-
Name
N-Aminoethylpiperazine
- EINECS 205-411-0
- CAS No. 140-31-8
- Article Data29
- CAS DataBase
- Density 0.958 g/cm3
- Solubility soluble in water
- Melting Point -19 °C
- Formula C6H15N3
- Boiling Point 220 °C at 760 mmHg
- Molecular Weight 129.205
- Flash Point 93.3 °C
- Transport Information UN 2815 8/PG 3
- Appearance colorless liquid
- Safety 26-36/37/39-45-61
- Risk Codes 21/22-34-43-52/53
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms Piperazine,1-(2-aminoethyl)- (6CI,7CI,8CI);1-(2-Aminoethyl)piperazine;1-(b-Aminoethyl)piperazine;1-Piperazineethylamine;2-(1-Piperazinyl)ethanamine;2-(1-Piperazinyl)ethylamine;2-Piperazinylethylamine;4-(2-Aminoethyl)piperazine;AEP;Ancamine AEP;N-(2-Aminoethyl)piperazine;N-(b-Aminoethyl)piperazine;N-AEP;NSC 38968;
- PSA 41.29000
- LogP -0.18270
Synthetic route
-
-
113585-93-6, 120574-64-3
2,5,8,10,13,16-Hexaazapentacyclo<8.6.1.12,5.09,18.013,17>octadecan

-
-
140-31-8
aminoethylpiperazine

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethanol at 125℃; under 41371.8 Torr; for 6h; | 94% |
-
-
18907-76-1
2,2’-(ethane-1,2-diylbis(azanediyl))diacetonitrile

-
A
-
140-31-8
aminoethylpiperazine

-
B
-
112-24-3
triethylentetramine

| Conditions | Yield |
|---|---|
| With hydrogen at 120℃; under 150015 Torr; Concentration; Temperature; Pressure; | A 5.2% B 87.3% |
| With hydrogen In tetrahydrofuran; toluene at 120℃; under 150015 Torr; for 500h; Autoclave; | A 6.1% B 83.6% |
| With hydrogen In tetrahydrofuran; water; toluene at 120℃; under 90009 Torr; for 25h; Autoclave; | A 12.1% B 71.9% |
| With hydrogen; Cr-doped Raney cobalt catalyst In tetrahydrofuran; water at 120℃; under 150015 Torr; for 3h; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| Stage #1: 2,2',2''-triaminotriethylamine With copper(II) perchlorate hexahydrate; acetonitrile Stage #2: With nitrogen(II) oxide In acetonitrile at 20℃; for 0.166667h; | A 60% B 30% |

-
A
-
140-31-8
aminoethylpiperazine

-
B
-
14057-91-1
tetrakis(acetonitrile)copper(I) perchlorate

| Conditions | Yield |
|---|---|
| With NO In acetonitrile NO purged into soln. of Cu complex in degassed CH3CN (O2-free conditions); stirred (10 min, room temp.); NO removed; layered with benzene; cooled; | A 60% B n/a C 30% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide | |
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With ammonia at 100℃; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 30 - 60℃; Rate constant; acid-catalyzed ring opening reactions at various temperature, Ea, ΔH are given; | |
| With hydrogenchloride In water at 30 - 60℃; Yield given; |

-
-
140-31-8
aminoethylpiperazine

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; Rate constant; Thermodynamic data; Ea, ΔH(excit.), ΔS(excit.); |

-
-
140-31-8
aminoethylpiperazine

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; Rate constant; Thermodynamic data; Ea, ΔH(excit.), ΔS(excit.); |

| Conditions | Yield |
|---|---|
| With water In acetonitrile at 30℃; Rate constant; |
-
-
107-15-3
ethylenediamine

-
A
-
110-85-0
piperazine

-
B
-
280-57-9
1,4-diaza-bicyclo[2.2.2]octane

-
C
-
140-31-8
aminoethylpiperazine

| Conditions | Yield |
|---|---|
| In water at 300℃; for 15h; Product distribution; Mechanism; other polyamines, amino alcohols and amino ethers; | A n/a B 30 % Chromat. C n/a |


| Conditions | Yield |
|---|---|
| In water at 75℃; for 3h; |

-
-
140-31-8
aminoethylpiperazine

| Conditions | Yield |
|---|---|
| Heating; |

| Conditions | Yield |
|---|---|
| at 100℃; analoge Reaktionen mit Aminen; |
-
-
107-15-3
ethylenediamine

