-
Name
glyceric acid
- EINECS 207-472-9
- CAS No. 473-81-4
- Article Data306
- CAS DataBase
- Density 1.558 g/cm3
- Solubility
- Melting Point <25 °C
- Formula C3H6O4
- Boiling Point 412 °C at 760 mmHg
- Molecular Weight 106.078
- Flash Point 217.1 °C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms glyceric acid;Propanoic acid, 2,3-dihydroxy-;C00258;DL-Glyceric Acid (20% in Water, ca. 2Mol/L);(20% in Water, ca. 2Mol/L);DL-Glyceric Acid
- PSA 77.76000
- LogP -1.57580
Synthetic route

| Conditions | Yield |
|---|---|
| With 1 wt% Au/TiO2; oxygen In water at 75℃; under 39547.2 Torr; for 3022h; Time; Flow reactor; | 100% |
| With sodium chlorite; dimethyl sulfoxide In aq. phosphate buffer at 0 - 20℃; pH=4; | 93% |
| With iodine; sodium carbonate |

| Conditions | Yield |
|---|---|
| Stage #1: glycerol With Rh(OTf)(trop2NH)(PPh3); water; cyclohexanone; sodium hydroxide at 20℃; for 8h; Inert atmosphere; Stage #2: With hydrogenchloride In water Inert atmosphere; chemoselective reaction; | 98% |
| With oxygen In water at 60℃; under 7500.75 Torr; Reagent/catalyst; | 75% |
| With water; oxygen In water at 24.84℃; under 760.051 Torr; for 6h; Catalytic behavior; Schlenk technique; | 42% |

| Conditions | Yield |
|---|---|
| Stage #1: oxiranyl-methanol With Rh(OTf)(trop2NH)(PPh3); water; cyclohexanone; sodium hydroxide at 20℃; for 8h; Inert atmosphere; Stage #2: With hydrogenchloride In water Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium tetrahydroborate In water for 4h; Product distribution; pH=10.8-11.0; | A n/a B 90% |

| Conditions | Yield |
|---|---|
| With oxygen In water at 60℃; Catalytic behavior; Reagent/catalyst; Autoclave; chemoselective reaction; | A 8.9% B 89.9% C 6.8% |
| With oxygen In water at 80℃; under 7500.75 Torr; for 2h; pH=6.7; Reagent/catalyst; Autoclave; | |
| With oxygen In water at 60℃; under 3750.38 Torr; for 24h; Catalytic behavior; Kinetics; Reagent/catalyst; Time; High pressure; | A 7.8 %Chromat. B 55.4 %Chromat. C 7.7 %Chromat. |


| Conditions | Yield |
|---|---|
| With oxygen In water at 60℃; Catalytic behavior; Reagent/catalyst; Autoclave; chemoselective reaction; | A n/a B n/a C 84.9% D 6.5% |


| Conditions | Yield |
|---|---|
| With oxygen In water at 60℃; Catalytic behavior; Reagent/catalyst; Autoclave; chemoselective reaction; | A 84.2% B 9.5% C 6.3% |
| With oxygen In water at 60℃; under 760.051 Torr; for 4h; Catalytic behavior; | |
| With Pt-MCM-41 catalyst; oxygen In water at 69.84℃; under 760.051 Torr; pH=Ca. 7; Kinetics; Catalytic behavior; Reagent/catalyst; Temperature; |


| Conditions | Yield |
|---|---|
| With perchloric acid In ethanol at 20℃; pH=1; Concentration; Reagent/catalyst; Electrochemical reaction; | A 9% B 70.6% |
| With Pseudomonas putida HK-5 pyrroloquinoline quinone-dependent alcohol dehydrogenase; 4,4'-azobis(1-methylpyridinium) bis(methyl sulfate); NADH In aq. buffer for 6h; pH=7.1; Catalytic behavior; Time; Electrolysis; Enzymatic reaction; | A 53% B 15% |
| With silica-supported platinum; oxygen In water at 89.84℃; for 24h; |

| Conditions | Yield |
|---|---|
| With water; oxygen In water at 24.84℃; under 760.051 Torr; for 6h; Catalytic behavior; Schlenk technique; | A 10% B 54% C 5% |
| With oxygen; sodium hydroxide In water at 59.84℃; under 760.051 Torr; for 7h; Product distribution / selectivity; Closed type batch reactor; | A n/a B 70 %Chromat. C n/a |
| With oxygen; sodium hydroxide In water at 60℃; under 2250.23 Torr; for 24h; |


