-
Name
N,N'-Methylenebisacrylamide
- EINECS 203-750-9
- CAS No. 110-26-9
- Density 1.054 g/cm3
- Solubility 0.01-0.1 g/100 mL at 18 °C in water
- Melting Point >300 °C(lit.)
- Formula C7H10N2O2
- Boiling Point 445.1 °C at 760 mmHg
- Molecular Weight 154.169
- Flash Point 215 °C
- Transport Information UN 2811
- Appearance white crystalline powder
- Safety 22-36/37-36-26
- Risk Codes 20/22-20/21/22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms Acrylamide,N,N'-methylenebis- (6CI,7CI,8CI);Bisacrylamide;Methylenebisacrylamide;Methylenediacrylamide;N,N'-Diacryloylmethylenediamine;N,N'-Methylenebis(2-propenamide);N,N'-Methylenebis(acrylamide);2-Propenamide,N,N'-methylenebis-;NSC 406836;NSC 7774;Triam 507;
- PSA 58.20000
- LogP 0.33010
Synthetic route

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; hydroquinone In water at 65 - 75℃; for 2.5h; Reagent/catalyst; Temperature; | 87.54% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
50-00-0
formaldehyd

-
-
107-06-2
1,2-dichloro-ethane

-
-
79-06-1
2-propenamide

-
-
110-26-9
N,N'-Methylenebisacrylamide

| Conditions | Yield |
|---|---|
| With hydrogenchloride |


| Conditions | Yield |
|---|---|
| With sulfuric acid; water | |
| With hydroquinone | |
| With sulfuric acid |
-
-
50-00-0
formaldehyd

-
-
7664-93-9
sulfuric acid

-
-
107-13-1
acrylonitrile

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
115-77-5
Pentaerythritol

-
-
924-42-5
N-Methylolacrylamid

-
B
-
85030-50-8
C13H22N2O6

-
I
-
110-26-9
N,N'-Methylenebisacrylamide

| Conditions | Yield |
|---|---|
| Stage #1: Pentaerythritol; N-Methylolacrylamid With tempol; toluene-4-sulfonic acid In methanol at 86℃; for 1.5h; Stage #2: With ammonia In methanol; propyleneglycol monomethylether; water |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; potassium carbonate; triethylamine In 1,4-dioxane; methanol; water; ethyl acetate | 200 mg (58%) |
-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
139031-06-4
bis(mesityleneformyl)phosphine

-
-
1591653-06-3
N,N-methylene(bis(bis(2,4,6-trimethylbenzoyl)phosphino)propanamide)

| Conditions | Yield |
|---|---|
| With triethylamine In 1,2-dimethoxyethane; toluene at 50℃; for 12h; Schlenk technique; Inert atmosphere; | 99% |
-
-
347371-56-6
N,N'-bis(2,6-diisopropylphenyl)-1,6,7,12-tetra[4-(2-aminoethyl)phenoxy]perylene-3,4,9,10-tetracarboxylic acid diimide

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
1549684-13-0
C136H158N22O24

| Conditions | Yield |
|---|---|
| In methanol; water at 50℃; for 48h; Michael Addition; Inert atmosphere; | 95% |
-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
79-10-7
acrylic acid

- poly(acrylic acid), cross-linked by 0.15 wt % of N,N'-methylenebisacrylamide, neutralized with degree of neutralization 0.3; monomer(s): acrylic acid; N,N'-methylenebisacrylamide
-
poly(acrylic acid), cross-linked by 0.15 wt % of N,N'-methylenebisacrylamide, neutralized with degree of neutralization 0.3; monomer(s): acrylic acid; N,N'-methylenebisacrylamide

| Conditions | Yield |
|---|---|
| With ammonium persulfate; N,N,N,N,-tetramethylethylenediamine In water at 30℃; for 1h; | 93% |

| Conditions | Yield |
|---|---|
| With triethylamine at 20℃; for 24h; Michael Addition; | 93% |
-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
-
122-52-1
triethyl phosphite

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 5.25h; diastereoselective reaction; | 92% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
1147550-11-5
ammonium thiocyanate

