-
Name
Pentafluorobenzoic acid
- EINECS 210-026-6
- CAS No. 602-94-8
- Article Data44
- CAS DataBase
- Density 1.724 g/cm3
- Solubility
- Melting Point 100-102 °C(lit.)
- Formula C7HF5O2
- Boiling Point 220.014 °C at 760 mmHg
- Molecular Weight 212.076
- Flash Point 86.862 °C
- Transport Information UN 2811 6.1/PG 3
- Appearance white to light yellow crystal powder
- Safety 26-45-36/37/39-24/25
- Risk Codes 25-36/37/38
-
Molecular Structure
-
Hazard Symbols
 T,
T, Xi
Xi
- Synonyms Perfluorobenzoic acid;2,3,4,5,6-Pentafluorobenzoic acid;2,3,4,5,6-pentafluorobenzoate;
- PSA 37.30000
- LogP 2.08030
Synthetic route

| Conditions | Yield |
|---|---|
| With cobalt(II) 2,9,16,23-phthalocyanine tetrasulfonic acid In water; acetonitrile at 20℃; under 760.051 Torr; for 150h; UV-irradiation; | 100% |
| With 9-oxyl-9-azabicyclo<3.3.1>nonan-3-one; oxygen; nitric acid; sodium nitrite In water; acetonitrile at 23℃; under 760.051 Torr; for 8h; Sealed tube; | 85% |
| Multi-step reaction with 2 steps 1: pyridine / benzene 2: H2SO4 View Scheme |
-
-
22236-19-7
pentafluorobenzoyl peroxide

-
-
150-78-7
1,4-dimethoxybezene

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
98040-87-0
2,3,4,5,6-Pentafluoro-benzoic acid 2,5-dimethoxy-phenyl ester

-
C
-
98040-88-1
C22H8F10O6

| Conditions | Yield |
|---|---|
| In 1,1,2-Trichloro-1,2,2-trifluoroethane at 25℃; Product distribution; Rate constant; Mechanism; different starting concentration ratios; | A 98% B n/a C n/a |
| In 1,1,2-Trichloro-1,2,2-trifluoroethane at 25℃; Yield given. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| With sulfuric acid In water at 155℃; for 2h; Temperature; | 95.3% |
| With sulfuric acid In water at 145 - 165℃; for 15h; | |
| With sulfuric acid hydrolysis at 180°C, with 75 % H2SO4; |
-
-
22236-19-7
pentafluorobenzoyl peroxide

-
-
151-10-0
1,3-Dimethoxybenzene

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
98040-85-8
2,3,4,5,6-Pentafluoro-benzoic acid 2,4-dimethoxy-phenyl ester

-
C
-
98040-86-9
C22H8F10O6

| Conditions | Yield |
|---|---|
| In 1,1,2-Trichloro-1,2,2-trifluoroethane at 50℃; Product distribution; Rate constant; Mechanism; different starting concentration ratios; | A 95% B n/a C n/a |
| In 1,1,2-Trichloro-1,2,2-trifluoroethane at 50℃; Yield given. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| With diethylene glycol dimethyl ether at 70℃; for 0.5h; Sonication; | 95% |
| With sodium hypochlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; potassium bromide In water; acetonitrile at 20℃; aq. buffer; | 82% |
-
-
22236-19-7
pentafluorobenzoyl peroxide

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
98040-93-8
tert-butyl 2,3,4,5,6-pentafluorobenzoate

| Conditions | Yield |
|---|---|
| In various solvent(s) at 20℃; Product distribution; Oxidation; | A 30% B 74% C 94% |
| In various solvent(s) at 20℃; Oxidation; | A 30% B 74% C 94% |


| Conditions | Yield |
|---|---|
| With carbon dioxide In diethyl ether; water react. of (C6F5)3SiH with CH3Li at -50°C, 1 h gives after treatment of the react. product with CO2 in H2O/H(1+) C6F5COOH; | 90% |
| With CO2 In diethyl ether; water react. of (C6F5)3SiH with CH3Li at -50°C, 1 h gives after treatment of the react. product with CO2 in H2O/H(1+) C6F5COOH; | 90% |

