-
Name
Methacrylic anhydride
- EINECS 212-084-8
- CAS No. 760-93-0
- Article Data46
- CAS DataBase
- Density 1.021 g/cm3
- Solubility 98g/L at 20℃
- Melting Point -20 °C
- Formula C8H10O3
- Boiling Point 202.3 °C at 760 mmHg
- Molecular Weight 154.166
- Flash Point 84.4 °C
- Transport Information UN 3265 8/PG 2
- Appearance liquid
- Safety 26-39
- Risk Codes 20-37/38-41
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 2-Propenoicacid, 2-methyl-, anhydride (9CI);Methacrylic anhydride (6CI,8CI);Methacrylicacid anhydride;Methacryloyl anhydride;NSC 24159;
- PSA 43.37000
- LogP 1.20840
Synthetic route

| Conditions | Yield |
|---|---|
| With N,N-bis[2-oxo-3-oxazolidinyl]phosphorodiamidic chloride; triethylamine In dichloromethane at 20℃; for 0.5h; | 99% |
| With magnesium dimethacrylate; di-tert-butyl dicarbonate at 25℃; for 4h; Reagent/catalyst; | 99% |
| With 10H-phenothiazine; trifluorormethanesulfonic acid; acetic anhydride at 95℃; Reagent/catalyst; Temperature; | 95% |

| Conditions | Yield |
|---|---|
| With pyridine; 1,1,1-trichloro-3,3,3-trifluoro-propan-2-one; water In toluene for 0.5h; Ambient temperature; | 93% |
| With sodium hydroxide; tetrabutylammomium bromide In dichloromethane at -10℃; for 3h; | 80% |
| With sodium hydroxide; tetrabutyl-ammonium chloride In toluene at -10℃; for 3h; | 7% |

| Conditions | Yield |
|---|---|
| With triethylamine In benzene at 70℃; | 92% |
| Stage #1: Methacryloyl chloride With pyridine; hydroquinone In toluene Stage #2: poly(methacrylic acid) In toluene at 50 - 65℃; | 83% |
| With triethylamine In benzene | |
| With sodium hydroxide In water | |
| With sodium hydroxide In water |
-
-
5536-61-8
sodium methacrylate

-
-
541-41-3
chloroformic acid ethyl ester

-
A
-
760-93-0
methacryloyl anhydride

-
B
-
21982-91-2
carboethoxymethacrylic anhydride

| Conditions | Yield |
|---|---|
| With 4-methoxy-phenol; tetrabutylammomium bromide In H2O (water) < 309.3g; toluene 192g at 15℃; for 4h; | A n/a B 87.5% |
| With 4-methoxy-phenol In water at 5 - 35℃; for 7 - 30h; | |
| tributylphenylphosphonium chloride at 10℃; for 3h; |

-
-
5536-61-8
sodium methacrylate

-
-
541-41-3
chloroformic acid ethyl ester

-
A
-
760-93-0
methacryloyl anhydride

-
B
-
21982-91-2
carboethoxymethacrylic anhydride

-
C
-
105-58-8
Diethyl carbonate

| Conditions | Yield |
|---|---|
| With 4-methoxy-phenol; tetrabutylammomium bromide In water at 20℃; for 3h; | A n/a B 80% C n/a |
| tetrabutyl phosphonium bromide at 10℃; for 4.5h; | |
| methyl(tri-n-octyl)ammonium bromide at 10℃; for 1.5h; |

-
-
702-98-7
2-methyladamantan-2-ol

-
A
-
177080-67-0
2-methyl-2-adamantyl methacrylate

-
B
-
760-93-0
methacryloyl anhydride

| Conditions | Yield |
|---|---|
| A 40% B n/a |

| Conditions | Yield |
|---|---|
| With oxygen; silica gel; Te/Pd-containing-silica-supported catalyst In water; tert-butyl alcohol at 75℃; under 18001.8 - 36003.6 Torr; for 0.75h; Product distribution / selectivity; Autoclave; | A 34.2% B 17.5% |

| Conditions | Yield |
|---|---|
| With copper (I) acetate |
-
-
79-41-4
poly(methacrylic acid)