-
A
-
110-85-0
piperazine

-
B
-
103-76-4
1-(2-hydroxyethyl)piperazine

-
C
-
140-31-8
aminoethylpiperazine

-
D
-
111-41-1
2-(2-Aminoethylamino)ethanol

-
E
-
111-40-0
1,5-diamino-3-azapentane

-
F
-
112-24-3
triethylentetramine

| Conditions | Yield |
|---|---|
| With hydrogen at 150 - 160℃; under 22502.3 Torr; |

-
-
141-43-5
ethanolamine

-
A
-
110-85-0
piperazine

-
B
-
103-76-4
1-(2-hydroxyethyl)piperazine

-
C
-
140-31-8
aminoethylpiperazine

-
D
-
111-41-1
2-(2-Aminoethylamino)ethanol

-
E
-
107-15-3
ethylenediamine

-
F
-
111-40-0
1,5-diamino-3-azapentane

-
G
-
112-24-3
triethylentetramine

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen at 170℃; under 150015 Torr; |

-
-
1051939-67-3
diethylenetriaminemonoacetonitrile

-
A
-
140-31-8
aminoethylpiperazine

-
B
-
112-24-3
triethylentetramine

| Conditions | Yield |
|---|---|
| With hydrogen |

-
-
18907-76-1
2,2’-(ethane-1,2-diylbis(azanediyl))diacetonitrile

-
A
-
110-85-0
piperazine

-
B
-
140-31-8
aminoethylpiperazine

-
C
-
111-40-0
1,5-diamino-3-azapentane

-
D
-
112-24-3
triethylentetramine

| Conditions | Yield |
|---|---|
| With hydrogen; Cr-doped Raney cobalt In tetrahydrofuran; water; ethylenediamine at 120℃; under 75007.5 Torr; for 3h; Product distribution / selectivity; Autoclave; |


| Conditions | Yield |
|---|---|
| With ammonia; hydrogen; Ni/Re (6.8wtpercent/1.8wtpercent) on silica/alumina (80/20) at 180℃; under 104192 - 119190 Torr; for 19h; Product distribution / selectivity; Autoclave; |

| Conditions | Yield |
|---|---|
| nickel on silica/alumina (Sud-Chemie C46-7-03) at 150℃; under 11775.6 - 38512.9 Torr; for 5h; Product distribution / selectivity; Autoclave; |
-
-
102-71-6
triethanolamine

-
A
-
110-85-0
piperazine

-
B
-
103-76-4
1-(2-hydroxyethyl)piperazine

-
C
-
140-31-8
aminoethylpiperazine

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen; Ni/Re (6.8wtpercent/1.8wtpercent) on silica/alumina (80/20) at 170℃; under 85781.4 - 99537.9 Torr; for 16h; Product distribution / selectivity; Autoclave; |

-
-
102-71-6
triethanolamine

-
A
-
110-85-0
piperazine

-
B
-
103-76-4
1-(2-hydroxyethyl)piperazine

-
C
-
140-31-8
aminoethylpiperazine

-
D
-
3197-06-6
N,N-bis(2-hydroxyethyl)ethylidenediamine

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen; Ni/Re (6.8wtpercent/1.8wtpercent) on silica/alumina (80/20) at 170℃; under 85781.4 - 99537.9 Torr; for 10h; Product distribution / selectivity; Autoclave; |

-
-
107-15-3
ethylenediamine

-
A
-
110-85-0
piperazine

-
B
-
140-31-8
aminoethylpiperazine

-
C
-
111-40-0
1,5-diamino-3-azapentane

-
D
-
112-24-3
triethylentetramine

| Conditions | Yield |
|---|---|
| With hydrogen; Ni/Re (6.8wtpercent/1.8wtpercent) on silica/alumina (80/20) at 162℃; under 9551.8 - 38512.9 Torr; for 6h; Product distribution / selectivity; Autoclave; |

