxy>-2'-methylcyclohexylidene>ethyl>diphenylphosphine oxide

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 85 percent / n-butyl lithium / hexane; tetrahydrofuran / a) -78 deg C, 30 min, b) -70 deg C, 2 h, c) to r.t., 2 h 2: 86 percent / tetra-n-butylammonium fluoride / tetrahydrofuran / 1 h / Ambient temperature View Scheme |
-
-
142980-89-0
ethyl <(1'R,2'S,5'S)-1',5-'dihydroxy-2'-methylcyclohexyl>ethanoate

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 90 percent / imidazole / dimethylformamide / 24 h 2: 93 percent / bis<2,2,2-trifluoro-1-phenyl-1-(trifluoromethyl)ethoxy>diphenylsulfurane / CCl4 / 70 h / Ambient temperature 3: 99 percent / diisobutylaluminium hydride / toluene / 1 h / -78 °C 5: 85 percent / n-butyl lithium / hexane; tetrahydrofuran / a) -78 deg C, 30 min, b) -70 deg C, 2 h, c) to r.t., 2 h 6: 86 percent / tetra-n-butylammonium fluoride / tetrahydrofuran / 1 h / Ambient temperature View Scheme |
-
-
142980-87-8, 143060-75-7
ethyl <(1'R,2'S,5'S)-6-hydroperoxy-2',6-'dimethylbicyclo<3.2.1>-7'-oxaoctyl>ethanoate

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: 85 percent / pyridine, DMAP / CH2Cl2 / a) 0 deg C, 1 h, b) r.t., 2 h, c) reflux, 12 h 2: 87 percent / 0.2 M NaOEt / ethanol / 3 h / 0 °C 3: 90 percent / imidazole / dimethylformamide / 24 h 4: 93 percent / bis<2,2,2-trifluoro-1-phenyl-1-(trifluoromethyl)ethoxy>diphenylsulfurane / CCl4 / 70 h / Ambient temperature 5: 99 percent / diisobutylaluminium hydride / toluene / 1 h / -78 °C 7: 85 percent / n-butyl lithium / hexane; tetrahydrofuran / a) -78 deg C, 30 min, b) -70 deg C, 2 h, c) to r.t., 2 h 8: 86 percent / tetra-n-butylammonium fluoride / tetrahydrofuran / 1 h / Ambient temperature View Scheme |
-
-
142980-88-9
ethyl <(1'R,2'S,5'S)-5'-acetoxy-1'-hydroxy-2'-methylcyclohexyl>ethanoate

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 87 percent / 0.2 M NaOEt / ethanol / 3 h / 0 °C 2: 90 percent / imidazole / dimethylformamide / 24 h 3: 93 percent / bis<2,2,2-trifluoro-1-phenyl-1-(trifluoromethyl)ethoxy>diphenylsulfurane / CCl4 / 70 h / Ambient temperature 4: 99 percent / diisobutylaluminium hydride / toluene / 1 h / -78 °C 6: 85 percent / n-butyl lithium / hexane; tetrahydrofuran / a) -78 deg C, 30 min, b) -70 deg C, 2 h, c) to r.t., 2 h 7: 86 percent / tetra-n-butylammonium fluoride / tetrahydrofuran / 1 h / Ambient temperature View Scheme |
-
-
142980-90-3
ethyl <(1'R,2'S,5'S)-5'-<(tert-butyldimethylsilyl)oxy>-1'-hydroxy-2'-methylcyclohexyl>ethanoate

-
-
67-96-9
Dihydrotachysterol2

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 93 percent / bis<2,2,2-trifluoro-1-phenyl-1-(trifluoromethyl)ethoxy>diphenylsulfurane / CCl4 / 70 h / Ambient temperature 2: 99 percent / diisobutylaluminium hydride / toluene / 1 h / -78 °C 4: 85 percent / n-butyl lithium / hexane; tetrahydrofuran / a) -78 deg C, 30 min, b) -70 deg C, 2 h, c) to r.t., 2 h 5: 86 percent / tetra-n-butylammonium fluoride / tetrahydrofuran / 1 h / Ambient temperature View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Irradiation.UV-Licht 2: sodium; N-methyl-aniline View Scheme | |
| Multi-step reaction with 2 steps 1: benzene / Irradiation.mit UV-Licht (Magnesium-Funken); Isolierung ueber das O-<3.5-Dinitro-4-methyl-benzoyl>-Derivat oder ueber das O-Acetyl-Derivat des Citraconsaeure-anhydrid-Addukts 2: sodium; ethanol / Isolierung als O-Allophanoyl-Derivat View Scheme |
-
-
110-86-1
pyridine