-
-
98-88-4
benzoyl chloride

-
A
-
760-93-0
methacryloyl anhydride

-
B
-
46318-87-0
benzoyl (meth)acrylate

-
C
-
93-97-0
benzoic acid anhydride

| Conditions | Yield |
|---|---|
| With P4-VP In dichloromethane at 0℃; for 1h; Yield given. Yields of byproduct given; |


-
-
760-93-0
methacryloyl anhydride

| Conditions | Yield |
|---|---|
| With thionyl chloride; Petroleum ether |
-
-
541-41-3
chloroformic acid ethyl ester

-
-
6900-35-2
potassium methacrylate

-
A
-
760-93-0
methacryloyl anhydride

-
B
-
21982-91-2
carboethoxymethacrylic anhydride

-
C
-
105-58-8
Diethyl carbonate

| Conditions | Yield |
|---|---|
| at 10℃; |

-
-
5536-61-8
sodium methacrylate

-
-
541-41-3
chloroformic acid ethyl ester

-
A
-
760-93-0
methacryloyl anhydride

-
B
-
1609-47-8
diethyl dicarbonate

-
C
-
21982-91-2
carboethoxymethacrylic anhydride

-
D
-
105-58-8
Diethyl carbonate


| Conditions | Yield |
|---|---|
| With phosphoric acid; acetic anhydride; sodium hydrogencarbonate In hexane; water |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; phosphoric acid; acetic anhydride In hexane; water | |
| With sodium hydroxide; phosphoric acid; acetic anhydride In hexane; water |
-
-
115-11-7
isobutene

-
A
-
78-85-3
2-methylpropenal

-
B
-
79-41-4
poly(methacrylic acid)

-
C
-
760-93-0
methacryloyl anhydride

| Conditions | Yield |
|---|---|
| With oxygen; 4-methoxy-phenol; palladium matal-containing silica-supported catalyst In water; tert-butyl alcohol at 90℃; for 6h; Product distribution / selectivity; | |
| With oxygen at 110℃; under 36003.6 Torr; Gas phase; | A 27.8 %Chromat. B 53 %Chromat. C 7.4 %Chromat. |


| Conditions | Yield |
|---|---|
| With TEMPOL; 2,6-di-tert-butyl-4-methyl-phenol; 2,4-dimethyl-6-tert-butylphenol at 93℃; under 18 - 140 Torr; Product distribution / selectivity; Air atmosphere; | |
| With 10H-phenothiazine at 80 - 90℃; under 760.051 Torr; | |
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; 10H-phenothiazine at 100 - 105℃; for 2h; | 135.5 g |
-
-
79-41-4
poly(methacrylic acid)

-
-
108-24-7
acetic anhydride

-
A
-
760-93-0
methacryloyl anhydride

-
B
-
76392-13-7
methacrylic acid acetate

-
C
-
64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| acidic ion exchanger Lewatit K2431 at 76℃; Polymerization inhibitior; |

-
-
79-41-4
poly(methacrylic acid)

-
-
76392-13-7
methacrylic acid acetate

-
-
108-24-7
acetic anhydride

-
A
-
760-93-0
methacryloyl anhydride

-
B
-
64-19-7
acetic acid

| Conditions | Yield |
|---|---|
| acidic ion exchanger Lewatit K2431 at 76℃; Polymerization inhibitior; |

-
-
67-56-1
methanol

-
-
78-85-3
2-methylpropenal

-
A
-
5634-54-8
1,1-dimethoxy-2-methylprop-1-ene

-
B
-
187737-37-7
propene

-
C
-
79-41-4
poly(methacrylic acid)

-
D
-
547-63-7
Methyl isobutyrate

-
E
-
97-86-9
Isobutyl methacrylate

-
F
-
760-93-0
methacryloyl anhydride

-
G
-
78-84-2
isobutyraldehyde

-
H
-
80-62-6
methacrylic acid methyl ester

-
I
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With magnesium hydroxide; oxygen at 80℃; under 2250.23 Torr; for 2h; Reagent/catalyst; |


| Conditions | Yield |
|---|---|
| With pyridine; hydroquinone at 20℃; | 100% |
-
-
760-93-0
methacryloyl anhydride

