-
Name
Propoxybenzene
- EINECS 210-756-5
- CAS No. 622-85-5
- Article Data80
- CAS DataBase
- Density 0.929g/cm3
- Solubility
- Melting Point -28°C(lit.)
- Formula C9H12 O
- Boiling Point 189.9°C at 760 mmHg
- Molecular Weight 136.194
- Flash Point 65°C
- Transport Information
- Appearance
- Safety Moderately toxic by ingestion. When heated to decomposition it emits acrid smoke and irritating fumes. See also ETHERS.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Ether, phenylpropyl (6CI,7CI,8CI); 1-Phenoxypropane; Phenyl propyl ether; Propoxybenzene;Propyl phenyl ether; Propyloxybenzene
- PSA 9.23000
- LogP 2.47540
Synthetic route

| Conditions | Yield |
|---|---|
| With C7H14N3(1+)*Cl(1-); sodium triethylborohydride; cobalt(II) chloride In tetrahydrofuran at 25℃; under 7500.75 Torr; for 16h; | 95% |
| With sodium tetrahydroborate; ruthenium trichloride In tetrahydrofuran; water at 20℃; for 1h; | 90% |
| With 10% Pd on charcoal; diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate In ethanol for 4h; Reflux; | 88% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; DCH-18-crown-6 immobilized on 2-vinylpyridine-styrene copolymer In chloroform for 6h; Heating; | 90% |
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 5h; | 1.29 g |

| Conditions | Yield |
|---|---|
| With caesium carbonate; [Cu8(S2P(OiPr)2)6(μ8-Cl)][PF6] at 110℃; for 11h; | 90% |
-
-
143885-02-3
propyloxy(diphenyl)-λ6-sulfanenitrile

-
-
108-95-2
phenol

-
A
-
622-85-5
propoxybenzene

-
B
-
22731-83-5
S,S-diphenylsulphoximine

| Conditions | Yield |
|---|---|
| In chloroform-d1 at 20℃; for 336h; | A 89% B n/a |


| Conditions | Yield |
|---|---|
| With [2-(di-tert-butylphosphinomethyl)-6-(diethylaminomethyl)pyridine]ruthenium(II) chlorocarbonyl hydride; isopropyl alcohol; sodium t-butanolate at 100℃; for 18h; Reagent/catalyst; Inert atmosphere; Sealed tube; Green chemistry; | 88% |
| With cetyltrimethylammonium bromide micelle; zinc In water Heating; | 83% |
| With tris-(trimethylsilyl)silane; oxygen at 60℃; for 6h; in sealed vial; | 80 % Chromat. |
-
-
118527-13-2
n-propoxy-tris(dimethylamino)phosphonium hexafluorophosphate

-
-
108-95-2
phenol

-
-
622-85-5
propoxybenzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide In N,N-dimethyl-formamide for 15h; Heating; | 86% |

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); tri-n-butyl-tin hydride In benzene for 1h; Mechanism; Heating; other aryl ethers; | A 85% B 2% |
| With 2,2'-azobis(isobutyronitrile); tri-n-butyl-tin hydride In benzene for 1h; Heating; | A 85% B 2% |

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate; iodine for 8h; Heating; other alkanol; | 81% |
| With ammonium cerium(IV) nitrate; iodine for 8h; Heating; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: potassium phenyltrifluoborate With dmap; copper diacetate; 4 A molecular sieve In dichloromethane at 20℃; for 0.0833333h; Stage #2: propan-1-ol With oxygen In dichloromethane at 20℃; for 24h; | 78% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 5h; | 72% |
| With potassium carbonate In ethanol at 140℃; | 62% |
| With sodium hydroxide; cetyltrimethylammonim bromide 1.) 60 deg C, 0.5 h 2.) 80 deg C, 5 h; Yield given. Multistep reaction; | |
| With sodium hydroxide; ethyltriphenylphosphonium bromide In water at 22℃; for 50h; | 7.0 %Chromat. |
-
-
87841-77-8
2-(2-Phenoxy-ethylazo)-prop-2-yl-hydroperoxide

-
-
109-92-2
ethyl vinyl ether

-
A
-
493-08-3
chromane

-
B
-
122-99-6
2-Phenoxyethanol

-
C
-
622-85-5
propoxybenzene

-
D
-
19790-62-6
4-phenoxybutyraldehyde

-
E
-
87841-94-9
2-phenoxypentanal

-
F
-
103-73-1
Phenetole

| Conditions | Yield |
|---|---|
| at 50℃; Mechanism; | A n/a B 1% C 0.6% D 52% E 0.7% F 7% G n/a |


| Conditions | Yield |
|---|---|
| With Co(dmgBF2)2(CH3CN)2; 3-cyano-N-methyl-quinolinium perchlorate In acetonitrile at 20℃; for 5h; Sealed tube; Inert atmosphere; Irradiation; High pressure; | 41% |
-
-
71-23-8
propan-1-ol

