-
Name
Ethyl hexadecanoate
- EINECS 211-064-6
- CAS No. 628-97-7
- Article Data67
- CAS DataBase
- Density 0.864 g/cm3
- Solubility Immiscible with water
- Melting Point 24-26 °C(lit.)
- Formula C18H36O2
- Boiling Point 342.2 °C at 760 mmHg
- Molecular Weight 284.483
- Flash Point 149 °C
- Transport Information
- Appearance clear colorless oily liquid after melting
- Safety 23-24/25
- Risk Codes
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Palmiticacid, ethyl ester (6CI,8CI);Ethyl hexadecanoate;Ethyl palmitate;NSC 8918;
- PSA 26.30000
- LogP 6.03080
Synthetic route

| Conditions | Yield |
|---|---|
| With monoammonium 12-tungstophosphate for 12h; Heating; | 98% |
| With alumina methanesulfonic acid at 120℃; for 0.333333h; Microwave irradiation; | 97% |
| With polysiloxane acidic ionic liquids containing pyridinium trifluoroacetate salts for 4h; Reflux; | 96% |
-
-
64-17-5
ethanol

-
-
555-44-2
glyceroltripalmitate

-
A
-
628-97-7
hexadecanoic acid ethyl ester

-
B
-
56-81-5
glycerol

| Conditions | Yield |
|---|---|
| With lithium perchlorate 1) electrolyzis; 2) reflux, 7h; | A 96% B n/a |

-
-
78-39-7
Triethyl orthoacetate

-
-
57-10-3
1-hexadecylcarboxylic acid

-
-
628-97-7
hexadecanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With 3-butyl-1-methyl-1H-imidazol-3-ium hexafluorophosphate at 100℃; for 3.5h; | 95% |
| In various solvent(s) at 100℃; for 3.5h; | 95% |

| Conditions | Yield |
|---|---|
| With CuCl2*2H2O for 3h; Ambient temperature; | 90% |

| Conditions | Yield |
|---|---|
| With CuCl2*2H2O for 3h; Ambient temperature; | 90% |

| Conditions | Yield |
|---|---|
| With palmitic acid imprinted Thermomyces lanuginosus Lypozyme TL 100L lipase nanogel In n-heptane at 40℃; for 1h; Reagent/catalyst; Green chemistry; Enzymatic reaction; | 77% |
| With constitutive mycelium-bound lipase from Aspergillus niger MYA 135 In hexane; acetone at 37℃; for 1h; Reagent/catalyst; Enzymatic reaction; | |
| With lipase encapsulated polyacrylamide nanogel In n-heptane Reagent/catalyst; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; titanium tetrachloride for 12h; Heating; | 74% |

| Conditions | Yield |
|---|---|
| With PDVB-VI-0.5 at 78℃; for 3h; | 64.3% |

| Conditions | Yield |
|---|---|
| With xylene at 100℃; |
-
-
50-00-0
formaldehyd

-
-
57-10-3
1-hexadecylcarboxylic acid

-
A
-
112-39-0
hexadecanoic acid methyl ester

-
B
-
628-97-7
hexadecanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| at 300℃; im Rohr; |


| Conditions | Yield |
|---|---|
| With diethyl ether; magnesium |
-
-
65260-62-0
Palmitoyl p-toluolsulphonyldiester

-
-
628-97-7
hexadecanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In N,N-dimethyl-formamide |

| Conditions | Yield |
|---|---|
| In methanol; acetone for 16h; Heating; |
-
-
64-17-5
ethanol

-
-
182698-28-8
3-O-palmitoyl-D-glucopyranose

-
A
-
2280-44-6
D-Glucose

-
B
-
628-97-7
hexadecanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With water; sulfuric acid at 50℃; Rate constant; |

