-
Name
Methyl carbazate
- EINECS 228-560-3
- CAS No. 6294-89-9
- Article Data24
- CAS DataBase
- Density 1.162 g/cm3
- Solubility Soluble in water
- Melting Point 70-73 °C(lit.)
- Formula C2H6N2O2
- Boiling Point 230.906 °C at 760 mmHg
- Molecular Weight 90.0818
- Flash Point 86.129 °C
- Transport Information UN 2811
- Appearance white to pink crystalline platelets
- Safety 26-36/37/39-22-45
- Risk Codes 36/37/38-48-23/25
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi,  T
T
- Synonyms Carbazicacid, methyl ester (8CI);(Methoxycarbonyl)hydrazide;Carbomethoxyhydrazide;Methyl hydrazinecarboxylate;Methyl hydrazinoformate;
- PSA 64.35000
- LogP 0.30730
Synthetic route
-
-
7803-57-8
hydrazine hydrate

-
-
616-38-6
carbonic acid dimethyl ester

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| at 20℃; | 96% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate at 20 - 50℃; for 20.33h; | 95% |
| With hydrazine hydrate | 95% |
| With hydrazine hydrate at 20 - 50℃; for 24.5h; | 94% |
-
-
616-38-6
carbonic acid dimethyl ester

-
A
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
B
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In neat (no solvent) at 80℃; Temperature; | A 53% B 35% |
-
-
14442-53-6
N-nitro-carbamide acid methylester

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With acetic acid Electrolysis.durch Reduktion an einer verzinnten Kupferkathode; |
-
-
14442-53-6
N-nitro-carbamide acid methylester

-
-
64-19-7
acetic acid

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| Reduktion an einer verzinnten Kupferkathode.Electrolysis; |

| Conditions | Yield |
|---|---|
| With methanol; hydrazine hydrate |
-
-
76529-39-0
methyl (phenylseleno)formate

-
A
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
B
-
1666-13-3
diphenyl diselenide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol at -10 - 20℃; |

-
A
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
B
-
22478-90-6
3-benzyl-4-phenyl-5-mercapto-1,2,4-triazole

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol; water at 20 - 60℃; for 10h; |
-
-
61985-23-7
methyl 1-imidazolecarboxylate

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In dichloromethane at 0 - 20℃; for 1h; |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
534-07-6
1,3-Dichloroacetone

-
-
87595-89-9
methyl 2-[2-chloro-1-(chloromethyl)ethylidene]-1-hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| In diethyl ether for 15h; Ambient temperature; | 100% |
| In methanol at 23℃; for 4h; | 54% |
| for 3h; Ambient temperature; Yield given; | |
| In methanol at 23℃; for 4h; |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
298-12-4
Glyoxilic acid

-
-
332868-35-6
[(methoxycarbonyl)hydrazono]acetic acid

| Conditions | Yield |
|---|---|
| In water for 7h; | 100% |

| Conditions | Yield |
|---|---|
| With acetic acid In methanol for 1h; Reflux; | 100% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
67-64-1
acetone

-
-
21732-11-6
methyl-2,3-diaza-4-methyl-4-acetoxypent-2-enoate

| Conditions | Yield |
|---|---|
| With magnesium sulfate for 6.5h; Reflux; Inert atmosphere; | 100% |
| With magnesium sulfate In acetone for 2h; Inert atmosphere; Reflux; | 87.8% |
| With acetic acid In methanol at 25 - 65℃; Molecular sieve; | |
| With acetic acid at 20℃; for 24h; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethyl acetate at 50℃; for 16h; Reagent/catalyst; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: chloroacetonitrile With sodium In methanol at 0 - 20℃; for 0.5h; Stage #2: hydrazinecarboxylic acid methyl ester With acetic acid In methanol at 20℃; for 0.666667h; | 99.4% |
| Stage #1: chloroacetonitrile With sodium methylate In methanol at 0 - 20℃; for 0.75h; Stage #2: hydrazinecarboxylic acid methyl ester With acetic acid In methanol at 20℃; for 0.5h; |
-
-
7803-57-8
hydrazine hydrate