-
-
112-24-3
triethylentetramine

-
A
-
110-85-0
piperazine

-
B
-
140-31-8
aminoethylpiperazine

-
C
-
24028-46-4
1-<2-(2-aminoethyl)aminoethyl>piperazine

| Conditions | Yield |
|---|---|
| With hydrogen; nickel on silica/alumina (Sud-Chemie C46-7-03) at 150℃; under 10379.3 - 32462.1 Torr; for 6h; Product distribution / selectivity; Autoclave; |

-
-
112-57-2
N-(2-aminoethyl)-N'-{2-[(2-aminoethyl)amino]ethyl}ethane-1,2-diamine

-
-
112-24-3
triethylentetramine

-
A
-
110-85-0
piperazine

-
B
-
140-31-8
aminoethylpiperazine

-
C
-
24028-46-4
1-<2-(2-aminoethyl)aminoethyl>piperazine

| Conditions | Yield |
|---|---|
| nickel on silica/alumina (Sud-Chemie C46-7-03) at 155℃; under 15550.9 - 38512.9 Torr; for 6h; Product distribution / selectivity; Autoclave; |

-
-
1211473-51-6
ethylenediaminediacetonitrile

-
-
540-61-4
2-aminoacetonitrile

-
A
-
140-31-8
aminoethylpiperazine

-
B
-
107-15-3
ethylenediamine

-
C
-
112-24-3
triethylentetramine

| Conditions | Yield |
|---|---|
| With hydrogen; Cr-doped Raney cobalt In tetrahydrofuran at 120℃; under 37503.8 Torr; Autoclave; |

-
-
1211473-51-6
ethylenediaminediacetonitrile

-
-
540-61-4
2-aminoacetonitrile

-
A
-
140-31-8
aminoethylpiperazine

-
B
-
107-15-3
ethylenediamine

-
C
-
111-40-0
1,5-diamino-3-azapentane

-
D
-
112-24-3
triethylentetramine

| Conditions | Yield |
|---|---|
| With hydrogen; Cr-doped Raney cobalt In tetrahydrofuran at 120℃; under 37503.8 Torr; Autoclave; |

-
-
18907-76-1
2,2’-(ethane-1,2-diylbis(azanediyl))diacetonitrile

-
A
-
110-85-0
piperazine

-
B
-
140-31-8
aminoethylpiperazine

-
C
-
111-40-0
1,5-diamino-3-azapentane

-
D
-
112-24-3
triethylentetramine

| Conditions | Yield |
|---|---|
| With hydrogen; Cr-doped Raney cobalt catalyst In tetrahydrofuran; water at 120℃; under 75007.5 Torr; for 3h; Product distribution / selectivity; |

-
-
111-40-0
1,5-diamino-3-azapentane

-
-
108-10-1
4-methyl-2-pentanone

-
-
67-63-0
isopropyl alcohol

-
-
140-31-8
aminoethylpiperazine

| Conditions | Yield |
|---|---|
| In methanol; water |

-
-
140-31-8
aminoethylpiperazine

-
-
100361-18-0
7-chloro-1-cyclopropyl-6-fluoro-4-oxo-1,4-dihydro-[1,8]naphthyridine-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In acetonitrile at 70℃; | 100% |
-
-
140-31-8
aminoethylpiperazine

-
-
107-13-1
acrylonitrile

-
-
96680-92-1
3-(4-{2-[bis-(2-cyanoethyl)amino]ethyl}piperazin-1-yl)propionitrile

| Conditions | Yield |
|---|---|
| Heating; | 99% |
| In methanol at 20℃; Michael condensation; | 94% |
-
-
140-31-8
aminoethylpiperazine

-
-
166186-30-7
3,3'-dibromo-4,4'-dimethoxy-5,6,5',6'-bis(methylenedioxy)biphenyl-2,2'-dicarboxylic anhydride

| Conditions | Yield |
|---|---|
| In benzene for 24h; Ambient temperature; | 99% |
-
-
140-31-8
aminoethylpiperazine

-
-
383-63-1
ethyl trifluoroacetate,

-
-
87980-84-5
1,1,1-trifluoro-N-(2-piperazin-1-ylethyl)acetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 20℃; for 2h; | 99% |

| Conditions | Yield |
|---|---|
| In ethanol at 20 - 75℃; for 60h; | 99% |
-
-
140-31-8
aminoethylpiperazine