-
-
64-17-5
ethanol

-
-
122156-12-1
3-O-Palmitoyl-1,2-O-isopropylidene-α-D-glucofuranose

-
A
-
70834-19-4, 138343-45-0, 138343-46-1
5,6-O-isopropylidene-D-glucofuranose

-
B
-
628-97-7
hexadecanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With water; sulfuric acid at 50℃; Rate constant; |

-
-
64-17-5
ethanol

-
-
24613-34-1
3-O-Palmitoyl-1,2:5,6-di-O-isopropylidene-α-D-glucofuranose

-
A
-
628-97-7
hexadecanoic acid ethyl ester

-
B
-
582-52-5
1,2:5,6-di-O-isopropylidene-α-D-glucofuranose

| Conditions | Yield |
|---|---|
| With water; sulfuric acid at 50℃; Rate constant; |

-
-
64-17-5
ethanol

-
-
2190-25-2
2-Oleodipalmitin

-
A
-
111-62-6
oleic acid ethyl ester

-
B
-
628-97-7
hexadecanoic acid ethyl ester

-
C
-
502-52-3
hexadecanoic acid, 2-hydroxy-1,3-propanediyl ester

-
D
-
3123-73-7
1-O-palmitoyl-2-O-oleoyl glycerol

| Conditions | Yield |
|---|---|
| With immobilized EL1 lipase In water; tert-butyl alcohol at 30℃; for 4.16667h; | A 45 mmol B 11 mmol C n/a D n/a E n/a |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. NH4OH / CH2Cl2 / 0 - 20 °C 2: 74 percent / TiCl4, aq. HCl / 12 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: benzene 2: NaBH4 / dimethylformamide View Scheme |

-
-
64-17-5
ethanol

-
B
-
111-62-6
oleic acid ethyl ester

-
C
-
544-35-4
ethyl (9Z,12Z)-9,12-octadecadienoate

-
D
-
1191-41-9
ethyl linolenate

-
E
-
628-97-7
hexadecanoic acid ethyl ester

-
F
-
14010-23-2
ethyl heptadecanoate

-
G
-
111-61-5
stearic acid ethyl ester

| Conditions | Yield |
|---|---|
| sulfuric acid at 60℃; for 1h; Conversion of starting material; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: iodine / acetonitrile / 0.17 h / Inert atmosphere 2.1: zinc trifluoromethanesulfonate / acetonitrile / 0.5 h / 60 °C / Inert atmosphere 2.2: 3.5 h / 60 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: C34H46O2P(1+)*I(1-) With zinc trifluoromethanesulfonate In acetonitrile at 60℃; for 0.5h; Inert atmosphere; Stage #2: ethanol In acetonitrile at 60℃; for 3.5h; Inert atmosphere; | 55 mg |

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; potassium carbonate In tetrahydrofuran for 21h; Inert atmosphere; Reflux; |
-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
36653-82-4
1-Hexadecanol

| Conditions | Yield |
|---|---|
| With C32H36ClNO2P2Ru; potassium tert-butylate; hydrogen In tetrahydrofuran at 120℃; under 38002.6 Torr; for 20h; Autoclave; Green chemistry; | 98% |
| With C30H34Cl2N2P2Ru; potassium methanolate; hydrogen In tetrahydrofuran at 100℃; under 38002.6 - 76005.1 Torr; for 15h; Glovebox; Autoclave; | 92% |
| With aluminum oxide; sodium; tert-butyl alcohol In toluene for 6h; Heating; | 70% |

| Conditions | Yield |
|---|---|
| With Novozym 435 at 37℃; | 98% |
-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
10597-60-1
hydroxytyrosol

| Conditions | Yield |
|---|---|
| With Novozym 435 at 37℃; | 98% |

| Conditions | Yield |
|---|---|
| With Novozym 435 In 1,4-dioxane for 0.0666667h; Microwave irradiation; Enzymatic reaction; chemoselective reaction; | 97.5% |
-
-
46118-02-9
3-(3',4'-dihydroxyphenyl)-1-propanol

