-
Name
FLAVONE
- EINECS 208-383-8
- CAS No. 525-82-6
- Article Data312
- CAS DataBase
- Density 1.24 g/cm3
- Solubility Soluble in acetone (25 mg/ml), methanol, alcohol, and chloroform. Insoluble in water.
- Melting Point 94-97 °C(lit.)
- Formula C15H10O2
- Boiling Point 367.013 °C at 760 mmHg
- Molecular Weight 222.243
- Flash Point 171.058 °C
- Transport Information
- Appearance white crystalline powder
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2-Phenyl-4H-1-benzopyran-4-one;2-Phenyl-4H-chromen-4-one;2-Phenyl-g-benzopyrone;Chromocor;NSC 19028;
- PSA 30.21000
- LogP 3.46000
Synthetic route

| Conditions | Yield |
|---|---|
| copper dichloride In ethanol at 80℃; for 0.0833333h; microwave irradiation; | 98% |
| With potassium hydrogensulfate at 120℃; for 2h; Temperature; Green chemistry; | 98% |
| With gallium(III) triflate In nitromethane at 80℃; for 2h; | 97% |

| Conditions | Yield |
|---|---|
| With C22H28Br2N4Pd; diethylamine In N,N-dimethyl-formamide at 80℃; under 3000.3 Torr; for 24h; Autoclave; | 98% |
| With diethylamine In N,N-dimethyl-formamide at 100℃; under 10343.2 Torr; for 24h; Sonogashira Cross-Coupling; Autoclave; | 96% |
| With (trihexyl)(tetradecyl)phosphonium bromide; triethylamine; palladium dichloride at 110℃; under 760.051 Torr; for 24h; Autoclave; | 95% |


| Conditions | Yield |
|---|---|
| With 1-butyl-3-methylimidazolium molybdate In hexan-1-ol at 70℃; for 3h; | 98% |

| Conditions | Yield |
|---|---|
| With pyrrolidone hydrotribromide In dimethyl sulfoxide at 80℃; for 2h; | 97% |
| With thallium(III) acetate In acetic acid for 3h; Heating; | 96% |
| With thallium(III) acetate In acetic acid for 3h; Heating; | 96% |

| Conditions | Yield |
|---|---|
| With indium(III) bromide; silica gel at 130 - 140℃; for 0.75h; | 96% |
| With iodine In dimethyl sulfoxide for 1h; Reflux; | 85% |
| With iodine In 2,2'-[1,2-ethanediylbis(oxy)]bisethanol at 150℃; for 4h; Product distribution; Further Variations:; Solvents; Temperatures; reaction times; | 83% |
-
-
16619-68-4
1-(2-hydroxyphenyl)-3-phenylprop-2-yn-1-one

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With dmap In N,N-dimethyl-formamide at 0 - 30℃; for 1.5h; Morita-Baylis-Hillman reaction; Inert atmosphere; regioselective reaction; | 96% |
| With thallium(III) toluene-p-sulfonate In methanol at 65℃; for 6h; Reagent/catalyst; Solvent; Time; regioselective reaction; | 91% |
| With lithium tert-butoxide In N,N-dimethyl-formamide at 20℃; for 0.166667h; Reagent/catalyst; Time; Temperature; | 90% |
-
-
81995-11-1
1-(2-hydroxyphenyl)-2-(triphenylphosphoranylidene)ethanone

-
-
98-88-4
benzoyl chloride

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With pyridine In toluene for 5h; Heating; | 96% |

| Conditions | Yield |
|---|---|
| With Iodine monochloride; dimethyl sulfoxide at 50℃; for 0.5h; Temperature; Reagent/catalyst; Sonication; Green chemistry; | 96% |
| With cerium (IV) sulfate tetrahydrate; silica gel In neat (no solvent) at 100℃; for 4h; Concentration; Time; Green chemistry; | 94% |
| With copper(l) iodide; oxygen at 50℃; under 760.051 Torr; for 48h; oxa-Michael-oxidation; Ionic liquid; | 92% |
-
-
223511-19-1
1-[2-(tert-butyldimethylsilyloxy)phenyl]-3-phenylprop-2-yn-1-one

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With diethylamine In ethanol for 24h; Heating; | 96% |
-
-
16619-51-5
2'-allyloxychalcone