| Conditions | Yield |
|---|---|
| With oxygen; sodium hydroxide In water at 70℃; pH=3.56; Temperature; pH-value; Flow reactor; | A 38% B 47.3% |
| With oxygen; sodium hydroxide In water at 89.84℃; under 760.051 Torr; | |
| With sodium hydroxide at 90℃; under 760.051 Torr; Reagent/catalyst; | |
| With oxygen; sodium hydroxide In water at 50℃; under 2250.23 Torr; Catalytic behavior; | |
| With oxygen; sodium hydroxide at 110℃; under 2250.23 Torr; for 0.5h; Reagent/catalyst; |

| Conditions | Yield |
|---|---|
| With oxygen; sodium hydroxide In water at 85℃; pH=3.56; Temperature; pH-value; Flow reactor; | A 46.5% B 5.5% C 38.1% |
| With oxygen; sodium hydroxide In water at 50℃; under 2250.23 Torr; for 4h; Catalytic behavior; | |
| With lithium hydroxide monohydrate at 90℃; under 760.051 Torr; for 6h; Catalytic behavior; Inert atmosphere; |

-
-
56-81-5
glycerol

-
A
-
849585-22-4
LACTIC ACID

-
B
-
473-81-4
glyceric acid

-
C
-
96-26-4
dihydroxyacetone

-
D
-
64-19-7
acetic acid

-
E
-
56-82-6
Glyceraldehyde

| Conditions | Yield |
|---|---|
| With oxygen In water at 60℃; under 3750.38 Torr; for 5h; Catalytic behavior; Autoclave; | A 45% B n/a C n/a D n/a E n/a |

-
-
56-81-5
glycerol

-
A
-
79-14-1
glycolic Acid

-
B
-
80-69-3
tartronic acid

-
C
-
473-81-4
glyceric acid

-
D
-
144-62-7
oxalic acid

| Conditions | Yield |
|---|---|
| With water; oxygen In water at 24.84℃; under 760.051 Torr; for 6h; Catalytic behavior; Reagent/catalyst; Concentration; Schlenk technique; | A 11% B 13% C 42% D 12% |
| With potassium carbonate In water at 80℃; for 24h; | |
| With water; oxygen; sodium hydroxide at 50℃; under 2280.15 Torr; Reagent/catalyst; |

-
-
107-18-6
allyl alcohol

-
A
-
473-81-4
glyceric acid

-
B
-
79-10-7
acrylic acid

-
C
-
503-66-2
3-hydroxypropionic acid

| Conditions | Yield |
|---|---|
| With oxygen; sodium hydroxide In water at 50℃; Reagent/catalyst; Sonication; | A 6.9% B 30.1% C 7.2% |

-
-
87-72-9, 608-45-7, 608-46-8, 608-47-9, 2460-44-8, 6748-95-4, 6763-34-4, 7261-26-9, 7283-06-9, 7283-07-0, 7296-55-1, 7296-56-2, 7296-58-4, 7296-59-5, 7296-60-8, 7296-61-9, 7296-62-0, 7322-30-7, 10257-31-5, 10257-32-6, 10257-33-7, 10257-34-8, 10257-35-9, 19982-83-3, 20242-88-0, 28697-53-2, 36562-42-2, 41546-41-2, 89299-64-9, 107655-34-5, 115794-06-4, 115794-07-5, 130550-15-1, 130606-21-2
D-lyxose

-
A
-
64-18-6
formic acid

-
B
-
79-14-1
glycolic Acid

-
C
-
473-81-4
glyceric acid

-
D
-
124-38-9
carbon dioxide

-
E
-
10191-35-2
2,3,4-Trihydroxybutanoic acid

| Conditions | Yield |
|---|---|
| With copper(II) sulfate at 100℃; Product distribution; Rate constant; Thermodynamic data; study of the oxidation reaction of D-lyxose by cupric ion, kinetic mesurements, ΔS(excit.),; | A 14% B 30% C 22% D 15% E 12% |


| Conditions | Yield |
|---|---|
| With oxygen In water at 160℃; under 13680.9 Torr; for 20h; Temperature; Pressure; Reagent/catalyst; Green chemistry; | A 29.3% B 17.8% |
| With oxygen In water at 160℃; under 13680.9 Torr; for 20h; |

| Conditions | Yield |
|---|---|
| With cerium(IV) oxide Reagent/catalyst; | A 19% B 23.2% |
| With nano gold salt supported on cerium oxide | A 10.7% B 9.4% |

| Conditions | Yield |
|---|---|
| With perchloric acid In ethanol at 20℃; pH=1; Reagent/catalyst; Electrochemical reaction; | A 7% B 7.6% C 20.3% |


| Conditions | Yield |
|---|---|
| With carbon dioxide; hydrogen; oxygen In water at 30℃; under 8274.59 Torr; for 6h; Catalytic behavior; Reagent/catalyst; Time; Autoclave; | A 14% B 10% |