-
-
616-38-6
carbonic acid dimethyl ester

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| at 20℃; | 96% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate at 20 - 50℃; for 20.33h; | 95% |
| With hydrazine hydrate | 95% |
| With hydrazine hydrate at 20 - 50℃; for 24.5h; | 94% |
-
-
616-38-6
carbonic acid dimethyl ester

-
A
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
B
-
497-18-7
carbonodihydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In neat (no solvent) at 80℃; Temperature; | A 53% B 35% |
-
-
14442-53-6
N-nitro-carbamide acid methylester

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With acetic acid Electrolysis.durch Reduktion an einer verzinnten Kupferkathode; |
-
-
14442-53-6
N-nitro-carbamide acid methylester

-
-
64-19-7
acetic acid

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| Reduktion an einer verzinnten Kupferkathode.Electrolysis; |

| Conditions | Yield |
|---|---|
| With methanol; hydrazine hydrate |
-
-
76529-39-0
methyl (phenylseleno)formate

-
A
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
B
-
1666-13-3
diphenyl diselenide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol at -10 - 20℃; |

-
A
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
B
-
22478-90-6
3-benzyl-4-phenyl-5-mercapto-1,2,4-triazole

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol; water at 20 - 60℃; for 10h; |
-
-
61985-23-7
methyl 1-imidazolecarboxylate

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In dichloromethane at 0 - 20℃; for 1h; |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
534-07-6
1,3-Dichloroacetone

-
-
87595-89-9
methyl 2-[2-chloro-1-(chloromethyl)ethylidene]-1-hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| In diethyl ether for 15h; Ambient temperature; | 100% |
| In methanol at 23℃; for 4h; | 54% |
| for 3h; Ambient temperature; Yield given; | |
| In methanol at 23℃; for 4h; |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
298-12-4
Glyoxilic acid

-
-
332868-35-6
[(methoxycarbonyl)hydrazono]acetic acid

| Conditions | Yield |
|---|---|
| In water for 7h; | 100% |

| Conditions | Yield |
|---|---|
| With acetic acid In methanol for 1h; Reflux; | 100% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
67-64-1
acetone

-
-
21732-11-6
methyl-2,3-diaza-4-methyl-4-acetoxypent-2-enoate

| Conditions | Yield |
|---|---|
| With magnesium sulfate for 6.5h; Reflux; Inert atmosphere; | 100% |
| With magnesium sulfate In acetone for 2h; Inert atmosphere; Reflux; | 87.8% |
| With acetic acid In methanol at 25 - 65℃; Molecular sieve; | |
| With acetic acid at 20℃; for 24h; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethyl acetate at 50℃; for 16h; Reagent/catalyst; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: chloroacetonitrile With sodium In methanol at 0 - 20℃; for 0.5h; Stage #2: hydrazinecarboxylic acid methyl ester With acetic acid In methanol at 20℃; for 0.666667h; | 99.4% |
| Stage #1: chloroacetonitrile With sodium methylate In methanol at 0 - 20℃; for 0.75h; Stage #2: hydrazinecarboxylic acid methyl ester With acetic acid In methanol at 20℃; for 0.5h; |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
141-97-9
ethyl acetoacetate

-
-
356102-30-2
3-methoxycarbonylhydrazonoacetic acid ethyl ester

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 10.5h; | 99% |
| In ethanol at 20℃; for 10.5h; | 99% |
| In ethanol at 20℃; | 98% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
58085-33-9, 60135-02-6, 51388-67-1
α-phenylcinnamoyl chloride

-
-
1194791-09-7
methyl 2-[(2E)-(2,3-diphenyl)prop-2-enoyl]hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; | 99% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
35086-79-4, 95602-71-4
cinnamoyl chloride

-
-
1194791-02-0
methyl 2-[(2E)-3-(4-chlorophenyl)prop-2-enoyl]hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; | 99% |
-
-
50-00-0
formaldehyd

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
99132-70-4
N'-Methylene-hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In methanol for 2h; Heating; | 98% |

| Conditions | Yield |
|---|---|
| In benzene for 0.333333h; | 98% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
102252-89-1
3-amino-1-(4-methoxyphenyl)but-2-en-1-one

