-
Name
Isobutyl vinyl ether
- EINECS 203-678-8
- CAS No. 109-53-5
- Article Data17
- CAS DataBase
- Density 0.773 g/cm3
- Solubility Insoluble (soluble in alcohol and ether)
- Melting Point -112 °C
- Formula C6H12O
- Boiling Point 83 °C at 760 mmHg
- Molecular Weight 100.161
- Flash Point 8°F
- Transport Information UN 1304 3/PG 2
- Appearance clear colourless liquid
- Safety 16-23-24/25-33-37
- Risk Codes 11-38
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xi
Xi
- Synonyms Isobutyl vinyl ether;Lutonal LR 8500;NSC 8265;Vinoflex MO 400;Vinyl isobutyl ether;iso-Butyl vinyl ether;Ether,isobutyl vinyl (8CI);1-(Ethenyloxy)-2-methylpropane;IVE;Isobutanol vinylether;Isobutoxyethene;
- PSA 9.23000
- LogP 1.80250
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium hydroxide; cesium fluoride at 135 - 140℃; under 7600 - 10640 Torr; for 2h; | 82% |
| With potassium hydroxide at 135 - 155℃; | |
| at 140℃; under 4500.45 Torr; |

| Conditions | Yield |
|---|---|
| With sodium hydrogen sulfate | |
| With quinoline; phosphorus pentoxide |
-
-
19865-33-9
α-chloroethyl isobutyl ether

-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| With pyridine |

| Conditions | Yield |
|---|---|
| With N,N-diethylaniline at 185 - 190℃; |

| Conditions | Yield |
|---|---|
| With zinc diacetate at 210 - 220℃; | |
| With copper(I) monacetylide at 210 - 220℃; |

| Conditions | Yield |
|---|---|
| Stage #1: 2-methyl-propan-1-ol; acetaldehyde With hydrogenchloride In dibutyl ether at 10 - 15℃; Stage #2: With 3-Methylpyridine In dibutyl ether at 50℃; for 1h; |

| Conditions | Yield |
|---|---|
| With potassium fluoride; water; potassium hydroxide In neat (no solvent) at 160℃; for 13h; Temperature; Autoclave; | 75.8 %Spectr. |
-
-
109-53-5
isobutyl vinyl ether

-
-
59938-06-6
(E)-4-Ethoxy-1,1,1-trifluoro-3-buten-2-one

-
-
1214317-50-6
4-Ethoxy-2-isobutoxy-6-trifluoromethyl-3,4-dihydro-2H-pyran

| Conditions | Yield |
|---|---|
| With hydroquinone at 80℃; for 30h; | 100% |
-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| In phosgene | 100% |
-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In tetrahydrofuran at 20℃; for 4h; Cooling; Large scale; | 100% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 20 - 25℃; | 100% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 60 - 65℃; | 99% |
| toluene-4-sulfonic acid In diethyl ether for 1h; Heating; | 72.4% |
-
-
109-53-5
isobutyl vinyl ether

-
-
107969-78-8
methyl benzylidenepyruvate

| Conditions | Yield |
|---|---|
| With 5percent Eu(fod)3 In dichloromethane for 48h; Cycloaddition; Heating; | 99% |

-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| at 80℃; for 5h; hetero-Diels-Alder reaction; | 99% |
-
-
109-53-5
isobutyl vinyl ether

-
-
107969-78-8, 6395-86-4
methyl 4-phenyl-2-oxo-3-butenoate

| Conditions | Yield |
|---|---|
| Stage #1: methyl 4-phenyl-2-oxo-3-butenoate With C39H60N4O4; erbium(III) triflate In dichloromethane at 25℃; for 0.5h; hetero Diels-Alder reaction; Inert atmosphere; Stage #2: isobutyl vinyl ether In dichloromethane at 0℃; for 12h; hetero Diels-Alder reaction; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 99% |
-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| With nickel(II) tetrafluoroborate hexahydrate; C33H48N4O4 In dichloromethane at 30℃; for 4h; Diels-Alder Cycloaddition; Inert atmosphere; stereoselective reaction; | 99% |
-
-
109-53-5
isobutyl vinyl ether

-
-
82849-49-8
dimethyl 2,2-dicyanoethene-1,1-dicarboxylate

-
-
82849-59-0
dimethyl 3-isobutoxy-2,2-dicyanocyclobutane-1,1-dicarboxylate

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; for 12h; Product distribution; other electrofilic olefins, var. reagents ratios; | 98% |
| at 25℃; for 12h; | 98% |
| In acetonitrile Ambient temperature; | 95% |
-
-
905560-26-1
(E)-2-(4-Chloro-1,1,2,2,3,3,4,4-octafluoro-butane-1-sulfonyl)-1,3-diphenyl-propenone