| Conditions | Yield |
|---|---|
| With formic acid; dihydrogen peroxide for 5h; | 89% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; | 87% |
| Multi-step reaction with 3 steps 1: 85 percent / pyridine / diethyl ether / 1.5 h / Heating 2: 80 percent / H2 / 5 percent Pd/C / methanol / 760 Torr / Ambient temperature 3: 5 percent / potassium carbonate / H2O / 10 h / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: bromopentafluorobenzene; carbon dioxide With tert.-butyl lithium In pentane at -70℃; Inert atmosphere; Stage #2: With sulfuric acid; water In pentane at 20℃; Inert atmosphere; | 86% |
-
-
853-75-8
N-pentafluorophenyl-pentafluorophenylaldimine

-
A
-
880-78-4
pentafluoronitrobenzen

-
B
-
602-94-8
Pentafluorobenzoic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; trifluoroacetic anhydride In dichloromethane for 10h; Heating; | A 85% B 82% |
-
-
22236-19-7
pentafluorobenzoyl peroxide

-
-
92101-95-6
2-methyl-5-tert-butyl-1,4-dimethoxybenzene

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
98040-93-8
tert-butyl 2,3,4,5,6-pentafluorobenzoate

| Conditions | Yield |
|---|---|
| In various solvent(s) at 20℃; Product distribution; Oxidation; | A 85% B 19% C 24% D 75% |
| In various solvent(s) at 20℃; Oxidation; | A 85% B 19% C 24% D 78% |

-
-
22236-19-7
pentafluorobenzoyl peroxide

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
98040-93-8
tert-butyl 2,3,4,5,6-pentafluorobenzoate

| Conditions | Yield |
|---|---|
| In various solvent(s) at 20℃; Product distribution; Oxidation; | A 85% B 15% C 17% D n/a |

-
-
22236-19-7
pentafluorobenzoyl peroxide

-
A
-
602-94-8
Pentafluorobenzoic acid

| Conditions | Yield |
|---|---|
| In various solvent(s) at 20℃; Oxidation; | A 85% B 17% C n/a D n/a |

-
-
124-38-9
carbon dioxide

-
-
17436-90-7
bis-(pentafluoro phenyl) magnesium

-
-
602-94-8
Pentafluorobenzoic acid

| Conditions | Yield |
|---|---|
| In tetrahydrofuran reaction at 20°C, 3 hours;; | 83% |
| In tetrahydrofuran reaction at 20°C, 3 hours;; | 83% |
-
-
97580-75-1
bis(α-pentafluoroanilino-2,3,4,5,6-pentafluorobenzyl)peroxide

-
-
407-25-0
trifluoroacetic anhydride

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
653-27-0
α,α,α,2,3,4,5,6-octafluoroacetanilide

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In dichloromethane for 5h; | A 71% B 79% |


| Conditions | Yield |
|---|---|
| With antimony pentafluoride; trifluoroacetic acid at 50℃; for 11h; | 76% |
| With carbon monoxide; fluorosulfonylchloride; antimony pentafluoride at 80℃; under 15200 Torr; for 24h; | 60% |
| With carbon monoxide; fluorosulfonylchloride; antimony pentafluoride at 80℃; under 15200 Torr; for 24h; Product distribution; -80 - -70deg C as temper. for 2 h.; | 60% |
| With sulfuric acid | |
| With water; antimony pentafluoride; silica gel 1.) 70 deg C, 5 h; Yield given. Multistep reaction; |
-
-
22236-19-7
pentafluorobenzoyl peroxide

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
98040-93-8
tert-butyl 2,3,4,5,6-pentafluorobenzoate

| Conditions | Yield |
|---|---|
| In various solvent(s) at 20℃; Product distribution; Oxidation; | A 75% B 24% C 31% D 69% |
| In various solvent(s) at 20℃; Oxidation; | A 75% B 24% C 31% D 69% |


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; acetic acid In dichloromethane at 20℃; for 5h; | A 58% B 70% |
-
-
22236-19-7
pentafluorobenzoyl peroxide