-
-
67-56-1
methanol

-
-
104-19-8
N-methyl-N'-(2-dimethylaminoethyl)piperazine

| Conditions | Yield |
|---|---|
| With 5percent silver supported on titanium oxide at 25℃; for 10h; Inert atmosphere; Sealed tube; UV-irradiation; | 99% |
-
-
140-31-8
aminoethylpiperazine

-
-
85-44-9
phthalic anhydride

-
-
6820-93-5
2-(2-(piperazin-1-yl)ethyl)isoindoline-1,3-dione

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 180℃; for 5h; | 98% |
| at 180 - 200℃; for 3h; | 93% |
| In neat (no solvent) at 160℃; for 4h; |
-
-
140-31-8
aminoethylpiperazine

-
-
2059-76-9
4-iodophenyl isothiocyanate

-
-
77995-00-7
4-{2-[3-(4-Iodo-phenyl)-thioureido]-ethyl}-piperazine-1-carbothioic acid (4-iodo-phenyl)-amide

| Conditions | Yield |
|---|---|
| In ethanol for 4h; Heating; | 98% |
-
-
140-31-8
aminoethylpiperazine

-
-
13280-60-9
5-amino-2-nitrobenzoic acid

| Conditions | Yield |
|---|---|
| With 1,1'-carbonyldiimidazole In pyridine at 20 - 80℃; for 2.5h; | 98% |
-
-
140-31-8
aminoethylpiperazine

-
-
1333-84-2, 1344-28-1
aluminum oxide

-
-
86119-84-8, 7664-38-2
phosphoric acid

-
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| In water High Pressure; Al2O3 was dispersed in aq. H3PO4 and stirred for 2 h at room temp. N-(2-aminoethyl)-piperazine was added dropwise and stirred for 2 h, gel formed was transferred into Teflon-lined stainless steel autoclave and heatedat 220°C for 36 h; react. mixt. was cooled rapidly, ppt. was filtered, washed with water and air-dried at room temp.; | 98% |


| Conditions | Yield |
|---|---|
| With 1.1 wt% Pd/NiO; hydrogen In ethanol at 25℃; under 760.051 Torr; for 10h; | 98% |
-
-
140-31-8
aminoethylpiperazine

-
-
90-02-8
salicylaldehyde

-
-
93968-73-1
2-(((2-(piperazin-1-yl)ethyl)imino)methyl)phenol

| Conditions | Yield |
|---|---|
| In methanol Reflux; | 97.39% |
| In acetone for 0.5h; Ambient temperature; | 96% |
| In methanol for 3h; Reflux; | 88% |
-
-
140-31-8
aminoethylpiperazine

-
-
75-15-0
carbon disulfide

-
-
129352-08-5
Potassium; 4-(2-dithiocarboxyamino-ethyl)-piperazine-1-carbodithioate

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 60℃; | 97% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 0.5h; | 97% |
| In methanol at 20℃; for 72h; | 74% |
-
-
140-31-8
aminoethylpiperazine

-
-
108-10-1
4-methyl-2-pentanone

-
-
911415-27-5
4-methyl-N-(2-(piperazin-1-yl)ethyl)pentan-2-imine

| Conditions | Yield |
|---|---|
| at 130℃; for 5h; | 97% |
| at 140℃; for 8h; | |
| for 4h; Heating / reflux; | |
| With sodium carbonate Reflux; Inert atmosphere; Dean-Stark; | |
| Stage #1: aminoethylpiperazine; 4-methyl-2-pentanone Inert atmosphere; Reflux; Stage #2: With pentaerythritol tetraglycidyl ether In methanol at 50 - 60℃; Dean-Stark; |
-
-
140-31-8
aminoethylpiperazine

-
-
60457-62-7
methyl 4-[4-(methyloxy)-4-oxobutyl]disulfanylbutanoate

| Conditions | Yield |
|---|---|
| In methanol at 25 - 65℃; for 40h; | 97% |
-
-
140-31-8
aminoethylpiperazine