-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| at 80℃; for 5h; hetero-Diels-Alder reaction; | 98% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; water In neat (no solvent) at 80 - 100℃; for 3.5h; Temperature; | 97% |
| Hydrolysis; |
-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| With NCNMe2 In tetrahydrofuran at 60 - 65℃; for 5h; | 97% |

-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| at 80℃; for 5h; hetero-Diels-Alder reaction; | 97% |
-
-
109-53-5
isobutyl vinyl ether

-
-
80657-57-4
3-hydroxy-(2S)-methylpropionate

| Conditions | Yield |
|---|---|
| With sodium hydroxide; pyridinium p-toluenesulfonate In water | 97% |

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate In toluene at 60℃; for 12h; Schlenk technique; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 60 - 65℃; | 97% |
-
-
109-53-5
isobutyl vinyl ether

-
-
61597-96-4
Isobutyl (R)-lactate

| Conditions | Yield |
|---|---|
| With trichlorophosphate In ethyl acetate | 96.9% |
-
-
109-53-5
isobutyl vinyl ether

-
-
95407-70-8
1-oxo-4-methoxy-2,2,6,6-tetramethylpiperidinium chloride

| Conditions | Yield |
|---|---|
| In chloroform at 20℃; for 0.05h; Addition; | 96% |
-
-
905560-32-9
(E)-2-(4-Chloro-1,1,2,2,3,3,4,4-octafluoro-butane-1-sulfonyl)-3-furan-2-yl-1-phenyl-propenone

-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; hetero-Diels-Alder reaction; | 96% |

-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; hetero-Diels-Alder reaction; | 96% |

| Conditions | Yield |
|---|---|
| pyridinium p-toluenesulfonate In water; ethyl acetate | 95.1% |
-
-
109-53-5
isobutyl vinyl ether

-
-
147666-20-4
succinimidyl diazoacetate

| Conditions | Yield |
|---|---|
| With [(4,5-dihydro-4,4-dimethyl-2-phenyloxazole)Ru(CH3CN)4]PF6 In dichloromethane at 20℃; for 0.0166667h; Inert atmosphere; optical yield given as %de; diastereoselective reaction; | 95% |
-
-
109-53-5
isobutyl vinyl ether

| Conditions | Yield |
|---|---|
| With C30H41N2Ni(1+)*C32H12BF24(1-) In toluene at 20℃; for 3h; Inert atmosphere; | 95% |
| With [(1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene)NiH(OTf)]; nitrobenzene In toluene at 20℃; for 24h; regioselective reaction; |
-
-
109-53-5
isobutyl vinyl ether

-
-
998-30-1
Triethoxysilane

-
-
1422284-26-1
triethoxy(2-isobutoxyethyl)silane

| Conditions | Yield |
|---|---|
| Stage #1: isobutyl vinyl ether With Wilkinson's catalyst for 0.0833333h; Stage #2: Triethoxysilane at 90℃; for 5h; | 94.2% |
| With N-heterocyclic carbene fuctionalized with methyl(trimethylsilane) and methoxypolyethylene glycol Pt(II) complex at 90℃; for 10h; | 85.7% |
| With Wilkinson's catalyst; 1-(2-carboxyethyl)-3-methylimidazolium inner salt at 90℃; for 5h; | |
| With Wilkinson's catalyst Catalytic behavior; Reagent/catalyst; Heating; |

| Conditions | Yield |
|---|---|
| With 2,6-di-tert-butyl-4-methyl-phenol at 135℃; under 22502.3 Torr; for 16h; | 94% |
-
-
56-23-5
tetrachloromethane

-
-
109-53-5
isobutyl vinyl ether

-
-
78846-85-2
Isobutyl-(1,3,3,3-tetrachlor-propyl)-ether

| Conditions | Yield |
|---|---|
| With dibenzoyl peroxide for 5h; Heating; | 93% |
-
-
696-63-9
4-Methoxybenzenethiol

-
-
109-53-5
isobutyl vinyl ether

-
-
355-43-1
n-perfluorohexyl iodide

-
-
135984-60-0
1-Methoxy-4-(3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluoro-1-isobutoxy-octylsulfanyl)-benzene

| Conditions | Yield |
|---|---|
| With sodium phenylselenide; sodium hydride In diethyl ether for 2h; Heating; | 93% |


| Conditions | Yield |
|---|---|
| n-butyl phosphoric acid In acetone at 65℃; for 16h; | 93% |
| Stage #1: isobutyl vinyl ether; Thiocyanuric acid; n-butyl phosphoric acid In acetone at 65℃; for 16h; Stage #2: With tetra(2-ethylhexyl)titanate In acetone | 93% |
| Stage #1: isobutyl vinyl ether; Thiocyanuric acid; n-butyl phosphoric acid In acetone at 65℃; for 16h; Stage #2: With hydrotalcite In acetone at 40℃; for 10h; | 84% |
-
-
109-53-5
isobutyl vinyl ether