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
98040-93-8
tert-butyl 2,3,4,5,6-pentafluorobenzoate

| Conditions | Yield |
|---|---|
| In various solvent(s) at 20℃; Product distribution; Oxidation; | A 70% B 30% C 33% D 66% |

-
-
22236-19-7
pentafluorobenzoyl peroxide

-
-
553-97-9
2-Methyl-1,4-benzoquinone

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
98040-93-8
tert-butyl 2,3,4,5,6-pentafluorobenzoate

| Conditions | Yield |
|---|---|
| In various solvent(s) at 20℃; Oxidation; | A 70% B 30% C 33% D 66% |

-
-
124-38-9
carbon dioxide

-
-
879-05-0
2,3,4,5,6-pentafluorophenylmagnesium bromide

-
-
602-94-8
Pentafluorobenzoic acid

| Conditions | Yield |
|---|---|
| In tetrahydrofuran 0.5 hours;; | 67% |
| In tetrahydrofuran 0.5 hours;; | 67% |
-
-
124-38-9
carbon dioxide

-
-
879-06-1
pentafluorophenylmagnesium chloride

-
-
602-94-8
Pentafluorobenzoic acid

| Conditions | Yield |
|---|---|
| In tetrahydrofuran 12 hours;; | 66% |
| In tetrahydrofuran 12 hours;; | 66% |
| In tetrahydrofuran reaction in BrCH2CH2Br, then in THF at 20°C, 1.5 hours;; | 41% |
| In tetrahydrofuran reaction in BrCH2CH2Br, then in THF at 20°C, 1.5 hours;; | 41% |
| In tetrahydrofuran reaction in BrCH2CH2Br, then in THF at 20°C, 1.5 hours;; | 41% |
-
-
344-07-0
1-chloro-2,3,4,5,6-pentafluorobenzene

-
-
124-38-9
carbon dioxide

-
-
602-94-8
Pentafluorobenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-chloro-2,3,4,5,6-pentafluorobenzene With magnesium In tetrahydrofuran at 47 - 65℃; for 1.5h; Stage #2: carbon dioxide In tetrahydrofuran at -10 - 20℃; for 0.416667h; | 65% |
| Stage #1: 1-chloro-2,3,4,5,6-pentafluorobenzene With magnesium In tetrahydrofuran at 10℃; Stage #2: carbon dioxide In tetrahydrofuran at -10 - 20℃; under 1137.76 - 1965.21 Torr; for 0.25 - 0.533333h; | 92 - 95 %Spectr. |
-
-
853-75-8
N-pentafluorophenyl-pentafluorophenylaldimine

-
-
407-25-0
trifluoroacetic anhydride

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
653-27-0
α,α,α,2,3,4,5,6-octafluoroacetanilide

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In dichloromethane for 10h; | A 59% B 57% |

-
-
2674-32-0
1,4-dimethoxy-2,5-dimethylbenzene

-
-
22236-19-7
pentafluorobenzoyl peroxide

-
A
-
602-94-8
Pentafluorobenzoic acid

| Conditions | Yield |
|---|---|
| In 1,1,2-Trichloro-1,2,2-trifluoroethane at 25℃; Product distribution; Rate constant; Mechanism; | A 53% B n/a C n/a |
| In 1,1,2-Trichloro-1,2,2-trifluoroethane at 25℃; Yield given. Yields of byproduct given; |

-
-
7323-63-9
1,4-di-tert-butyl-2,5-dimethoxybenzene

-
-
22236-19-7
pentafluorobenzoyl peroxide

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
98040-93-8
tert-butyl 2,3,4,5,6-pentafluorobenzoate

-
C
-
98040-90-5
2,3,4,5,6-Pentafluoro-benzoic acid 4-tert-butyl-2,5-dimethoxy-phenyl ester

-
E
-
98040-88-1
C22H8F10O6

| Conditions | Yield |
|---|---|
| In 1,1,2-Trichloro-1,2,2-trifluoroethane at 25℃; Product distribution; Rate constant; Mechanism; | A 53% B 40% C n/a D n/a E n/a |