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With iodine In dimethyl sulfoxide at 120℃; for 0.5h; regioselective reaction; | 96% |
| Multi-step reaction with 2 steps 1: acetic acid / 24 h / 20 °C 2: iodine / dimethyl sulfoxide / 0.5 h / 130 °C View Scheme |

| Conditions | Yield |
|---|---|
| With palladium(II) trifluoroacetate; 5-Nitro-1,10-phenanthroline; oxygen In dimethyl sulfoxide at 100℃; for 48h; Reagent/catalyst; Solvent; | 96% |
-
-
16619-68-4
1-(2-hydroxyphenyl)-3-phenylprop-2-yn-1-one

-
A
-
525-82-6
FLAVONE

-
B
-
36685-41-3
(Z)-4'-methoxyaurone

| Conditions | Yield |
|---|---|
| With silver nitrate In methanol Ambient temperature; | A 5% B 95% |

| Conditions | Yield |
|---|---|
| With silver nitrate In methanol Ambient temperature; | A 5% B 95% |
| With 2-pyridone sodium salt In tetrahydrofuran at 20℃; for 6h; Reagent/catalyst; Solvent; Temperature; Time; regioselective reaction; | A 3% B 93% |
| With tricyclohexylphosphine In ethanol at 30℃; for 2h; | A n/a B 55% |
-
-
16619-67-3
1-(2-acetoxy-phenyl)-3-phenyl-2-propyn-1-one

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With piperazine In acetonitrile at 25℃; for 3h; Schlenk technique; | 95% |
| With piperazine In acetonitrile at 20℃; for 6h; Schlenk technique; | 95% |
| With 18-crown-6 ether; potassium methanolate In tetrahydrofuran at 20℃; for 0.25h; Reagent/catalyst; Time; Temperature; Solvent; | 76% |

| Conditions | Yield |
|---|---|
| With piperazine; palladium diacetate In acetonitrile at 50℃; for 12h; Schlenk technique; | 95% |

-
-
93514-42-2, 6053-99-2
2-phenylchromene

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; copper(ll) bromide In decane; toluene at 80℃; for 0.0833333h; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; | 94% |
| With thallium(III) nitrate In methanol Ambient temperature; overnight; | 20% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-benzopyran-4(4H)-one; phenylboronic acid With iron(III) trifluoromethanesulfonate; palladium diacetate; Trimethylacetic acid at 60℃; under air; Stage #2: With potassium nitrite; 2,3-dicyano-5,6-dichloro-p-benzoquinone | 94% |
| Stage #1: 1-benzopyran-4(4H)-one With 1,10-Phenanthroline; oxygen; palladium diacetate In N,N-dimethyl-formamide for 0.0833333h; Heck type reaction; Stage #2: phenylboronic acid In N,N-dimethyl-formamide at 100℃; for 24h; Heck type reaction; regioselective reaction; | 86% |
-
-
491-37-2
2,3-dihydro-4H-1-benzopyran-4-one

-
-
24388-23-6
2-phenyl-4,4,5,5-tetramethyl-1,3,2-dioxoborole

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With palladium(II) trifluoroacetate; 5-Nitro-1,10-phenanthroline; oxygen In dimethyl sulfoxide at 100℃; for 48h; | 93% |
| With palladium(II) trifluoroacetate; 5-Nitro-1,10-phenanthroline; oxygen In dimethyl sulfoxide at 100℃; Heck Reaction; | 93% |

| Conditions | Yield |
|---|---|
| pyrex In benzene Irradiation; | A 4% B 92% |
-
-
32865-61-5
2-iodophenyl acetate

-
-
201230-82-2
carbon monoxide

-
-
536-74-3
phenylacetylene

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene; diethylamine; thiourea; bis-triphenylphosphine-palladium(II) chloride; 1,3-bis-(diphenylphosphino)propane at 40℃; for 48h; Addition; cyclization; | 92% |

-
-
98153-12-9
3-iodo-2-phenyl-4H-1-benzopyran-4-one

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With sodium formate; bis-triphenylphosphine-palladium(II) chloride In N,N-dimethyl-formamide at 90℃; | 92% |
| With copper(l) iodide; potassium carbonate; isopropyl alcohol at 90℃; for 12h; Green chemistry; regioselective reaction; | 92% |
-
-
137527-18-5
4-oxo-2-phenyl-4H-chromen-6-yl 4-methylbenzenesulfonate