-
-
628-97-7
hexadecanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With Novozym 435 at 37℃; | 97% |
-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
100-46-9
benzylamine

-
-
74058-71-2
hexadecanoic acid benzylamide

| Conditions | Yield |
|---|---|
| With indium (III) iodide at 110 - 120℃; for 8.5h; | 91% |
| at 150℃; |
-
-
554-62-1
D-ribo-phytosphingosine

-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
111149-09-8
N-palmitoyl-D-ribo-phytosphingosine

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 55℃; | 88.6% |
-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
56555-03-4
<1,1-D2>cetyl alcohol

| Conditions | Yield |
|---|---|
| With lithium aluminium deuteride In diethyl ether at 25℃; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: hexadecanoic acid ethyl ester With sodium diisobutyl-tert-butoxyaluminium hydride In tetrahydrofuran at 0℃; for 2h; Inert atmosphere; Stage #2: With 1,3-Diiodo-5,5-dimethyl-2,4-imidazolidinedione; ammonia In tetrahydrofuran; water at 0 - 20℃; for 3h; | 86% |
| Stage #1: hexadecanoic acid ethyl ester With sodium diisobutyl-tert-butoxyaluminium hydride In tetrahydrofuran at 0℃; for 4h; Inert atmosphere; Stage #2: With ammonium hydroxide; 1,3-Diiodo-5,5-dimethyl-2,4-imidazolidinedione In tetrahydrofuran at 0℃; for 2h; Inert atmosphere; | 351 mg |

| Conditions | Yield |
|---|---|
| With indium; iodine for 4.5h; transesterification; Heating; | 85% |
| With Dowex DR2030 ion exchange resin at 59.84℃; under 760.051 Torr; for 3h; |
-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
119119-53-8
α-Tetradecyl-β-keto-stearinsaeureethylester

| Conditions | Yield |
|---|---|
| With sodium ethanolate at 140℃; for 2h; Inert atmosphere; | 80% |
| With sodium ethanolate Heating; |

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran; N,N,N,N,N,N-hexamethylphosphoric triamide 1.) -78 deg C, 20 min, 2.) r.t., 1.5 h; | 73% |

| Conditions | Yield |
|---|---|
| With Novozym 435 at 37℃; | 71% |
-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
106-95-6
allyl bromide

-
-
123546-59-8
ethyl 2-(2-propenyl)hexadecanoate

| Conditions | Yield |
|---|---|
| With n-butyllithium; diisopropylamine In tetrahydrofuran | 67% |
| With lithium diisopropyl amide 1.) THF, hexane, 0 deg C, 1 h, 2.) RT, 12 h; Yield given. Multistep reaction; |
-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
10314-06-4
4-phthalimidobutyryl chloride

-
-
322391-58-2
β-ketoester (32)

| Conditions | Yield |
|---|---|
| Stage #1: 4-phthalimidobutyric acid With thionyl chloride for 3h; Heating / reflux; Stage #2: hexadecanoic acid ethyl ester; 4-phthalimidobutyryl chloride; lithium hexamethyldisilazane In tetrahydrofuran at -20 - 20℃; for 2.33333h; Heating / reflux; Stage #3: With ammonium chloride In water | 37% |

| Conditions | Yield |
|---|---|
| With Lipozyme TL IM In tert-Amyl alcohol at 40℃; Enzymatic reaction; | 20% |
-
-
10467-10-4
ethylmagnesium iodide

-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
35185-53-6
3-ethyl-octadecan-3-ol

| Conditions | Yield |
|---|---|
| With ethanol |
-
-
693-03-8
n-butyl magnesium bromide