-
-
1332581-01-7
bis(2-isobutoxy-2-bromoethyl) selenide

| Conditions | Yield |
|---|---|
| With selenium dibromide In tetrachloromethane at -20 - 20℃; regioselective reaction; | 93% |
Isobutyl vinyl ether Chemical Properties
IUPAC Name: 1-ethenoxy-2-methylpropane
Empirical Formula: C6H12O
Molecular Weight: 100.1589g/mol
Structure of Propane,1-(ethenyloxy)-2-methyl- (CAS NO.109-53-5):
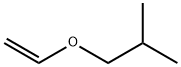
EINECS: 203-678-8
Index of Refraction: 1.398
Molar Refractivity: 31.27 cm3
Molar Volume: 129.4 cm3
Polarizability: 12.39×10-24 cm3
Surface Tension: 20.9 dyne/cm
Density: 0.773 g/cm3
vapor density 3.45 (vs air)
Melting point: -112 °C
Boiling Point: 83 °C at 760 mmHg
Enthalpy of Vaporization: 31.01 kJ/mol
Vapour Pressure: 85.6 mmHg at 25°C
storage temp.: Flammables area
Water Solubility: 0.7 g/L (25 oC)
Canonical SMILES: CC(C)COC=C
InChI: InChI=1S/C6H12O/c1-4-7-5-6(2)3/h4,6H,1,5H2,2-3H3
InChIKey: OZCMOJQQLBXBKI-UHFFFAOYSA-N
Isobutyl vinyl ether Toxicity Data With Reference
| 1. | orl-rat LD50:17 g/kg | AIHAAP American Industrial Hygiene Association Journal. 23 (1962),95. | ||
| 2. | ihl-rat LCLo:16,000 ppm/4H | AIHAAP American Industrial Hygiene Association Journal. 23 (1962),95. | ||
| 3. | skn-rbt LD50:20 g/kg | AIHAAP American Industrial Hygiene Association Journal. 23 (1962),95. |
Isobutyl vinyl ether Consensus Reports
Reported in EPA TSCA Inventory.
Isobutyl vinyl ether Safety Profile
Very mildly toxic by ingestion, inhalation, and skin contact. A very dangerous fire hazard when exposed to heat, flame, oxidizers. Severe explosion hazard when exposed to sparks or open flame. Can react vigorously with oxidizing materials. When heated to decomposition it emits acrid smoke and fumes. To fight fire, use alcohol foam, CO2, dry chemical. See also ETHERS.
Hazard Codes:  F,
F, Xi
Xi
Risk Statements: 11-38
R11:Highly flammable.
R38:Irritating to skin.
Safety Statements: 16-23-24/25-33-37
S16:Keep away from sources of ignition.
S23:Do not breathe vapour.
S24/25:Avoid contact with skin and eyes.
S33:Take precautionary measures against static discharges.
S37:Wear suitable gloves.
RIDADR: UN 1304 3/PG 2
WGK Germany: 1
RTECS: KO1300000
F: 9
HazardClass: 3.1
PackingGroup: II
Isobutyl vinyl ether Standards and Recommendations
DOT Classification: 3; Label: Flammable Liquid
Isobutyl vinyl ether Specification
Propane,1-(ethenyloxy)-2-methyl- , its cas register number is 64058-30-6. It also can be called 2-Methyl-1-vinyloxypropane ; 4-01-00-02054 (Beilstein Handbook Reference) ; AI3-18425 ; IVE ; Isobutanol vinyl ether ; Isobutoxyethene ; Isobutyl vinyl ether ; Lutanol LR 8500 ; NSC 8265 ; Vinyl isobutyl ether .
Propane,1-(ethenyloxy)-2-methyl- (CAS NO.109-53-5) is a clear colourless liquid.
Related Products
- Isobutyl 1,2,3,4,5,6,7,8-octahydro-2,8,8-trimethyl-2-naphthoate
- Isobutyl 1,2-dihydro-2-isobutoxy-1-quinoline-carboxylate
- Isobutyl 2,2-dimethyl-5-(2,5-xylyloxy)valerate
- Isobutyl 2,4-dichlorophenoxyacetate
- Isobutyl 2-butenoate
- Isobutyl 3,5-diamino-4-chloro benzoate
- Isobutyl 4-hydroxybenzoate
- Isobutyl 5-chloro-2,2-dimethylvalerate
- Isobutyl acetate
- Isobutyl acetoacetate
- 1095382-05-0
- 109543-53-5
- 109545-01-9
- 109545-84-8
- 109-54-6
- 109552-15-0
- 109555-87-5
- 1095565-81-3
- 1095565-87-9
- 109-55-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View