-
-
108-86-1
bromobenzene

-
-
6819-41-6
sodium n-propoxide

-
-
142-71-2
copper diacetate

-
-
622-85-5
propoxybenzene


| Conditions | Yield |
|---|---|
| beim Behandeln von Benzoldiazoniumchlorid; |

| Conditions | Yield |
|---|---|
| With carbon monoxide at 180℃; |

| Conditions | Yield |
|---|---|
| With propan-1-ol; ultra-violet light; copper (I) acetate Irradiation; |

| Conditions | Yield |
|---|---|
| With toluene |

| Conditions | Yield |
|---|---|
| With ammonia | |
| With water at 80℃; |

| Conditions | Yield |
|---|---|
| With propan-1-ol; Pd-BaSO4 Hydrogenation; |
-
-
20549-68-2
1-(3-iodopropoxy)benzene

-
-
622-85-5
propoxybenzene

| Conditions | Yield |
|---|---|
| With etheric solution; sodium |

| Conditions | Yield |
|---|---|
| With ethanol |

| Conditions | Yield |
|---|---|
| at 100 - 110℃; |

| Conditions | Yield |
|---|---|
| With ethanol | |
| In tetrahydrofuran |

| Conditions | Yield |
|---|---|
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| With aluminum oxide at 380 - 400℃; unter Druck; | |
| at 420℃; beim Leiten ueber ThO2; | |
| With aluminum oxide at 450 - 500℃; |

| Conditions | Yield |
|---|---|
| With Fuller's Earth under 140 Torr; |


| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 160℃; |

| Conditions | Yield |
|---|---|
| at 100℃; for 24h; |
-
-
71-23-8
propan-1-ol

-
-
108-95-2
phenol

-
A
-
645-56-7
4-n-Propylphenol

-
B
-
644-35-9
5-propylphenol

-
C
-
622-85-5
propoxybenzene

-
D
-
6626-32-0
2,6-di-n-propylphenol

-
E
-
74663-48-2
2-(1-methylethyl)-6-propylphenol

-
F
-
74663-45-9
n-Propyl 2-n-propylphenyl ether

| Conditions | Yield |
|---|---|
| aluminum oxide at 250℃; Product distribution; Mechanism; other temperatures, other catalysts; | A 1.4 % Chromat. B 3.1 % Chromat. C 5.8 % Chromat. D 49.5 % Chromat. E 9.2 % Chromat. F 6.5 % Chromat. |

-
-
622-85-5
propoxybenzene

-
-
33382-58-0
1-chloro-4-propoxybenzene

| Conditions | Yield |
|---|---|
| With kaolin; sodium chlorite; manganese(III) acetylacetonate In dichloromethane at 25℃; for 1.33333h; | 98% |
| With aluminum oxide; sodium chlorite; (Salen)Mn(III) catalyst In dimethyl sulfoxide for 0.5h; Ambient temperature; | 96% |
| With aluminum oxide; sodium chlorite; (salen)Mn(III) In dichloromethane at 25℃; for 0.5h; | 96% |
| With aluminum oxide; sodium chlorite; water; manganese(III) acetylacetonate In dichloromethane at 25℃; for 0.75h; | 93% |
| With aluminum oxide; copper dichloride In chlorobenzene at 100℃; for 3h; | 95 % Chromat. |
-
-
622-85-5
propoxybenzene

-
-
39969-56-7
1-bromo-4-propoxybenzene

| Conditions | Yield |
|---|---|
| With aluminum oxide; sodium chlorite; water; manganese(III) acetylacetonate; sodium bromide In dichloromethane at 20℃; for 1.33333h; | 97% |
| With kaolin; sodium chlorite; manganese(III) acetylacetonate; sodium bromide In dichloromethane at 25℃; for 2.66667h; | 97% |
| With sodium chlorite; Montmorillonite K10; manganese(III) acetylacetonate; sodium bromide In dichloromethane at 25℃; for 1h; | 96% |
| With sodium chlorite; silica gel; manganese(III) acetylacetonate; sodium bromide In dichloromethane at 25℃; for 2h; | 95% |
| With carbon disulfide; bromine at 0℃; |

| Conditions | Yield |
|---|---|
| Stage #1: monomethyl oxalyl chloride With iron(III) chloride In 1,2-dichloro-ethane at 0 - 5℃; for 0.5h; Friedel-Crafts Acylation; Stage #2: propoxybenzene In dichloromethane; 1,2-dichloro-ethane at 20℃; for 3h; Friedel-Crafts Acylation; | 92% |

| Conditions | Yield |
|---|---|
| With phosphoric acid; trifluoroacetic acid at 20℃; for 0.5h; | 88% |
-
-
622-85-5
propoxybenzene

-
-
872415-81-1
chloro(5,10,15,20-tetratolylporphyrinato)carbonyliridium(III)

-
-
872415-73-1
(5,10,15,20-tetra-p-tolylporphyrinato)propyliridium(III)