-
-
17728-11-9
1-amino-4-hydroxy-2-[2'-[4'-(2'-hydroxyethoxy)phenoxy]ethoxy]anthraquinone

| Conditions | Yield |
|---|---|
| Stage #1: methacryloyl anhydride; 1-amino-4-hydroxy-2-[2'-[4'-(2'-hydroxyethoxy)phenoxy]ethoxy]anthraquinone With triethylamine; dmap; hydroquinone In DMF (N,N-dimethyl-formamide) at 20℃; for 24h; Stage #2: Heating / reflux; | 100% |

| Conditions | Yield |
|---|---|
| With pyridine at 80℃; for 6.5h; | 100% |

| Conditions | Yield |
|---|---|
| With dmap; 2,6-di-tert-butyl-4-methyl-phenol In toluene at 80℃; for 40h; | 100% |
-
-
54731-72-5
dimethyl 2-hydroxyethylphosphonate

-
-
760-93-0
methacryloyl anhydride

-
-
22432-83-3
dimethyl (2-methacryloyloxyethyl)phosphonate

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole In butanone at 60℃; for 6h; Inert atmosphere; | 100% |
-
-
760-93-0
methacryloyl anhydride

-
-
54731-74-7
dimethyl (γ-hydroxypropyl)phosphonate

-
-
252210-28-9
dimethyl (3-methacryloyloxypropyl)phosphonate

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole In butanone at 60℃; for 6h; Inert atmosphere; | 100% |
-
-
760-93-0
methacryloyl anhydride

-
-
131991-61-2
6A-(6-aminohexylamino)-6A-deoxy-β-cyclodextrin

-
-
1080030-24-5
(1-methacrylamidohexyl)amino-6-deoxy-β-cyclodextrin

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; Inert atmosphere; | 100% |
-
-
19700-96-0, 19700-97-1, 58208-04-1, 112571-38-7, 144829-86-7, 1069-23-4
hexa-1,5-diene-3,4-diol

-
-
760-93-0
methacryloyl anhydride

-
-
1361054-19-4
4-[(2-methylprop-2-enoyl)oxy]hexa-1,5-dien-3-yl 2-methylprop-2-enoate

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; for 1.5h; | 100% |

| Conditions | Yield |
|---|---|
| With triphenyl phosphite; 2,6-di-tert-butyl-4-methyl-phenol; magnesium oxide In toluene at 80℃; for 3h; Reagent/catalyst; | 100% |
| With magnesium oxide In toluene at 80℃; for 3h; Catalytic behavior; Concentration; Reagent/catalyst; Temperature; | 100% |
-
-
760-93-0
methacryloyl anhydride

-
-
1137-42-4
4-Hydroxybenzophenone

-
-
56467-43-7
4-hydroxybenzophenone ester of metacrylic acid

| Conditions | Yield |
|---|---|
| With 2,4-dimethyl-6-tert-butylphenol; sulfuric acid; 4-methoxy-phenol at 90℃; for 6h; Air introduction; | 99.4% |
| With sulfuric acid In methanol at 90℃; for 2h; Temperature; Large scale; | 99.7% |
| With tempol; 2,4-dimethyl-6-tert-butylphenol; 4-methoxy-phenol; magnesium bromide at 90℃; for 6h; Reagent/catalyst; |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 80℃; for 8h; Product distribution / selectivity; | 99.1% |
| at 25℃; for 24h; | 90% |
| With sulfuric acid at 80℃; for 8h; Product distribution / selectivity; Industry scale; | |
| at 120℃; for 5h; Product distribution / selectivity; | |
| With magnesium monohydroxide; di-tert-butyl dicarbonate; lithium chloride at 25℃; under 760.051 Torr; for 24h; Time; |

| Conditions | Yield |
|---|---|
| Stage #1: inulin, MW: ca. 5000 Da, isolated from Dahlia Tubers; methacryloyl anhydride; triethylamine In N,N-dimethyl-formamide at 25℃; for 24h; Stage #2: succinic acid anhydride; triethylamine In N,N-dimethyl-formamide at 25℃; for 24h; Further stages.; | 99% |

-
-
760-93-0
methacryloyl anhydride

-
-
1664-40-0
N-(2-aminoethyl)benzeneamine

-
-
1149340-19-1
N-phenylethylenediamine methacrylamide

| Conditions | Yield |
|---|---|
| In methanol at 0℃; for 3h; | 99% |

| Conditions | Yield |
|---|---|
| In methanol at 15 - 25℃; for 2h; Solvent; | 99% |