-
-
369-57-3
benzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 10 - 15℃; for 1.25h; | 91% |
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 10 - 15℃; for 0.75h; | 91% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
178949-82-1
trisodium (S,S)-ethylenediamine-N,N’-disuccinic acid

| Conditions | Yield |
|---|---|
| With lithium hydroxide at 20℃; for 48h; | 90.1% |

| Conditions | Yield |
|---|---|
| With 2,4-di-tert-Butylphenol; toluene-4-sulfonic acid In chloroform Ambient temperature; 2-4d; | 90% |
-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
1147550-11-5
ammonium thiocyanate

| Conditions | Yield |
|---|---|
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 10 - 15℃; | 90% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
762-21-0
acetylenedicarboxylic acid diethyl ester

-
-
121-45-9
phosphorous acid trimethyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 5.25h; diastereoselective reaction; | 90% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
2210-25-5
N-Isopropylacrylamide

-
-
25999-13-7
N-propylacrylamide

-
-
627546-76-3
4-N-(2-acryloyloxyethyl)-N-methylamino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole

- polymer, emulsion polymerization; Monomer(s): N-isopropylacrylamide, 75 mM; N-n-propylacrylamide, 25 mM; N,N\-methylenebisacrylamide, 1 mM; 4-N-(2-acryloyloxyethyl)-N-methylamino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole, 0.1 mM
-
polymer, emulsion polymerization; Monomer(s): N-isopropylacrylamide, 75 mM; N-n-propylacrylamide, 25 mM; N,N\-methylenebisacrylamide, 1 mM; 4-N-(2-acryloyloxyethyl)-N-methylamino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole, 0.1 mM

| Conditions | Yield |
|---|---|
| With ammonium persulfate; N,N,N,N,-tetramethylethylenediamine; sodium dodecyl-sulfate at 70℃; for 4h; | 88.3% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
-
121-45-9
phosphorous acid trimethyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 5.25h; diastereoselective reaction; | 87% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
333-20-0
potassium thioacyanate

-
-
369-57-3
benzenediazonium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 15℃; for 1.75h; | 86% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
459-64-3
4-methoxybenzenediazonium tetrafluoroborate

-
-
1147550-11-5
ammonium thiocyanate

-
-
524067-02-5
N-{[3-(4-methoxy-phenyl)-2-thiocyanato-propionylamino]-methyl}-acrylamide

| Conditions | Yield |
|---|---|
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 10 - 15℃; | 85% |
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 10 - 15℃; | 85% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
1147550-11-5
ammonium thiocyanate

-
-
459-44-9
4-methylbenzenediazonium tetrafluoroborate

-
-
524067-01-4
N-[(2-thiocyanato-3-p-tolyl-propionylamino)-methyl]-acrylamide

| Conditions | Yield |
|---|---|
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 10 - 15℃; | 85% |
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 10 - 15℃; | 85% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
79-10-7
acrylic acid

- poly(acrylic acid), cross-linked by 0.2 wt % of N,N'-methylenebisacrylamide, neutralized with degree of neutralization 0.4; monomer(s): acrylic acid; N,N'-methylenebisacrylamide
-
poly(acrylic acid), cross-linked by 0.2 wt % of N,N'-methylenebisacrylamide, neutralized with degree of neutralization 0.4; monomer(s): acrylic acid; N,N'-methylenebisacrylamide

| Conditions | Yield |
|---|---|
| With ammonium persulfate; N,N,N,N,-tetramethylethylenediamine In water at 50℃; for 1h; | 85% |
-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
66086-33-7
Di-tert-butyl acetylenedicarboxylate

-
-
121-45-9
phosphorous acid trimethyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 5.25h; diastereoselective reaction; | 85% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
2210-25-5
N-Isopropylacrylamide

-
-
627546-76-3
4-N-(2-acryloyloxyethyl)-N-methylamino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole

- polymer, emulsion polymerization; Monomer(s): N-isopropylacrylamide, 100 mM; N,N\-methylenebisacrylamide, 1 mM; 4-N-(2-acryloyloxyethyl)-N-methylamino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole, 0.1 mM
-
polymer, emulsion polymerization; Monomer(s): N-isopropylacrylamide, 100 mM; N,N\-methylenebisacrylamide, 1 mM; 4-N-(2-acryloyloxyethyl)-N-methylamino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole, 0.1 mM

| Conditions | Yield |
|---|---|
| With ammonium persulfate; N,N,N,N,-tetramethylethylenediamine; sodium dodecyl-sulfate at 70℃; for 4h; | 84.9% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
13749-61-6
N-isopropylmethacrylamide

-
-
627546-76-3
4-N-(2-acryloyloxyethyl)-N-methylamino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole

- polymer, emulsion polymerization; Monomer(s): N-isopropylmethacrylamide, 100 mM; N,N\-methylenebisacrylamide, 1 mM; 4-N-(2-acryloyloxyethyl)-N-methylamino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole, 0.1 mM
-
polymer, emulsion polymerization; Monomer(s): N-isopropylmethacrylamide, 100 mM; N,N\-methylenebisacrylamide, 1 mM; 4-N-(2-acryloyloxyethyl)-N-methylamino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole, 0.1 mM

| Conditions | Yield |
|---|---|
| With ammonium persulfate; N,N,N,N,-tetramethylethylenediamine; sodium dodecyl-sulfate at 70℃; for 4h; | 84.9% |


| Conditions | Yield |
|---|---|
| With ammonium persulfate; N,N,N,N,-tetramethylethylenediamine In various solvent(s) at 65℃; Product distribution; Further Variations:; Solvents; Temperatures; | 84% |
-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
66086-33-7
Di-tert-butyl acetylenedicarboxylate

-
-
122-52-1
triethyl phosphite

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 5.25h; diastereoselective reaction; | 84% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
5205-93-6
2-methyl-acrylic acid 3-dimethylamino-propyl amide

-
-
79-06-1
2-propenamide

-
-
99349-68-5
(3-acrylamidophenyl)boronic acid

| Conditions | Yield |
|---|---|
| With α,α'-azodiizobutyramidine-dihydrochloride In water; dimethyl sulfoxide at 60℃; for 20h; | 83.7% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
2093-46-1
2-methylphenyl diazonium tetrafluoroborate

-
-
1147550-11-5
ammonium thiocyanate

| Conditions | Yield |
|---|---|
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 10 - 15℃; | 82% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
99685-96-8, 161105-99-3, 161106-00-9, 111138-12-6, 133318-63-5, 134053-11-5, 134931-35-4, 134931-36-5, 139703-76-7, 145633-27-8, 175414-73-0, 175414-74-1, 175414-75-2, 175519-12-7, 175519-13-8, 175519-14-9, 175519-15-0, 136376-46-0, 144906-37-6, 144906-38-7, 151767-00-9, 152882-97-8, 152882-98-9, 152882-99-0, 153062-34-1, 154171-74-1, 154171-75-2, 154333-99-0, 154334-00-6, 154397-63-4, 154460-59-0, 199456-56-9, 108739-25-9, 120329-57-9, 120329-58-0
fullerene-C60

-
-
79-10-7
acrylic acid

- poly(acrylic acid), cross-linked by 0.15 wt % of N,N'-methylenebisacrylamide, modified by 0.1 wt % of [60]fullerene, neutralized with degree of neutralization 0.4; monomer(s): acrylic acid; N,N'-methylenebisacrylamide; [60]fullerene
-
poly(acrylic acid), cross-linked by 0.15 wt % of N,N'-methylenebisacrylamide, modified by 0.1 wt % of [60]fullerene, neutralized with degree of neutralization 0.4; monomer(s): acrylic acid; N,N'-methylenebisacrylamide; [60]fullerene

| Conditions | Yield |
|---|---|
| With ammonium persulfate; N,N,N,N,-tetramethylethylenediamine In water at 50℃; for 1.4h; | 82% |

