-
Name
DIMETHYLCADMIUM
- EINECS 208-055-4
- CAS No. 506-82-1
- Article Data32
- CAS DataBase
- Density 1.986 g/cm3
- Solubility
- Melting Point -4.5 °C
- Formula C2H6Cd
- Boiling Point 105.5 °C
- Molecular Weight 142.48
- Flash Point -18 °C
- Transport Information UN 2845
- Appearance colorless toxic liquid
- Safety 7/8-16-23-36/37/39-45
- Risk Codes 17-20/21/22
-
Molecular Structure
- Hazard Symbols
- Synonyms (CH3)2Cd;DIMETHYLCADMIUM;CADMIUM DIMETHYL;Dimethylcadmiumelecgrcolorlessliq;Dimethylcadmiuminhexane;CADMIUM DIMETHYL 95%;Dimethylcadmium,elec.gr.(99.995+%-Cd)PURATREM;Dimethylcadmium,min.97%
- PSA 0.00000
- LogP 1.16510
Synthetic route

-
A
-
754-25-6
trimethyl(trifluoromethyl)tin

-
B
-
506-82-1
dimethylcadmium

-
C
-
65059-36-1
dimethylbis(trifluoromethyl)tin

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: Sn(CH3)4, C2F4, CH3I; excess of Cd compd. quickly added to SnI4 in CH2Cl2, sepd. after 15 min; solvent removed; | A 23% B n/a C 10% |


| Conditions | Yield |
|---|---|
| With cadmium(II) chloride | |
| With diethyl ether; cadmium(II) bromide | |
| With diethyl ether; cadmium(II) iodide at 25℃; |

| Conditions | Yield |
|---|---|
| With diethyl ether; cadmium(II) bromide | |
| With diethyl ether; cadmium(II) chloride |

| Conditions | Yield |
|---|---|
| With cadmium at 110℃; |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether room temp.; distn.; | >90 |

| Conditions | Yield |
|---|---|
| In diethyl ether under N2 atm. Et2O soln. Grignard reagent was added dropwise for 2 h to stirred suspn. CdCl2 in Et2O in protected from light vessel and stirred for 12 h; Et2O was removed at reduced pressure; |

-
-
506-82-1
dimethylcadmium

| Conditions | Yield |
|---|---|
| byproducts: diethyl ether; heated; vacuum distilled, held at 2°C, distilled into two traps (-30°C, -196°C); |

-
-
506-82-1
dimethylcadmium

| Conditions | Yield |
|---|---|
| decompn.; |

| Conditions | Yield |
|---|---|
| at 28.69℃; Temperature; |

| Conditions | Yield |
|---|---|
| In (2)H8-toluene at 26.84℃; Equilibrium constant; Solvent; Inert atmosphere; Schlenk technique; Glovebox; |

-
-
506-82-1
dimethylcadmium

-
-
65094-19-1
trifluoromethyltribromostannane

-
-
754-25-6
trimethyl(trifluoromethyl)tin

| Conditions | Yield |
|---|---|
| tenfold excess of Cd(CH3)2, 20°C (15 min); | 100% |
| tenfold excess of Cd(CH3)2, 20°C (15 min); | 100% |
| small excess of Cd(CH3)2, 20°C (15 min); | 92% |
| small excess of Cd(CH3)2, 20°C (15 min); | 92% |

| Conditions | Yield |
|---|---|
| twofold excess of Cd(CH3)2; 20°C, 24 h; | 100% |
-
-
10556-98-6
1,3,5-Triisopropyl-1,3,5-triazacyclohexane