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With dimethylamine borane; potassium carbonate; triphenylphosphine; nickel In N,N-dimethyl-formamide at 120℃; for 12h; | 92% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; potassium carbonate; isopropyl alcohol at 90℃; for 12h; Catalytic behavior; Reagent/catalyst; Green chemistry; regioselective reaction; | 92% |

| Conditions | Yield |
|---|---|
| With dibutylamine In dimethyl sulfoxide for 72h; Ambient temperature; | A 91.5% B 6.2% |
| With sodium acetate In dimethyl sulfoxide at 25℃; for 144h; | A 80.1% B 10.9% |
| With sodium acetate In dimethyl sulfoxide at 25℃; for 144h; Product distribution; other reaction temperatures, solvents, reaction time, reagents, catalists, other amount of catalists and use other 3-mesyloxy-flavanone; | A 80.1% B 10.9% |
| With potassium cyanate; 18-crown-6 ether In benzene for 21h; Heating; | A 31.3% B 48% |
| With isopropylamine In dimethyl sulfoxide for 72h; Ambient temperature; | A 57.5 % Spectr. B 32.9 % Spectr. |
-
-
98153-26-5
1-(2-Hydroxy-4-methoxyphenyl)-3-phenylpropyn-1-one

-
A
-
525-82-6
FLAVONE

-
B
-
22395-22-8
7-methoxoyflavone

| Conditions | Yield |
|---|---|
| With silver nitrate In methanol Ambient temperature; | A 91% B n/a |
-
-
108378-93-4
3-<(4-Methylphenyl)sulphinyl>-4H-1-benzopyran-4-one

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With copper(I) bromide dimethylsulfide complex In tetrahydrofuran at -78 - 0℃; for 2.5h; | 91% |
-
-
36081-80-8
1-(2'-bromophenyl)-3-phenylpropane-1,3-dione

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 1h; Ullmann type reaction; under air; | 91% |
| With tert-butylisonitrile; palladium diacetate; potassium carbonate; bis[2-(diphenylphosphino)phenyl] ether In N,N-dimethyl-formamide at 120℃; for 2h; Reagent/catalyst; Solvent; chemoselective reaction; | 86% |
-
-
1657006-73-9
C18H16Br2O2

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With iodine In dimethyl sulfoxide at 130℃; for 0.5h; | 91% |

| Conditions | Yield |
|---|---|
| With Clayfen for 0.025h; microwave irradiation; | 90% |
| With tetraethylammonium perchlorate; ethidium Bromide In N,N-dimethyl-formamide cathodic reduction; | |
| With air In ethanol at 20℃; Photolysis; |
-
-
75630-53-4
2'-hydroxychalcone dichloride

-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| at 210 - 220℃; for 1.5h; | 90% |
-
-
525-82-6
FLAVONE

-
-
134051-30-2
1a,7a-Dihydro-1a-phenyl-7H-oxireno<1>benzopyran-7-one

| Conditions | Yield |
|---|---|
| With 3,3-dimethyldioxirane In dichloromethane; acetone at 0℃; for 36h; | 100% |
| With 3,3-dimethyldioxirane In dichloromethane; acetone at 0℃; for 36h; | 100% |
| With Oxone; 1,1,1-trifluoro-2-propanone; edetate disodium; sodium hydrogencarbonate In acetonitrile at 0 - 1℃; for 1.25h; | 99% |

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; copper(II) acetate monohydrate In tert-Amyl alcohol at 20 - 100℃; for 12.33h; Schlenk technique; Sealed tube; regioselective reaction; | 99% |
-
-
525-82-6
FLAVONE

-
A
-
487-26-3, 17002-31-2, 24271-98-5, 27439-12-9
(2S)-flavanone

-
B
-
487-26-3, 17002-31-2, 24271-98-5, 27439-12-9
(R)-flavanone

| Conditions | Yield |
|---|---|
| Stage #1: FLAVONE With Dimethylphenylsilane; C50H52N2; bis(pentafluorophenyl)borohydride In toluene at 20℃; for 1h; Inert atmosphere; Glovebox; Stage #2: With trifluoroacetic acid In toluene at 20℃; for 0.166667h; Inert atmosphere; Glovebox; enantioselective reaction; | A n/a B 99% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Solvent; | 98.72% |
-
-
525-82-6
FLAVONE