-
-
101-02-0
triphenyl phosphite

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
66086-33-7
Di-tert-butyl acetylenedicarboxylate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 5.25h; diastereoselective reaction; | 82% |


| Conditions | Yield |
|---|---|
| With copper(II) bis(tetrafluoroborate) In water; N,N-dimethyl-formamide at 15℃; | 81% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
762-21-0
acetylenedicarboxylic acid diethyl ester

-
-
122-52-1
triethyl phosphite

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 5.25h; diastereoselective reaction; | 81% |

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
99685-96-8, 161105-99-3, 161106-00-9, 111138-12-6, 133318-63-5, 134053-11-5, 134931-35-4, 134931-36-5, 139703-76-7, 145633-27-8, 175414-73-0, 175414-74-1, 175414-75-2, 175519-12-7, 175519-13-8, 175519-14-9, 175519-15-0, 136376-46-0, 144906-37-6, 144906-38-7, 151767-00-9, 152882-97-8, 152882-98-9, 152882-99-0, 153062-34-1, 154171-74-1, 154171-75-2, 154333-99-0, 154334-00-6, 154397-63-4, 154460-59-0, 199456-56-9, 108739-25-9, 120329-57-9, 120329-58-0
fullerene-C60

-
-
79-10-7
acrylic acid

- poly(acrylic acid), cross-linked by 0.1 wt % of N,N'-methylenebisacrylamide, modified by 0.1 wt % of [60]fullerene, neutralized with degree of neutralization 0.4; monomer(s): acrylic acid; N,N'-methylenebisacrylamide; [60]fullerene
-
poly(acrylic acid), cross-linked by 0.1 wt % of N,N'-methylenebisacrylamide, modified by 0.1 wt % of [60]fullerene, neutralized with degree of neutralization 0.4; monomer(s): acrylic acid; N,N'-methylenebisacrylamide; [60]fullerene

| Conditions | Yield |
|---|---|
| With ammonium persulfate; N,N,N,N,-tetramethylethylenediamine In water at 50℃; for 1.5h; | 80% |

-
-
101-02-0
triphenyl phosphite

-
-
110-26-9
N,N'-Methylenebisacrylamide

-
-
762-21-0
acetylenedicarboxylic acid diethyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 5.25h; diastereoselective reaction; | 80% |