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 35℃; Flow reactor; | 99% |

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 0 - 20℃; for 18h; | 98.2% |
| With dmap; triethylamine In dichloromethane at 0℃; Inert atmosphere; Cooling with ice; | 90.8% |

-
-
760-93-0
methacryloyl anhydride

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl acetamide at 40℃; for 48h; | 98% |
-
-
760-93-0
methacryloyl anhydride

-
-
7321-13-3
1,4-bis-[(2-hydroxy-ethylsulfanyl)-methyl]-benzene

-
-
112503-98-7
1,4-bis(2-methacryloyloxyethylthio)xylylene

| Conditions | Yield |
|---|---|
| Stage #1: methacryloyl anhydride; 1,4-bis-[(2-hydroxy-ethylsulfanyl)-methyl]-benzene With triethylamine In dichloromethane at 20℃; for 48h; Stage #2: With sodium hydrogencarbonate In dichloromethane; water for 0.5h; | 98% |
-
-
760-93-0
methacryloyl anhydride

-
-
117420-69-6
1,2-bis(hydroxyethylthiomethyl)benzene

-
-
154865-01-7
1,2-bis(methacryloyloxyethylthiomethyl)benzene

| Conditions | Yield |
|---|---|
| Stage #1: methacryloyl anhydride; 1,2-bis(hydroxyethylthiomethyl)benzene With triethylamine In dichloromethane at 20℃; for 48h; Stage #2: With sodium hydrogencarbonate In dichloromethane; water for 0.5h; | 98% |

| Conditions | Yield |
|---|---|
| With dmap at 60℃; | 98% |

| Conditions | Yield |
|---|---|
| With lithium carbonate at 80℃; for 2h; Reagent/catalyst; | 98% |
-
-
760-93-0
methacryloyl anhydride

-
-
135304-82-4
1,3,6-trimethyl-isobarbituric acid

| Conditions | Yield |
|---|---|
| With pyridine; hydroquinone at 20℃; for 1h; | 97% |
-
-
79-41-4
poly(methacrylic acid)

-
-
760-93-0
methacryloyl anhydride

-
-
123-31-9
hydroquinone

-
A
-
5363-70-2
hex-1-ene-2,5-dicarboxylic acid

-
B
-
31480-93-0
4-hydroxyphenyl methacrylate

| Conditions | Yield |
|---|---|
| at 120℃; for 4h; Product distribution / selectivity; | A 1% B 97% |

-
-
700-58-3
2-Adamantanone

-
-
760-93-0
methacryloyl anhydride

-
-
917-54-4
methyllithium

-
-
177080-67-0
2-methyl-2-adamantyl methacrylate

| Conditions | Yield |
|---|---|
| Stage #1: 2-Adamantanone; methyllithium In tetrahydrofuran; diethyl ether at -25 - -20℃; for 1h; Stage #2: methacryloyl anhydride With triethylamine In tetrahydrofuran; diethyl ether at 25℃; for 1.5h; | 97% |
| Stage #1: 2-Adamantanone; methyllithium In tetrahydrofuran; diethyl ether at -25 - -20℃; for 1h; Stage #2: methacryloyl anhydride In tetrahydrofuran; diethyl ether at 55℃; for 3h; | 90% |

-
-
760-93-0
methacryloyl anhydride

-
-
491878-98-9
bis(O,S-triphenylsilyl)-2-mercaptoethanol

-
-
1113062-54-6
C24H24O2SSi

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 3h; | 97% |
-
-
760-93-0
methacryloyl anhydride

-
-
94594-90-8
(2R)-bornane-10,2-sultam

-
-
116195-15-4
(3aS,6R,7aR)-1-[(2E)-but-2-enoyl]-8,8-dimethylhexahydro-3a,6-methano-2,1-benzisothiazole 2,2-dioxide

| Conditions | Yield |
|---|---|
| Stage #1: (2R)-bornane-10,2-sultam With triethylamine; lithium chloride In tetrahydrofuran at -20℃; for 0.166667h; Stage #2: methacryloyl anhydride In tetrahydrofuran at -20 - -10℃; for 0.0833333h; Product distribution / selectivity; | 96% |
| With triethylamine; lithium chloride In tetrahydrofuran for 4h; Ambient temperature; | 95% |