-
-
13178-98-8
3-chloro-2-phenyl-chromen-4-one

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride; Montmorillonite-K-10 clay In dichloromethane for 0.25h; Ambient temperature; | 98% |
| With pyridine; chloro-trimethyl-silane; [bis(acetoxy)iodo]benzene In dichloromethane at 0℃; for 3.5h; | 82% |
| With sulfuryl dichloride In tetrachloromethane | 63% |
-
-
525-82-6
FLAVONE

-
-
71840-34-1, 71840-35-2, 82908-23-4
(2R*,2’R*)-2,2’-diphenyl-[2,2’-bichromane]-4,4’-dione

| Conditions | Yield |
|---|---|
| With samarium diiodide In tetrahydrofuran; methanol at -40℃; for 24h; Reagent/catalyst; Temperature; Solvent; Inert atmosphere; stereoselective reaction; | 98% |
-
-
525-82-6
FLAVONE

-
-
71840-35-2
(2R*,2’S*)-2,2’-diphenyl-[2,2’-bichromane]-4,4’-dione

| Conditions | Yield |
|---|---|
| With samarium diiodide In tetrahydrofuran; methanol at 68℃; for 24h; Inert atmosphere; stereoselective reaction; | 98% |
-
-
525-82-6
FLAVONE

-
-
98153-13-0
3-Mercapto-2-phenyl-4H-1-benzopyran-4-one

| Conditions | Yield |
|---|---|
| With sulfur; lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.583333h; | 97% |
-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With methanol; sodium tetrahydroborate; cobalt(II) 5,10,15,20-tetraphenylporphyrin; oxygen at 20℃; for 0.166667h; Kinetics; Reagent/catalyst; Time; | 97% |
-
-
525-82-6
FLAVONE

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 20℃; for 3h; Inert atmosphere; | 95% |
-
-
941-55-9
4-toluenesulfonyl azide

-
-
525-82-6
FLAVONE

-
-
1607017-87-7
4-methyl-N-(4-oxo-2-phenyl-4H-chromen-5-yl)benzenesulfonamide

| Conditions | Yield |
|---|---|
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; lithium carbonate; acetic acid; silver(I) triflimide In 1,2-dichloro-ethane at 25℃; for 12h; Inert atmosphere; | 95% |
-
-
525-82-6
FLAVONE

-
-
19726-12-6
3-(2-hydroxyphenyl)-5-phenyl-pyrazole

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol at 90℃; | 95% |

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; Trimethylacetic acid In 1,2-dichloro-ethane at 80℃; for 20h; Sealed tube; | 94% |
Flavone Chemical Properties
Synonyms :2-Phenyl-4H-1-benzopyran-4-one; 2-Phenylchromone
MF: C15H10O2
MW: 222.24
EINECS: 208-383-8
2-PHENYLCHROMONE(525-82-6),its melting point is about 94-97 °C(lit.),boiling point is 185 °C / 1mmHg .
Following is the molecular structure of 2-PHENYLCHROMONE(525-82-6):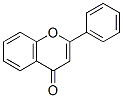
Flavone Toxicity Data With Reference
| 1. | dni-hmn:fbr 100 mg/L | BCPCA6 Biochemical Pharmacology. 33 (1984),3823. | ||
| 2. | orl-mus LD50:2500 mg/kg | JHTCAO Journal of Heterocyclic Chemistry. 21 (1984),193. | ||
| 3. | ipr-mus LD50:380 mg/kg | FRPSAX Farmaco, Edizione Scientifica. 13 (1958),561. | ||
| 4. | scu-mus LD >400 mg/kg | ANTCAO Antibiotics and Chemotherapy. 3 (1953),603. |
RTECS : DJ3100630
Flavone Consensus Reports
Flavone Safety Profile
Poison by intraperitoneal route. Moderately toxic by ingestion and subcutaneous routes. Mutation data reported. When heated to decomposition it emits acrid smoke and irritating vapors.
Other safty informations about 2-PHENYLCHROMONE(525-82-6):
Hazard Codes: Xi (Irritant)
Risk Statements
R 36/37/38 (Irritating to eyes, respiratory system and skin )
Safety Statements :
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S36 :Wear suitable protective clothing
WGK Germany : 2
Related Products
- Flavone
- 52583-42-3
- 52583-54-7
- 52583-87-6
- 52584-47-1
- 52589-11-4
- 52589-14-7
- 52589-39-6
- 52590-49-5
- 52590-61-1
- 52591-27-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View