-
-
254734-22-0
methyl 4,5-dihydro-5-hydroxy-5-(4-methoxyphenyl)-3-methylpyrazole-1-carboxylate

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 20℃; for 24h; Cycloaddition; | 98% |
-
-
31021-02-0
2-phenylmethylenecyclohexan-1-one

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| In ethanol | 98% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
28424-61-5, 73186-06-8, 119924-24-2
(2E)-3-(2-thiophenyl)-2-propenoyl chloride

-
-
1194790-95-8
methyl 2-[(2E)-3-(2-thienyl)prop-2-enoyl]hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; | 98% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
53901-95-4, 108608-06-6, 58523-18-5
p-acetoxycinnamoyl chloride

-
-
1194790-88-9
methyl 2-[(2E)-3-(4-acetoxyphenyl)prop-2-enoyl]hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; | 98% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| In butan-1-ol at 130℃; for 48h; | 98% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
121557-79-7
Ethyl <<2-(4-t-Butylphenyl)-1-thioxoethyl>thio>acetate

-
-
114373-67-0
2-[2-[4-(1,1-dimethylethyl)phenyl]-1-thioxoethyl]hydrazinecarboxylic acid, methyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane for 2h; Heating; | 97% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
108-94-1
cyclohexanone

-
-
14702-42-2
methoxycarbonylhydrazone of cyclohexanone

| Conditions | Yield |
|---|---|
| With acetic acid In methanol for 0.5h; Heating; | 97% |
| 91% | |
| With acetic acid In methanol for 6h; Heating; Yield given; | |
| With acetic acid In methanol at 25 - 65℃; Molecular sieve; |
-
-
1118-66-7
4-amino-3-penten-2-one

-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
53921-04-3
methyl 3,5-dimethyl-1H-pyrazole-1-carboxylate

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 20℃; for 110h; Cyclization; condensation; | 97% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
102252-95-9
3-amino-1-(4-nitrophenyl)-2-buten-1-one

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 20℃; for 24h; Dehydration; condensation; | 97% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
169559-48-2
[perfluoro(sulfanyl)vinyl]-diiron complex

-
-
436162-67-3
[(Fe(CO)3)2( μ-S(CH3)C(CF3)C β (NHNHC(O)OMe) C α (NHNHC(O)OMe))]

| Conditions | Yield |
|---|---|
| In dichloromethane room temp., 2 hs, (Ar); filtration (deoxygenated silica gel), vac. evapn., crystn. (pentane), elem. anal.; | 97% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
119924-22-0
(E)-3-(thien-3-yl)acryloyl chloride

-
-
1194790-97-0
methyl 2-[(2E)-3-(2-thienyl)prop-2-enoyl]hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; | 97% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
121557-83-3
Ethyl <<2-(3-Methoxyphenyl)-1-thioxoethyl>thio>acetate

-
-
114373-92-1
2-[2-(3-methoxyphenyl)-1-thioxoethyl]hydrazinecarboxylic acid, methyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane for 2h; Heating; | 96% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
114373-99-8
[[1-thioxo-2-[3-(trifluoromethyl)phenyl]ethyl]thio]acetic acid, ethyl ester

-
-
114373-96-5
2-[1-thioxo-2-[3-(trifluoromethyl)phenyl]ethyl]hydrazinecarboxylic acid, methyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane for 2h; Heating; | 96% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
93222-85-6
2-<(nitrooxy)methyl>quinoxaline 1,4-dioxide

-
-
6804-07-5
carbadox

| Conditions | Yield |
|---|---|
| In dichloromethane | 96% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
67130-27-2
ethyl-2 benzothiazine-3,1 thione-4

-
-
83389-34-8
(2-Ethyl-4-thioxo-4H-quinazolin-3-yl)-carbamic acid methyl ester

| Conditions | Yield |
|---|---|
| In ethanol; benzene for 4h; Heating; | 96% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
23652-90-6, 80012-15-3, 1128-85-4
3-amino-1-phenylbut-2-en-1-one

-
-
254734-23-1
methyl 4,5-dihydro-5-hydroxy-3-methyl-5-phenylpyrazole-1-carboxylate