-
-
67899-41-6, 83976-95-8
1,2,3,4,5-pentafluoro-6-(perfluoroallyl)benzene

-
A
-
602-94-8
Pentafluorobenzoic acid

-
B
-
91407-89-5
heptafluorobenzeneacetic acid

| Conditions | Yield |
|---|---|
| With (iron phthalocyanine)(μ-nitrido)(iron octapropylporphyrazine); dihydrogen peroxide In [D3]acetonitrile at 60℃; for 6h; Reagent/catalyst; | A 6% B 37% |
-
-
80684-62-4
F-benzohydroxamic acid

-
A
-
771-60-8
2,3,4,5,6-pentafluoroaniline

-
B
-
602-94-8
Pentafluorobenzoic acid

-
C
-
80684-66-8
1,2-(F-benz)isoxazol-3(2H)-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In water for 10h; Heating; | A 11% B 5% C 15% |


| Conditions | Yield |
|---|---|
| With fluorine In sulfuric acid Ambient temperature; | 5% |

| Conditions | Yield |
|---|---|
| With thionyl chloride for 2h; Reflux; | 100% |
| With thionyl chloride for 3h; Reflux; | 99% |
| With phosphorus pentachloride | 78% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride | 100% |
| With thionyl chloride at 0℃; for 12h; Reflux; | 88% |
| With thionyl chloride for 1h; Heating; |
-
-
176763-62-5
(1R,2R)-(-)-N,N'-bis(3,5-di-tert-butylsalicydene)-1,2-cyclohexanediaminocobalt(II)

-
-
602-94-8
Pentafluorobenzoic acid

| Conditions | Yield |
|---|---|
| In toluene at 22℃; for 12h; | 100% |
| In toluene (N2); std. Schlenk technique; toluene was added to mixt. of Co complex and acid; stirred in air at 22°C for 12 h; evapd.; pentane added; filtered; dried (vac.); |

| Conditions | Yield |
|---|---|
| With water In aq. phosphate buffer at 25℃; for 15h; pH=2.7 - 3.2; Electrochemical reaction; | 100% |

| Conditions | Yield |
|---|---|
| With water In ethanol at 0 - 20℃; for 0.0833333h; | 100% |

| Conditions | Yield |
|---|---|
| With triethylamine In toluene at 100 - 110℃; for 24h; Solvent; Temperature; | 99% |
| With ammonia; copper In water at 180℃; for 0.25h; Temperature; Reagent/catalyst; | 94.6% |
| In N,N-dimethyl-formamide at 160℃; for 2h; | 94.4% |
-
-
602-94-8
Pentafluorobenzoic acid

-
-
100-51-6
benzyl alcohol

-
-
104856-46-4
benzyl 2,3,4,5,6-pentafluorobenzoate

| Conditions | Yield |
|---|---|
| With [Cl(C6F13C2H4)2SnOSn(C2H4C6F13)2Cl]2 In various solvent(s) at 150℃; for 10h; | 99% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 3h; | 78% |
-
-
602-94-8
Pentafluorobenzoic acid

-
-
5970-45-6
zinc(II) acetate dihydrate

-
-
15967-65-4
zinc pentafluorobenzoate

| Conditions | Yield |
|---|---|
| at 150℃; for 1.5h; | 99% |
-
-
602-94-8
Pentafluorobenzoic acid

-
-
58521-27-0
pentafluorophenyl potassium carboxylate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In ethanol at 20℃; for 1.5h; | 99% |
| With potassium tert-butylate In ethanol at 20℃; for 1h; Inert atmosphere; | 99% |
| With potassium tert-butylate In ethanol at 20℃; for 3h; Inert atmosphere; | |
| With potassium tert-butylate In ethanol for 3h; Inert atmosphere; | |
| With potassium tert-butylate In ethanol at 20℃; for 1.5h; |

| Conditions | Yield |
|---|---|
| With air In dichloromethane at 20℃; Irradiation; | 99% |