-
-
1761-61-1
5-bromosalicyclaldehyde

-
-
639452-69-0
2-((2-(piperazin-1-yl)ethylimino)methyl)-4-bromophenol

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 0.5h; | 97% |
| In ethanol at 20℃; for 1.5h; Reflux; | 95% |
| In methanol for 6h; Reflux; | 74.3% |
| In methanol |

| Conditions | Yield |
|---|---|
| at 30 - 60℃; | 97% |

-
-
140-31-8
aminoethylpiperazine

-
-
527-69-5
2-furancarbonyl chloride

-
-
80838-44-4
Furan-2-carboxylic acid {2-[4-(furan-2-carbonyl)-piperazin-1-yl]-ethyl}-amide

| Conditions | Yield |
|---|---|
| In benzene for 1h; Heating; | 96% |

| Conditions | Yield |
|---|---|
| In methanol for 8h; Heating; | 96% |
| In methanol for 8h; Heating; | 95% |
| Yield given; |
-
-
140-31-8
aminoethylpiperazine

-
-
49627-27-2
sulindac sulfide

| Conditions | Yield |
|---|---|
| With O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate; triethylamine In acetonitrile at 20℃; Inert atmosphere; | 96% |
-
-
140-31-8
aminoethylpiperazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 72h; Inert atmosphere; | 96% |
-
-
140-31-8
aminoethylpiperazine

-
-
4808-48-4
2,3-diphenylmaleic anhydride

-
-
128143-36-2
3,4-diphenyl-1-<2-(1-piperazinyl)ethyl>-1H-pyrrole-2,5-dione

| Conditions | Yield |
|---|---|
| In ethanol for 2h; Ambient temperature; | 95% |
-
-
140-31-8
aminoethylpiperazine

-
-
107-13-1
acrylonitrile

-
-
68310-66-7
3-(2-Piperazin-1-yl-ethylamino)-propionitrile

| Conditions | Yield |
|---|---|
| In benzene for 6h; Heating; | 95% |
-
-
140-31-8
aminoethylpiperazine

-
-
79-11-8
chloroacetic acid

-
-
136369-41-0
N-methoxycarbonyl-N'-(β-dimethoxycarbonylaminoethyl)piperazine

| Conditions | Yield |
|---|---|
| In toluene for 4h; Heating; | 95% |
-
-
140-31-8
aminoethylpiperazine

-
-
6590-96-1
2, 4-dichlorophenyl isothiocyanate

-
-
77995-01-8
4-{2-[3-(2,4-Dichloro-phenyl)-thioureido]-ethyl}-piperazine-1-carbothioic acid (2,4-dichloro-phenyl)-amide

| Conditions | Yield |
|---|---|
| In ethanol for 4h; Heating; | 95% |
-
-
140-31-8
aminoethylpiperazine

-
-
85-43-8
1,2,3,6-Tetrahydrophthalic anhydride

-
-
52599-47-0
2-(2-Piperazin-1-yl-ethyl)-3a,4,7,7a-tetrahydro-isoindole-1,3-dione

| Conditions | Yield |
|---|---|
| at 180 - 200℃; for 3h; | 95% |

| Conditions | Yield |
|---|---|
| In water High Pressure; Sb, S and org. compd. diluted with water and heated in autoclave at 170°C for 7 d; cooled to room temp., filtered, washed with deionised water and acetone;elem. anal.; | 95% |

-
-
140-31-8
aminoethylpiperazine

-
-
635-93-8
5-chlorosalicyclaldehyde

-
-
1245707-53-2
2-((2-(piperazin-1-yl)ethylimino)methyl)-4-chlorophenol

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 0.5h; | 95% |
| In methanol for 6h; Reflux; | 73.2% |
| In methanol for 0.5h; Reflux; | |
| In ethanol for 0.5h; Reflux; Green chemistry; | |
| In methanol |
-
-
140-31-8
aminoethylpiperazine

-
-
98-03-3
thiophene-2-carbaldehyde

-
-
67-52-7
BARBITURIC ACID

-
-
126-81-8
dimedone

-
-
1309833-69-9
8,8‐dimethyl‐10‐[2‐(piperazin‐1‐yl)ethyl]‐5‐(thiophen‐2‐yl)‐8,9‐dihydropyrimido[4,5‐b]quinoline‐2,4,6(1H,3H,5H,7H,10H)‐trione

| Conditions | Yield |
|---|---|
| With phosphotungstic acid In ethanol for 6h; Reflux; | 95% |
| Stage #1: thiophene-2-carbaldehyde; BARBITURIC ACID; dimedone With polyphosphoric acid supported on CoFe2O4 nanoparticles In ethanol for 0.0166667h; Sonication; Green chemistry; Stage #2: aminoethylpiperazine In ethanol for 0.05h; Sonication; Green chemistry; | 95% |