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 20℃; for 18h; Cyclization; condensation; | 96% |
-
-
6294-89-9
hydrazinecarboxylic acid methyl ester

-
-
35086-87-4, 38449-13-7
(E)-2-methyl-3-phenylacryloyl chloride

-
-
1194791-06-4
methyl 2-[(2E)-(2-methyl-3-phenyl)prop-2-enoyl]hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; | 96% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol; water for 0.5h; Reflux; | 96% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol Reflux; | 96% |
Methyl carbazate Consensus Reports
Methyl carbazate is reported in EPA TSCA Inventory.
Methyl carbazate Specification
The Methyl carbazate, with the CAS registry number 6294-89-9, is also known as Methoxycarbonylhydrazine. It belongs to the product categories of Amines; Isotope Labelled Compounds. Its EINECS number is 228-560-3. This chemical's molecular formula is C2H6N2O2 and molecular weight is 90.08. What's more, its systematic name is Methyl hydrazinecarboxylate.
Physical properties of Methyl carbazate are: (1)ACD/LogP: -0.719; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.72; (4)ACD/LogD (pH 7.4): -0.72; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 9.64; (8)ACD/KOC (pH 7.4): 9.67; (9)#H bond acceptors: 4; (10)#H bond donors: 3; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 64.35 Å2; (13)Index of Refraction: 1.435; (14)Molar Refractivity: 20.22 cm3; (15)Molar Volume: 77.525 cm3; (16)Polarizability: 8.016×10-24cm3; (17)Surface Tension: 38.8 dyne/cm; (18)Density: 1.162 g/cm3; (19)Flash Point: 86.129 °C; (20)Enthalpy of Vaporization: 46.755 kJ/mol; (21)Boiling Point: 230.906 °C at 760 mmHg; (22)Vapour Pressure: 0.06 mmHg at 25°C.
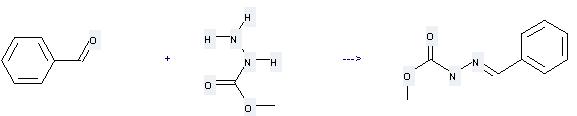
Uses of Methyl carbazate: it can be used to produce benzylidene-carbazic acid methyl ester by heating. It will need reagent acetic acid and solvent methanol with the reaction time of 30 min. The yield is about 90%.
When you are using this chemical, please be cautious about it as the following:
This chemical is irritating to eyes, respiratory system and skin. It it toxic by inhalation and if swallowed. It has a danger of serious damage to health by prolonged exposure. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing, gloves and eye/face protection. You should not breathe dust. In case of accident or if you feel unwell, you must seek medical advice immediately (show the label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(OC)NN
(2)Std. InChI: InChI=1S/C2H6N2O2/c1-6-2(5)4-3/h3H2,1H3,(H,4,5)
(3)Std. InChIKey: WFJRIDQGVSJLLH-UHFFFAOYSA-N
Related Products
- Methyl (((methoxymethylphosphinothioyl)thio)acetyl)methylcarbamate
- Methyl (2R)-2-[(tert-butoxycarbonyl)amino]-3-iodopropanoate
- Methyl (2R)-2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate
- Methyl (2R)-2-amino-2-cyclohexylethanoate hydrochloride
- Methyl (2R)-2-bromo-2-(2-chlorophenyl)acetate
- Methyl (2R,3S)-3-(4-methoxyphenyl)-2-oxiranecarboxylate
- Methyl (2R,3S)-3-(benzoylamino)-2-hydroxy-3-phenylpropanoate
- Methyl (2R,3S)-3-(tert-butoxycarbonylamino)-2-hydroxy-3-phenylpropionate
- Methyl (2S)-1-(1,2-dioxo-3,3-dimethypentyl)-2-pyrrolidinecarboxylate
- Methyl (2S)-2,3-epoxypropanoate
- 6294-93-5
- 629-50-5
- 6295-18-7
- 6295-19-8
- 62952-06-1
- 6295-21-2
- 6295-22-3
- 6295-24-5
- 6295-28-9
- 6295-29-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View