-
-
506-82-1
dimethylcadmium

| Conditions | Yield |
|---|---|
| In neat (no solvent) (N2 free of O2) amine slowly added to dimethylcadmium at -196°C, warmed to room temp.; distd. (140°C, E-2 Torr); elem. anal.; | 99.2% |

| Conditions | Yield |
|---|---|
| In diethyl ether (N2); stirring (overnight), reflux (1 h); solvent removal (vac.), washing (Et2O), drying; elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) (N2 free of O2) amine slowly added to dimethylcadmium at -196°C, warmed to room temp.; sublimed (120°C, E-2 Torr); elem. anal.; | 98.9% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) (N2 free of O2) amine slowly added to dimethylcadmium at -196°C, warmed to room temp.; distd. (140°C, E-2 Torr); elem. anal.; | 98.4% |

| Conditions | Yield |
|---|---|
| In toluene byproducts: EtBMe2, CH4; Ar-atmosphere; dropwise addn. of 1 equiv. Et2BOH to CdMe2 (-20°C,stirring), stirring at room temp. for 30 min; removal of volatiles (reduced pressure); | 98% |

| Conditions | Yield |
|---|---|
| In benzene | 96.3% |

| Conditions | Yield |
|---|---|
| In toluene at 20℃; for 0.5h; | 96% |

| Conditions | Yield |
|---|---|
| In hexane hexane soln. of Me2Cd added to hexane; then 1-amino-4-methylpiperazine added; mixt. stirred; elem. anal.; | 94% |
-
-
506-82-1
dimethylcadmium

-
-
754-36-9
perfluormethylgermanium iodide

-
-
66348-20-7
Dimethyl-bis-trifluoromethyl-germane

| Conditions | Yield |
|---|---|
| twofold excess of Cd(CH3)2; 20°C, 24 h; | 92% |

| Conditions | Yield |
|---|---|
| In toluene Schlenk technique; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: Si(CH3)4; stirred at 25°C for 4 h; filtered, powder annealed in vacuo (400°C, 4 h); X-ray powder diffraction; | 91% |
| In toluene byproducts: Si(CH3)4; stirred at 25°C, reaction is fairly slow, taked days to go compled; proton NMR; | |
| In further solvent(s) under argon, soln. of Me2Cd in (n-C8H17)3P and soln. of ((CH3)3Si)2Se in (n-C8H17)3P are combined and added under vigorous stirring to (n-C8H17)3PO at 300°C, heat removed (180°C) then gradually raised to 230-260°C; cooled to 60°C, methanol added, centrifugated, dispersed (1-butanol), centrifugated, precipitate discarded, methanol added to the supernatant, flocculate rinsed (methanol), dried (vacuum); |
-
-
506-82-1
dimethylcadmium

-
-
65094-20-4
bis(trifluoromethyl)dibromostannane

-
A
-
754-25-6
trimethyl(trifluoromethyl)tin

-
B
-
65059-36-1
dimethylbis(trifluoromethyl)tin

| Conditions | Yield |
|---|---|
| educts separately condensed into a reactor held at -196°C, then allowed to react for 35 min at room temp.; volatile products were removed and analyzed, identification by NMR; | A n/a B 91% |

-
-
506-82-1
dimethylcadmium

-
-
66348-18-3
tris(trifluoromethyl)germyl iodide

-
-
66348-19-4
Methyl-tris-trifluoromethyl-germane

| Conditions | Yield |
|---|---|
| twofold excess of Cd(CH3)2; 20°C, 24 h; | 90% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) V-compd. added to Cd-compd. at room temp., immediate reaction; extd. (pentane); | A 90% B n/a |


| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: CdCl2; components (5% Cd-compd. excess) vac. distn. into Pyrex tube with Teflonstopcock, mixt. allowing to warm to room temp.; Ge-compd. pumping off the solid byproduct; FTIR spectroscopy; | 90% |

| Conditions | Yield |
|---|---|
| In toluene Schlenk technique; Inert atmosphere; | 90% |
-
-
506-82-1
dimethylcadmium

-
-
130-15-4
[1,4]naphthoquinone

-
-
42860-82-2
4-hydroxy-4-methyl-1(4H)-naphthalenone

| Conditions | Yield |
|---|---|
| In tetrahydrofuran 1.) -10 deg C, 1 h, 2.) -10 deg C -> room temperature; | 89% |

| Conditions | Yield |
|---|---|
| With CS2 In toluene byproducts: CH4; (N2); dimethylcadmium and diethylamine stirred and heated at 70°C for 12 h; cooled; added CS2; warmed to 40°C for 5 h; filtered; evapd.; recrystd. from benzene; elem. anal.; | 89% |