N,N'-Methylenebisacrylamide Chemical Properties
Molecular structure of N,N'-Methylenebisacrylamide (CAS NO.110-26-9):
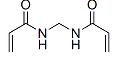
IUPAC Name: N-[(Prop-2-enoylamino)methyl]prop-2-enamide
Molecular Formula: C7H10N2O2
Molecular Weight: 154.1665 g/mol
Density: 1.054 g/cm3
Melting Point: >300 °C(lit.)
Boiling Point: 445.1 °C at 760 mmHg
Flash Point: 215 °C
Index of Refraction: 1.475
Molar Refractivity: 41.17 cm3
Molar Volume: 146.1 cm3
Surface Tension: 34.7 dyne/cm
Enthalpy of Vaporization: 70.3 kJ/mol
Vapour Pressure: 4.06E-08 mmHg at 25 °C
Storage Temp.: 2-8 °C
Water Solubility: 0.01-0.1 g/100 mL at 18 °C
XLogP3-AA: 0.1
H-Bond Donor: 2
H-Bond Acceptor: 2
Rotatable Bond Count: 4
Tautomer Count: 3
Exact Mass: 154.074228
MonoIsotopic Mass: 154.074228
Topological Polar Surface Area: 58.2
Heavy Atom Count: 11
Canonical SMILES: C=CC(=O)NCNC(=O)C=C
InChI: InChI=1S/C7H10N2O2/c1-3-6(10)8-5-9-7(11)4-2/h3-4H,1-2,5H2,(H,8,10)(H,9,11)
InChIKey: ZIUHHBKFKCYYJD-UHFFFAOYSA-N
EINECS: 203-750-9
Product Categories: Biochemistry; Reagents for Electrophoresis; Acrylamide; Molecular Biology; Nucleic Acid Electrophoresis
N,N'-Methylenebisacrylamide Uses
N,N'-Methylenebisacrylamide (CAS NO.110-26-9) is an important material for the separation of Amino Acids. It is essential to photosensitive nylon and plastics. N,N'-Methylenebisacrylamide is also used as plugging agent in oilfield drilling and construction grouting. It is used as cross-linking agent in the synthesis of acrylic resin and adhesives.
N,N'-Methylenebisacrylamide Toxicity Data With Reference
| 1. | mma-sat 1 mg/plate | EMMUEG Environmental and Molecular Mutagenesis. 11 (Suppl 12)(1988),1. | ||
| 2. | dlt-mus-ipr 225 mg/kg | MUREAV Mutation Research. 229 (1990),161. | ||
| 3. | orl-rat LD50:390 mg/kg | 37ASAA Kirk-Othmer Encyclopedia of Chemical Technology. 1 (1978),306. | ||
| 4. | orl-mus LD50:380 mg/kg | JACTDZ Journal of the American College of Toxicology. 1 (1990),111. |
N,N'-Methylenebisacrylamide Consensus Reports
Reported in EPA TSCA Inventory.
N,N'-Methylenebisacrylamide Safety Profile
Poison by ingestion. Experimental reproductive effects. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx. See also AMIDES.
Hazard Codes:  Xn,
Xn,  Xi
Xi
Risk Statements: 20/22-20/21/22-36/37/38
20/22: Harmful by inhalation and if swallowed
20/21/22: Harmful by inhalation, in contact with skin and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
Safety Statements: 22-36/37-36-26
22: Do not breathe dust
36/37: Wear suitable protective clothing and gloves
36: Wear suitable protective clothing
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
RIDADR: UN 2811
WGK Germany: 2
RTECS: AS3678000
F: 8
F8: Photosensitive
HazardClass: 6.1(b)
PackingGroup: III
HS Code: 29241900
N,N'-Methylenebisacrylamide Specification
N,N'-Methylenebisacrylamide (CAS NO.110-26-9) is also named as 2-Propenamide, N,N'-methylenebis- ; 4-02-00-01472 (Beilstein Handbook Reference) ; AI3-08643 ; BRN 1706297 ; CCRIS 4672 ; Methylenebisacrylamide ; Methylenediacrylamide ; N,N'-Methylenebis(2-propenamide) ; N,N'-Methylenebis(acrylamide) ; N,N'-Methylenediacrylamide ; N,N'-Methylidenebisacrylamide ; NSC 406836 . N,N'-Methylenebisacrylamide (CAS NO.110-26-9) is white crystalline powder with a neutral odor. It is very slightly water soluble. N,N'-Methylenebisacrylamide is incompatible with strong oxidizers, strong acids and strong bases. Flash point data for N,N'-Methylenebisacrylamide are not available; however, it is probably combustible.
Related Products
- N-[(10-Oxido-9,10-dihydro-9-oxa-10-phosphaphenanthrene)methyl]-1,3,5-triazine-2,4,6-triamine
- N10-(Trifluoroacetyl)pteroic acid
- N-[1,1'-Biphenyl]-4-yl-9,9-dimethyl-9H-fluoren-2-amine
- N-[1,1'-Biphenyl]-4-yl-9,9-dimethyl-9H-fluoren-3-amine
- N-[1,1'-Biphenyl]-4-yl-N-(4-bromophenyl)-9,9-dimethyl-9H-fluoren-2-amine
- N-[1,1-Bis[(acetyloxy)methyl]-3-(4-octylphenyl)propyl]acetamide
- N'-[(1,1-Dimethylethoxy)carbonyl]-N-[(9H-fluoren-9-ylmethoxy)carbonyl]-N'-methyl-L-lysine
- N1,1-Diphenyl-1,2-ethanediamine
- N-(1,2-Dimethylpropyl)-2-pyridinamine
- N<sup xmlns="">1</sup>-(3,4-DIMETHYL-5-ISOXAZOLYL)SULFANIL-AMIDE LITHIUM SALT
- 110-27-0
- 11027-58-0
- 11027-63-7
- 11028-42-5
- 110284-79-2
- 11028-71-0
- 11029-06-4
- 11029-12-2
- 110-30-5
- 110-31-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View