-
-
628-97-7
hexadecanoic acid ethyl ester

-
A
-
105359-53-3
5-butyl-eicosan-5-ol

-
B
-
13287-17-7
5-butyl-eicos-4-ene

| Conditions | Yield |
|---|---|
| With diethyl ether |

-
-
917-64-6
methyl magnesium iodide

-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
35177-29-8
2-methylheptadecan-2-ol

| Conditions | Yield |
|---|---|
| With diethyl ether |

| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
628-97-7
hexadecanoic acid ethyl ester

-
-
925-90-6
ethylmagnesium bromide

-
-
35185-53-6
3-ethyl-octadecan-3-ol

| Conditions | Yield |
|---|---|
| With diethyl ether |
Ethyl palmitate Chemical Properties
The Molecular Weight of Ethyl palmitate(628-97-7):284.4772
The Molecular Structure of Ethyl palmitate(628-97-7) is:
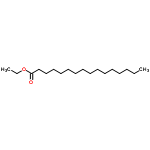
Density:0.864 g/cm3
Melting point:24-26 °C(lit.)
Boiling point:342.2 °C at 760 mmHg
Flash point:149 °C
Solubility in water:IMMISCIBLE
Index of Refraction :1.442
Molar Refractivity: 87.21 cm3
Molar Volume:329.1 cm3
Polarizability:34.57 10-24cm3
Surface Tension:30.4 dyne/cm
Enthalpy of Vaporization:58.6 kJ/mol
Vapour Pressure:7.63E-05 mmHg at 25°C
Appearance:colorless needle-like crystals
IUPAC Name:ethyl hexadecanoate
Synonyms:PALMITIC ACID ETHYL ESTER;HEXADECANOIC ACID ETHYL ESTER;Ethyl palmitate;Ethyl cetylate;ETHYL HEXADECANOATE;ETHYL PALMITATE;ETHYL N-HEXADECANOATE;FEMA 2451;Ethyl hexadecanoate
Ethyl palmitate Uses
Ethyl palmitate Toxicity Data With Reference
LD50/LC50: RTECS: Not available.
Carcinogenicity: Ethyl palmitate - Not listed as a carcinogen by ACGIH, IARC, NTP, or CA Prop 65.
Other: The toxicological properties have not been fully investigated.
Ethyl palmitate Safety Profile
Hazard Codes:Xi
Xi:Irritant
Safety Statements :
S23:Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer) .
S24/25:Avoid contact with skin and eyes .
WGK Germany:3
Hazard Note:Irritant
HS Code:29157020
Ethyl palmitate Specification
Conditions to Avoid: Incompatible materials.
Incompatibilities with Other Materials Strong oxidizing agents.
Hazardous Decomposition Products Carbon monoxide, carbon dioxide.
Hazardous Polymerization Will not occur.
Related Products
- Ethyl (13-cis)-9-(4-methoxy-2,3,6-trimethylphenyl)-3,7-dimethyl-2,4,6,8-nonatetraenoate
- ethyl (1R,2R)-1-phenyl-2-(trideuteriomethylamino)cyclohex-3-ene-1-carboxylate,hydrochloride
- Ethyl (1S,2R)-2-(dimethylamino)-1-phenylcyclohex-3-ene-1-carboxylate hydrochloride
- Ethyl (2,4,6-trimethylbenzoyl) phenylphosphinate
- Ethyl (2-amino-4-hydroxy-6-methyl-5-pyrimidinyl)acetate
- Ethyl (2-bromopropionamido)acetate
- Ethyl (2-cyanoimino-5,6-dichloro-1,2,3,4-tetrahydroquinazolin-3-yl)acetate
- ETHYL (2E,4Z)-DECADIENOATE
- Ethyl (2-hydroxyethyl)dimethyl-ammonium benzilate chloride
- Ethyl (2-mercaptoethyl) carbamate S-ester with O,O-dimethyl phosphorodithioate
- 62897-94-3
- 62899-78-9
- 628-99-9
- 6290-03-5
- 6290-05-7
- 6290-17-1
- 6290-24-0
- 629-03-8
- 629-04-9
- 6290-49-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View