-
-
760-93-0
methacryloyl anhydride

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 20℃; | 96% |
| With dmap In dichloromethane at 20℃; | 96% |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; for 12h; | 96% |

| Conditions | Yield |
|---|---|
| With dmap at 60℃; | 96% |
Methacrylic anhydride Chemical Properties
Product Name: Methacrylic anhydride (CAS NO.760-93-0)
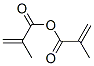
MF: C8H10O3
MW: 154.16
Mol File: 760-93-0.mol
EINECS: 212-084-8
Surface Tension: 29 dyne/cm
Density: 1.021 g/ml
Flash Point: 84.4 °C
MP: -20°C
Enthalpy of Vaporization: 43.85 kJ/mol
Boiling Point: 202.3 °C at 760 mmHg
Vapour Pressure: 0.294 mmHg at 25°C
BRN: 1761982
Index of Refraction: 1.443
Molar Refractivity: 40.05 cm3
Molar Volume: 150.9 cm3
XLogP3-AA: 1.8
H-Bond Donor: 0
H-Bond Acceptor: 3
Structure Descriptors of Methacrylic anhydride (CAS NO.760-93-0):
IUPAC Name: 2-methylprop-2-enoyl 2-methylprop-2-enoate
Canonical SMILES: CC(=C)C(=O)OC(=O)C(=C)C
InChI: InChI=1S/C8H10O3/c1-5(2)7(9)11-8(10)6(3)4/h1,3H2,2,4H3
InChIKey: DCUFMVPCXCSVNP-UHFFFAOYSA-N
Product Categories: Organic acids; Carbonyl Compounds; Carboxylic Acid Anhydrides; Organic Building Blocks; Anhydride Monomers; Monomers; Polymer Science
Methacrylic anhydride Uses
Methacrylic anhydride (CAS NO.760-93-0) is used as light-curing coatings, cross-linked materials such as synthetic resin.
Methacrylic anhydride Toxicity Data With Reference
| 1. | ihl-mus LC50:450 mg/m3/2H | 85GMAT Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure Izmerov, N.F., et al.,Moscow, USSR.: Centre of International Projects, GKNT,1982,80. |
Methacrylic anhydride Consensus Reports
EPA Extremely Hazardous Substances List. Reported in EPA TSCA Inventory.
Methacrylic anhydride Safety Profile
Poison by inhalation. A combustible liquid. When heated to decomposition it emits acrid smoke and irritating fumes. See also ANHYDRIDES.
Safety information of Methacrylic anhydride (CAS NO.760-93-0):
Hazard Codes:  Xn
Xn
Risk Statements: 20-37/38-41
R20:Harmful by inhalation ;
R37/38:Irritating to respiratory system and skin ;
R41:Risk of serious damage to eyes ;
Safety Statements: 26-39
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice ;
S39:Wear eye/face protection ;
RIDADR: UN 3265 8/PG 2
WGK Germany: 1
RTECS: OZ5700000
F: 10-21
HazardClass: 8
PackingGroup: II
Methacrylic anhydride Specification
Methacrylic anhydride , its CAS NO. is 760-93-0, the synonyms are 1-Methylacrylic anhydride ; 2-Propenoicacid,2-methyl-,anhydride ; 2-Methyl-2-propenoicacianhydride ; Methacrylic acid anhydride ; Methacryloyl anhydride ; Methacrylicacidanhydride ; Methacryloylanhydride . Methacrylic anhydride (CAS NO.760-93-0) reacts exothermically with water. The reactions are sometimes slow, but can become violent when local heating accelerates their rate. Acids accelerate the reaction with water. Incompatible with acids, strong oxidizing agents, alcohols, amines, and bases.
Related Products
- Methacrylic acid
- Methacrylic acid, butyl ester, polymer with 2-(dimethylamino)ethyl meth-acrylate and methyl methacrylate
- Methacrylic acid, polymer with methy acrylate
- Methacrylic anhydride
- 760937-92-6
- 76093-98-6
- 76-09-5
- 76095-16-4
- 760958-13-2
- 76098-97-0
- 760990-15-6
- 76099-05-3
- 76099-38-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View