| Conditions | Yield |
|---|---|
| In diethyl ether at 20℃; for 24h; | 99% |

-
-
602-94-8
Pentafluorobenzoic acid

-
-
2170-05-0
pentaphenylantimony

-
-
360572-77-6
tetraphenylantimony pentafluorobenzoate

| Conditions | Yield |
|---|---|
| In toluene heating at 90 °C for 1 h; solvent was removed, dry residue was recrystd. from a toluene-heptane (1:1) mixt.; | 98% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In diethyl ether; water byproducts: H2O; react. of antimony compd. and acid with aq. H2O2 at room temp. in ether; elem. anal.; | 98% |
-
-
2170-05-0
pentaphenylantimony

-
-
602-94-8
Pentafluorobenzoic acid

-
-
360572-77-6
tetraphenylantimony pentafluorobenzoate

| Conditions | Yield |
|---|---|
| In toluene heating a mixt. of antimony compd. and acid in toluene at 90°C for 1 h; evapn., crystn. (toluene/heptane 1:1); elem. anal.; | 98% |

| Conditions | Yield |
|---|---|
| With H2O2 In diethyl ether (C6F5)COOH and 30% aq. H2O2 added to soln. of (p-tolyl)3Sb in ether; kept at 20°C for 12 h; crystals filtered off; dried; elem. anal.; | 98% |
-
-
602-94-8
Pentafluorobenzoic acid

-
-
126747-14-6
4-cyanophenylboronic acid

| Conditions | Yield |
|---|---|
| With palladium(II) trifluoroacetate; silver carbonate In dimethyl sulfoxide at 120℃; for 2h; Suzuki coupling; | 98% |

| Conditions | Yield |
|---|---|
| With C58H34F18O6P2Ru; silver trifluoromethanesulfonate In toluene at 70℃; for 1h; Inert atmosphere; Schlenk technique; regioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With C58H34F18O6P2Ru; silver trifluoromethanesulfonate In toluene at 25℃; for 4h; Inert atmosphere; Schlenk technique; regioselective reaction; | 98% |
-
-
602-94-8
Pentafluorobenzoic acid

-
-
108-24-7
acetic anhydride

-
-
15989-99-8
2,3,4,5,6-pentafluorobenzoic anhydride

| Conditions | Yield |
|---|---|
| at 128℃; for 0.0833333h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| In chlorobenzene for 24h; Heating; | 97% |

-
-
602-94-8
Pentafluorobenzoic acid

-
-
603-36-1
triphenylantimony

-
-
360572-79-8
triphenylantimony bis(pentafluorobenzoate)

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In diethyl ether; water byproducts: H2O; addn. of acid and aq. H2O2 to a soln. of antimony compd. in ether, keeping for 12 h at 20°C; filtration, drying; elem. anal.; | 97% |
| With H2O2 In diethyl ether (C6F5)COOH and 30% aq. H2O2 added to soln. of Ph3Sb in ether; kept at 20°C for 12 h; crystals filtered off; dried; elem. anal.; | 97% |
-
-
602-94-8
Pentafluorobenzoic acid

-
-
33756-42-2
tri-p-fluorophenylstibine

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In diethyl ether; water at 20℃; for 24h; | 97% |
| With tert.-butylhydroperoxide In diethyl ether at 24℃; for 18h; | 94% |
-
-
602-94-8
Pentafluorobenzoic acid

-
-
71-48-7, 917-69-1, 5931-89-5, 55881-15-7, 68931-68-0, 68931-69-1, 93029-27-7
cobalt(II) acetate

| Conditions | Yield |
|---|---|
| Stage #1: C105H138N6O12; cobalt(II) acetate In toluene at 20℃; for 5h; Schlenk technique; Inert atmosphere; Stage #2: Pentafluorobenzoic acid In dichloromethane at 20℃; for 5h; | 97% |


| Conditions | Yield |
|---|---|
| With sulfuric acid In tetrachloromethane Heating; | 96% |
| With thionyl chloride at 20 - 50℃; for 21h; Reflux; Large scale; | 91% |
| With sulfuric acid Reflux; | 75% |