-
A
-
849585-22-4
LACTIC ACID

-
B
-
57-55-6
propylene glycol

-
C
-
473-81-4
glyceric acid

-
D
-
50-70-4
D-sorbitol

-
E
-
107-21-1
ethylene glycol

| Conditions | Yield |
|---|---|
| With hydrogen In water at 240℃; under 30003 Torr; for 1h; | A 12.6% B 11.6% C 10.2% D 10.1% E 9.1% |


| Conditions | Yield |
|---|---|
| With copper(II) hydroxide; sodium hydroxide |

| Conditions | Yield |
|---|---|
| With copper(II) hydroxide; sodium hydroxide |

| Conditions | Yield |
|---|---|
| im Tierkoerper; | |
| With cis-nitrous acid |

| Conditions | Yield |
|---|---|
| With cis-nitrous acid | |
| With hydrogenchloride; sodium nitrite In water at 0 - 20℃; for 17.0833h; Schlenk technique; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With cis-nitrous acid at 40 - 50℃; |

| Conditions | Yield |
|---|---|
| With water; silver(l) oxide |

| Conditions | Yield |
|---|---|
| With copper(II) hydroxide; sodium hydroxide | |
| With air in alkalischer Loesungen; | |
| With dihydrogen peroxide in alkalischer Loesungen; |

| Conditions | Yield |
|---|---|
| With copper(II) hydroxide; sodium hydroxide | |
| With air in alkalischer Loesungen; | |
| With dihydrogen peroxide in alkalischer Loesungen; |

| Conditions | Yield |
|---|---|
| With Fehling's solution | |
| With air in alkalischer Loesungen; | |
| With dihydrogen peroxide in alkalischer Loesungen; |
-
-
67525-74-0
(+/-)-calcium glycerate dihydrate

-
-
473-81-4
glyceric acid

| Conditions | Yield |
|---|---|
| Gaerung durch den Bacillus aethaceticus; |

| Conditions | Yield |
|---|---|
| With hydrogen iodide; hydrogen; palladium dichloride at 100℃; under 20686.5 Torr; for 10h; Reagent/catalyst; Temperature; Sealed tube; | 99% |
| With hydrogen iodide; hydrogen; palladium dichloride In water at 100℃; under 20686.5 Torr; for 10h; Pressure; Reagent/catalyst; Temperature; | 99% |
| With hydrogen iodide; hydrogen; palladium dichloride at 170℃; under 155149 Torr; for 25h; Temperature; Pressure; Sealed tube; | 99.9% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In water for 12h; Inert atmosphere; Reflux; | 99% |
| With boron trifluoride methanol complex for 1h; Heating; | 75.7% |
| at 180℃; methyl ester of/the/ d-glyceric acid; |
-
-
473-81-4
glyceric acid

-
-
129867-48-7
mono(6-amino-6-deoxy)-per(2,3,6-O-methyl)-β-cyclodextrin

-
-
1407657-67-3
6I-(2,3-dihydroxypropionamido)-6I-deoxy-2I,3I-di-O-methylhexakis(2II-VII,3II-VII,6II-VII-tri-O-methyl)cyclomaltoheptaose

| Conditions | Yield |
|---|---|
| Stage #1: glyceric acid With benzotriazol-1-ol; diisopropyl-carbodiimide In N,N-dimethyl-formamide at 20℃; for 1.5h; Inert atmosphere; Stage #2: mono(6-amino-6-deoxy)-per(2,3,6-O-methyl)-β-cyclodextrin With triethylamine In chloroform; N,N-dimethyl-formamide at 20℃; for 2.5h; Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| With oxygen In water at 60℃; under 2250.23 Torr; for 9h; Mechanism; Autoclave; | 75% |
-
-
473-81-4
glyceric acid

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In N,N-dimethyl-formamide at 20℃; | 67% |

| Conditions | Yield |
|---|---|
| With aluminium(III) triflate; dihydrogen peroxide In acetonitrile at 70℃; for 12h; | A 57.8% B 8.1% |

| Conditions | Yield |
|---|---|
| With rhodium on carbon; hydrogen iodide; hydrogen; iodine In water at 99.84℃; under 20686.5 Torr; for 1h; Reagent/catalyst; Temperature; Autoclave; | A 52% B 14% |

| Conditions | Yield |
|---|---|
| Stage #1: glyceric acid; 3,3'-((2,2'-dichloro-[1,1'-biphenyl]-3,3'-diyl)bis(oxy))bis(propan-1-amine) With 4-methyl-morpholine; 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In water; N,N-dimethyl-formamide at 20℃; for 18h; Stage #2: trifluoroacetic acid In water; acetonitrile | 50% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 48h; | 46% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride Heating; | 32% |
| at 180 - 190℃; ethyl ester of/the/ d-glyceric acid; | |
| at 190℃; Man fraktionert das Produkt im Vakuum; | |
| at 190℃; |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; potassium hydroxide In water at 20℃; pH=6 - 7; | 32% |
-
-
473-81-4
glyceric acid