-
-
67-96-9
Dihydrotachysterol2

-
-
108-24-7
acetic anhydride

-
-
71045-54-0
(3S,5E,7E,10S)-3-acetoxy-9,10-seco-ergosta-5,7,22t-triene


| Conditions | Yield |
|---|---|
| With aluminum isopropoxide; toluene Behandeln des Reaktionsprodukts mit Semicarbazid-hydrochlorid und Kaliumacetat in wss. Methanol; |
-
-
67-96-9
Dihydrotachysterol2

-
-
55812-80-1, 55870-05-8, 70144-73-9
(1R,3aR,7aR)-7a-Methyl-1-((E)-(1R,4R)-1,4,5-trimethyl-hex-2-enyl)-octahydro-inden-4-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: CrO3; acetic acid View Scheme |
-
-
67-96-9
Dihydrotachysterol2

-
-
2234-73-3
2,3,3a,4,5,6,7,7aβ-octahydro-7aα-methyl-1R-(1α,1R,4R,5-trimethyl-2E-hexenyl)-4H-inden-4-one semicarbazide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 2: CrO3; acetic acid View Scheme |
Dihydrotachysterol Chemical Properties
Product Name: Dihydrotachysterol (CAS NO.67-96-9)
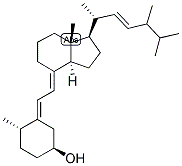
Molecular Formula: C28H46O
Molecular Weight: 398.66g/mol
Mol File: 67-96-9.mol
Einecs: 200-672-7
Boiling point: 499.5 °C at 760 mmHg
Storage Temperature: 2-8°C
Flash Point: 217.6 °C
Density: 1.003 g/cm3
Surface Tension: 43 dyne/cm
Enthalpy of Vaporization: 88.44 kJ/mol
Vapour Pressure: 4.52E-12 mmHg at 25°C
XLogP3-AA: 7.7
H-Bond Donor: 1
H-Bond Acceptor: 1
Structure Descriptors of Dihydrotachysterol (CAS NO.67-96-9):
IUPAC Name: (1S,2E,3S)-2-[(2E)-2-[(7aR)-1-[(E,2R,5R)-5,6-dimethylhept-3-en-2-yl]-7a-methyl-2,3,3a,5,6,7-hexahydro-1H-inden-4-ylidene]ethylidene]-3-methylcyclohexan-1-ol
Canonical SMILES: CC1CCCC(C1=CC=C2CCCC3(C2CCC3C(C)C=CC(C)C(C)C)C)O
Isomeric SMILES: C[C@H]\1CCC[C@@H](/C1=C/C=C/2\CCC[C@]3(C2CCC3[C@H](C)/C=C/[C@H](C)C(C)C)
C)O
InChI: InChI=1S/C28H46O/c1-19(2)20(3)12-13-22(5)25-16-17-26-23(10-8-18-28(25,26)6)14-15-24-21(4)9-7-11-27(24)29/h12-15,19-22,25-27,29H,7-11,16-18H2,1-6H3/b13-12+,23-14+,24-15+/t20-,21-,22+,25?,26?,27-,28+/m0/s1
InChIKey: DTSXXSAWQHPLEF-GFVAUXBKSA-NUHFFFAOYSA-N
Dihydrotachysterol Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 104mg/kg (104mg/kg) | Drugs in Japan Vol. 6, Pg. 330, 1982. | |
| mouse | LD50 | oral | 288mg/kg (288mg/kg) | Drugs in Japan Vol. 6, Pg. 330, 1982. |
Dihydrotachysterol Safety Profile
Poison by ingestion and intraperitoneal routes. Experimental reproductive effects. When heated to decomposition it emits acrid smoke and irritating fumes.
Safety Information of Dihydrotachysterol (CAS NO.67-96-9):
Hazard Codes: Xn![]()
Risk Statements: 22
22: Harmful if swallowed
Safety Statements: 36
36: Wear suitable protective clothing
Dihydrotachysterol Specification
Dihydrotachysterol ,its CAS NO. is 67-96-9,the synonyms is 9,10-Secoergosta-5,7,22-trien-3beta-ol ; 9,10-Secoergosta-5,7,22-trien-3b-ol ; Dichystrolum ; Dihydrotachysterol ; Calcamine ; At-10 ; Antitanil ; 7,22-Trien-3-ol,(3-beta,5e,7e,10-alpha,22e)-10-secoergosta-5 .
Related Products
- Dihydrotachysterol
- 679-69-6
- 67969-69-1
- 67-97-0
- 67970-17-6
- 67970-19-8
- 67970-22-3
- 67973-19-7
- 67973-20-0
- 67973-32-4
- 67973-34-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View