N-AMINOETHYLPIPERAZINE Chemical Properties
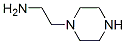
MF: C6H15N3
MW: 129.2
Synonyms: LABOTEST-BB LTBB000498;AMINOETHYLPIPERAZINE;AKOS BBS-00004342;AEP;1-(2-AMINOETHYL)PIPERAZINE;2-(1-PIPERAZINYL)ETHYLAMINE;1-piperazineethanamine;2-PIPERAZINOETHYLAMINE
Product Categories: Piperaizine;Piperazines
Mol File: 140-31-8.mol
mp -19 °C
bp 218-222 °C(lit.)
density 0.985 g/mL at 25 °C(lit.)
vapor density 4.4 (vs air)
vapor pressure 0.05 mm Hg ( 20 °C)
refractive index n20/D 1.500
Fp 200 °F
Water Solubility soluble
Sensitive Air Sensitive
BRN 104363
CAS DataBase Reference 140-31-8(CAS DataBase Reference)
NIST Chemistry Reference 140-31-8(NIST)
N-AMINOETHYLPIPERAZINE Uses
N-aminoethylpiperazine (140-31-8) could be used with Pharmaceutical Intermediates.
Also,Delamine N-Aminoethylpiperazine(140-31-8) is appropriate for use in:
Bitumen Chemicals
Corrosion Inhibitors
Epoxy Curing Agents
Lube Oil and fuel additives
Mineral Processing Aids
Polyamide Resins
Urethane chemicals
Wet Strength Resins
N-AMINOETHYLPIPERAZINE Production
N-aminoethylpiperazine (140-31-8) is a reaction to produce ethylene dichloride and ammonia by-product of ethylenediamine class products.
N-aminoethylpiperazine (140-31-8) of the consumption of raw materials fixed: dichloroethane 2100kg / t, liquid ammonia 620kg / t, caustic soda (42%) 4143kg / t.
N-AMINOETHYLPIPERAZINE Toxicity Data With Reference
| 1. | skn-rbt 100 µg/24H open | AIHAAP American Industrial Hygiene Association Journal. 23 (1962),95. | ||
| 2. | skn-rbt 5 mg/24H SEV | 85JCAE Prehled Prumyslove Toxikologie; Organicke Latky Marhold, J.,Prague, Czechoslovakia.: Avicenum,1986,864. | ||
| 3. | eye-rbt 20 mg/24H MOD | 85JCAE Prehled Prumyslove Toxikologie; Organicke Latky Marhold, J.,Prague, Czechoslovakia.: Avicenum,1986,864. | ||
| 4. | sce-ham:ovr 125 µg/L | MUREAV Mutation Research. 320 (1994),31. | ||
| 5. | msc-ham:ovr 500 µg/L | MUREAV Mutation Research. 320 (1994),31. | ||
| 6. | otr-mus:lym 1 mL/L | ENMUDM Environmental Mutagenesis. 4 (1982),390. | ||
| 7. | orl-rat LD50:2140 mg/kg | AIHAAP American Industrial Hygiene Association Journal. 23 (1962),95. | ||
| 8. | ipr-mus LD50:250 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) AD277-689 . | ||
| 9. | skn-rbt LD50:880 mg/kg | UCDS** Union Carbide Data Sheet. (Industrial Medicine and Toxicology Dept., Union Carbide Corp., 270 Park Ave., New York, NY 10017) 6/13/69 . |
N-AMINOETHYLPIPERAZINE Consensus Reports
N-AMINOETHYLPIPERAZINE Safety Profile
Hazard Codes C
Safety Statements 26-36/37/39-45-61
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
37: Wear suitable gloves
39: Wear eye/face protection
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
61: Avoid release to the environment. Refer to special instructions safety data sheet
N-AMINOETHYLPIPERAZINE Standards and Recommendations
Related Products
- N-AMINOETHYLPIPERAZINE
- 14032-21-4
- 1403-36-7
- 1403-45-8
- 14034-59-4
- 14034-79-8
- 14035-33-7
- 14035-96-2
- 14036-06-7
- 14036-55-6
- 1403-66-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View