-
-
506-82-1
dimethylcadmium

| Conditions | Yield |
|---|---|
| In diethyl ether | 89% |

| Conditions | Yield |
|---|---|
| In toluene Schlenk technique; Inert atmosphere; | 89% |

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: Si(i-Pr)Me3; stirred at 25°C for 15 min; filtered, annealed (400°C, 2 h); | 88% |
-
-
862277-33-6
1,3-bis(dimethylamino)-2-(dimethylaminomethyl)-propan-2-ol

-
-
544-97-8
dimethyl zinc(II)

-
-
506-82-1
dimethylcadmium

| Conditions | Yield |
|---|---|
| In hexane (inert atm.); addn. of aminoalcohol deriv. to hexane soln. of cadmium compd. and zinc compd. at -78°C, stirring for 1 h, warming to room temp.; heating to 60°C, cooling to room temp., elem. anal.; | 87% |

-
-
506-82-1
dimethylcadmium

-
-
81603-45-4
8-Methyl-1,2,3,4-tetrahydro-naphthalene-1-carbonyl chloride

-
-
81603-31-8
8-methyl-1,2,3,4-tetrahydro-1-naphthyl methyl ketone

| Conditions | Yield |
|---|---|
| In benzene for 1h; Heating; | 85% |
-
-
506-82-1
dimethylcadmium

-
-
289618-59-3
2-((2,6-diisopropylphenyl)amino)-4-((2,6-diisopropylphenyl)imino)-2-pentene

-
-
1234205-23-2
(CH3)2C2NCHN((CH(CH3)2)2C6H3)2CdCH3

| Conditions | Yield |
|---|---|
| In benzene (under N2, Schlenk); Cd(CH3)2 added to soln. of ligand in benzene, refluxed for 16 h, Cd(CH3)2 added, refluxed for 16 h; volatiles removed in vacuo, extd. into benzene, lyophilized; | 85% |
-
-
41203-22-9
1,4,8,11-tetramethyl-1,4,8,11-tetra-azacyclotetradecane

-
-
2999-74-8
dimethylmagnesium

-
-
506-82-1
dimethylcadmium

-
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran | 83% |

Dimethylcadmium Consensus Reports
Dimethylcadmium Standards and Recommendations
ACGIH TLV: TWA 0.002 mg(Cd)/m3 (respirable dust), Suspected Human Carcinogen); BEI: 5 μg/g creatinine in urine; 5 μg/L in blood
DFG MAK: DFG BAT: Blood 1.5 μg/dL; Urine 15 μg/dL; Suspected Carcinogen
NIOSH REL: (Cadmium) Reduce to lowest feasible level
Dimethylcadmium Specification
The Cadmium, dimethyl- has the CAS registry number 506-82-1. Its EINECS number is 208-055-4. This chemical's molecular formula is C2H6Cd and molecular weight is 142.48. What's more, its systematic name is dimethylcadmium. It is used as compound semiconductors, polymerization catalysts, or in organic synthesis.
Preparation of Cadmium, dimethyl-: this chemical can be prepared by halogenated cadmium and fresh Grignard reagent.
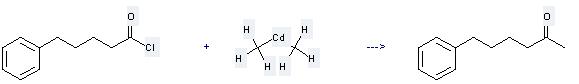
Uses of Cadmium, dimethyl-: it can be used to produce 6-phenyl-hexan-2-one. It will need solvent diethyl ether. The yield is about 73%.
When you are using this chemical, please be cautious about it as the following:
This chemical is spontaneously flammable in air, so you should keep it away from sources of ignition - No smoking. It is harmful by inhalation, in contact with skin and if swallowed. You should keep the container tightly closed and dry. You must not breathe gas/fumes/vapour/spray (appropriate wording to be specified by the manufacturer). When using it, you need wear suitable protective clothing, gloves and eye/face protection. In case of accident or if you feel unwell, you must seek medical advice immediately (show the label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: C[Cd]C
(2)Std. InChI: InChI=1S/2CH3.Cd/h2*1H3;
(3)Std. InChIKey: KVVGSXJGEUULNM-UHFFFAOYSA-N
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View