-
-
602-94-8
Pentafluorobenzoic acid

| Conditions | Yield |
|---|---|
| In toluene byproducts: H2O, CO2; N2 or air, 1:1 mixt. refluxed at 110°C for 6 h; dried (vac.); | 96% |
-
-
602-94-8
Pentafluorobenzoic acid

-
-
71-48-7, 917-69-1, 5931-89-5, 55881-15-7, 68931-68-0, 68931-69-1, 93029-27-7
cobalt(II) acetate

| Conditions | Yield |
|---|---|
| Stage #1: C72H94N4O8; cobalt(II) acetate In toluene at 20℃; for 5h; Schlenk technique; Inert atmosphere; Stage #2: Pentafluorobenzoic acid In dichloromethane at 20℃; for 5h; | 96% |

Pentafluorobenzoic acid Consensus Reports
Pentafluorobenzoic acid Specification
Pentafluorobenzoic acid is an organic compound with the formula C7HF5O2, and its systematic name is the same with the product name. With the CAS registry number 602-94-8, it is also named as 2,3,4,5,6-Pentafluorobenzoic acid. It belongs to the product categories of Benzoic acid; Fluorobenzene; C7; Carbonyl Compounds; Carboxylic Acids; Organofluorine compounds. Its EINECS number is 210-026-6. In addition, the molecular weight is 212.07. Its classification code is Drug / Therapeutic Agent. It is stable at common pressure and temperature, and it should be sealed and stored in a cool and dry place. Moreover, it should be protected from oxides. This chemical is used as a pharmaceutical intermediate.
Physical properties of Pentafluorobenzoic acid are: (1)ACD/LogP: 2.635; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.44; (4)ACD/LogD (pH 7.4): -0.51; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 1.00; (8)ACD/KOC (pH 7.4): 1.00; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 37.3 Å2; (13)Index of Refraction: 1.451; (14)Molar Refractivity: 33.152 cm3; (15)Molar Volume: 123.011 cm3; (16)Polarizability: 13.142×10-24cm3; (17)Surface Tension: 37.81 dyne/cm; (18)Density: 1.724 g/cm3; (19)Flash Point: 86.862 °C; (20)Enthalpy of Vaporization: 48.245 kJ/mol; (21)Boiling Point: 220.014 °C at 760 mmHg; (22)Vapour Pressure: 0.07 mmHg at 25°C.
Preparation of Pentafluorobenzoic acid: this chemical can be prepared by 2,3,4,5,6-pentafluorobenzylidene aniline. This reaction will need reagents 90% H2O2, formic acid with the reaction time of 5 hours. The yield is about 89%.
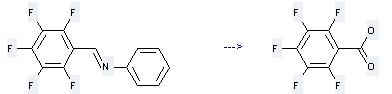
Uses of Pentafluorobenzoic acid: it can be used to produce methyl pentafluorobenzoate by heating. It will need reagent conc. H2SO4 and solvent CCl4. The yield is about 96%.

When you are using this chemical, please be cautious about it as the following:
This chemical is toxic if swallowed. It is irritating to eyes, respiratory system and skin. When using it, you must avoid contact with skins and eyes. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need to wear suitable protective clothing, gloves and eye/face protection. In case of accident or if you feel unwell, you must seek medical advice immediately (show the label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: Fc1c(c(F)c(F)c(F)c1F)C(=O)O
(2)Std. InChI: InChI=1S/C7HF5O2/c8-2-1(7(13)14)3(9)5(11)6(12)4(2)10/h(H,13,14)
(3)Std. InChIKey: YZERDTREOUSUHF-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 178mg/kg (178mg/kg) | BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) BEHAVIORAL: REGIDITY | Journal of Medicinal Chemistry. Vol. 11, Pg. 1020, 1968. |
Related Products
- Pentafluorobenzoic acid
- 60295-14-9
- 60296-02-8
- 60-29-7
- 60299-11-8
- 603-00-9
- 6030-36-0
- 60303-68-6
- 60304-36-1
- 60308-49-8
- 603107-76-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View