-
-
1207632-14-1
6-(3-fluoro-4-methoxybenzyl)-2,3,4,7-tetrahydro-1H-indolo[2,3-c][1,7]naphthyridine

-
-
1207633-34-8
1-[6-(3-fluoro-4-methoxybenzyl)-1,2,4,7-tetrahydro-3H-indolo[2,3-c][1,7]naphthyridin-3-yl]-2,3-dihydroxypropan-1-one

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane; water at 20℃; for 18h; Molecular sieve; | 25% |
Propanoic acid,2,3-dihydroxy- Specification
The Propanoic acid,2,3-dihydroxy-, with the CAS registry number 473-81-4, is also known as Glycerolic acid. Its EINECS number is 207-472-9. This chemical's molecular formula is C3H6O4 and molecular weight is 106.08. What's more, its systematic name is 2,3-dihydroxypropanoic acid.
Physical properties of Propanoic acid,2,3-dihydroxy- are: (1)ACD/LogP: -1.93; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -4.51; (4)ACD/LogD (pH 7.4): -5.61; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 4; (10)#H bond donors: 3; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 44.76 Å2; (13)Index of Refraction: 1.515; (14)Molar Refractivity: 20.54 cm3; (15)Molar Volume: 68 cm3; (16)Polarizability: 8.14×10-24cm3; (17)Surface Tension: 80.9 dyne/cm; (18)Density: 1.558 g/cm3; (19)Flash Point: 217.1 °C; (20)Enthalpy of Vaporization: 76.78 kJ/mol; (21)Boiling Point: 412 °C at 760 mmHg; (22)Vapour Pressure: 1.65E-08 mmHg at 25°C.
Preparation of Propanoic acid,2,3-dihydroxy-: this chemical can be prepared by 2-acetylamino-2,3-dibromo-propionic acid at the pH value of 10.8-11.0. This reaction will need reagents 0.4 M sodium borohydride, 0.1 M sodium hydroxide and solvent H2O with the reaction time of 4 hours. The yield is about 90%.
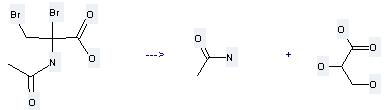
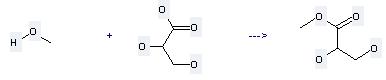
Uses of Propanoic acid,2,3-dihydroxy-: it can be used to produce 2,3-dihydroxy-propionic acid methyl ester by heating. It will need reagent 14% borontrifluoride-methanol complex with the reaction time of 1 hour. The yield is about 75.7%.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)C(O)CO
(2)InChI: InChI=1S/C3H6O4/c4-1-2(5)3(6)7/h2,4-5H,1H2,(H,6,7)
(3)InChIKey: RBNPOMFGQQGHHO-UHFFFAOYSA-N
Related Products
- Propanoic acid, 1-methylpropyl ester
- Propanoic acid, 2-(2,4-dichlorophenoxy)-, magnesium salt (2:1)
- Propanoic acid, 2-(2,4-dichlorophenoxy)-, mixt. with (2,4-dichlorophenoxy)acetic acid, (2,4,5-trichlorophenoxy)acetic acid and 2-(2,4,5-trichlorophenoxy)propanoic acid
- Propanoic acid, 2-[4-(trifluoromethoxy)phenoxy]-
- Propanoic acid, 2-bromo-, ethyl ester, (2S)-
- Propanoic acid, 2-chloro-3-methoxy-
- Propanoic acid, 2-hydroxy-, decylester
- Propanoic acid, 2-hydroxy-3-(phenylmethoxy)-, methyl ester, (2R)-
- Propanoic acid, 2-iodo-2-methyl-, methyl ester
- Propanoic acid, 2-methoxy-, ethyl ester, (2R)-
- 473838-96-9
- 473-86-9
- 473902-31-7
- 473914-76-0
- 473918-41-1
- 473923-97-6
- 473927-64-9
- 473927-69-4
- 473981-11-2
- 473-98-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View