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 150℃; for 2h; Inert atmosphere; Darkness; | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: dichloroacethyl chloride With aluminum (III) chloride In dichloromethane at 0℃; for 0.166667h; Friedel-Crafts Acylation; Stage #2: propoxybenzene In dichloromethane Friedel-Crafts Acylation; | 82.2% |

| Conditions | Yield |
|---|---|
| With phosphoric acid; trifluoroacetic acid at 20℃; for 0.75h; | 80% |
-
-
622-85-5
propoxybenzene

-
-
10313-60-7
3,4-dimethoxyphenylacetyl chloride

| Conditions | Yield |
|---|---|
| With aluminium trichloride In 1,2-dichloro-ethane at 40℃; | 78% |

| Conditions | Yield |
|---|---|
| With aluminium trichloride at 40℃; for 3h; | 66% |

| Conditions | Yield |
|---|---|
| With H-β zeolite In neat (no solvent) at 150℃; for 12h; regioselective reaction; | 63% |
-
-
622-85-5
propoxybenzene

-
-
872415-81-1
chloro(5,10,15,20-tetratolylporphyrinato)carbonyliridium(III)

-
-
100-66-3
methoxybenzene

-
-
386707-40-0
(5,10,15,20-tetrakis(p-tolyl)porphyrinato)(methyl)iridium(III)

-
-
872415-73-1
(5,10,15,20-tetra-p-tolylporphyrinato)propyliridium(III)

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 150℃; for 2h; Inert atmosphere; Darkness; | A 23% B 61% |

-
-
26118-57-0
2-(4-(n-propoxy)phenyl)acetic acid

-
-
622-85-5
propoxybenzene

-
-
94686-48-3
4,4'-Dipropyloxy-deoxybenzoin

| Conditions | Yield |
|---|---|
| Stage #1: 2-(4-(n-propoxy)phenyl)acetic acid With thionyl chloride In chloroform at 0℃; Stage #2: propoxybenzene With aluminum (III) chloride In dichloromethane at 0 - 20℃; Stage #3: With water In dichloromethane Cooling; | 51.86% |
-
-
622-85-5
propoxybenzene

-
-
1597-49-5
trifluoroacetyl-DL-alanine

-
-
742095-38-1
(RS)-2-trifluoroacetamido-1-(4-propoxyphenyl)-1-propanone

| Conditions | Yield |
|---|---|
| With aluminium trichloride; oxalyl dichloride | 50% |
-
-
7790-94-5
chlorosulfonic acid

-
-
622-85-5
propoxybenzene

-
-
58076-32-7
4-n-propoxy-1-benzenesulfonyl chloride

| Conditions | Yield |
|---|---|
| In dichloromethane for 2h; | 50% |
-
-
622-85-5
propoxybenzene

-
-
872415-81-1
chloro(5,10,15,20-tetratolylporphyrinato)carbonyliridium(III)

-
-
103-73-1
Phenetole

-
-
872415-72-0
(5,10,15,20-tetra-p-tolylporphyrinato)ethyliridium(III)

-
-
872415-73-1
(5,10,15,20-tetra-p-tolylporphyrinato)propyliridium(III)

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 150℃; for 2h; Inert atmosphere; Darkness; | A 25% B 46% |

-
-
622-85-5
propoxybenzene

-
-
386707-40-0
(5,10,15,20-tetrakis(p-tolyl)porphyrinato)(methyl)iridium(III)

-
-
872415-73-1
(5,10,15,20-tetra-p-tolylporphyrinato)propyliridium(III)

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 150℃; for 17h; Inert atmosphere; Darkness; | 5% |

| Conditions | Yield |
|---|---|
| at 150℃; Erwaermen des Reaktionsprodukts mit methanol. KOH; |
-
-
108-30-5
succinic acid anhydride

-
-
622-85-5
propoxybenzene

-
-
39496-82-7
4-oxo-4-(4-propoxyphenyl)butanoic acid

| Conditions | Yield |
|---|---|
| With aluminium trichloride; nitrobenzene |
Propoxybenzene Chemical Properties
IUPAC Name: Propoxybenzene
Synonyms: Phenyl propyl ether ; Benzene, propoxy- ; Ether, phenyl propyl ; Ether, propyl phenyl ; Ether,propylphenyl ; Propoxy-benzen ; Propoxy-benzene ; Propoxyphenyl
Molecular Structure of Propyl phenyl ether (CAS NO. 622-85-5) : 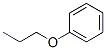
Molecular Formula of Propyl phenyl ether (CAS NO. 622-85-5) : C9H12O
Molecular Weight of Propyl phenyl ether (CAS NO. 622-85-5) : 136.19
CAS NO: 622-85-5
EINECS: 210-756-5
Index of Refraction: 1.488
Surface Tension: 30.2 dyne/cm
Density: 0.929 g/cm3
Flash Point: 65 °C
Enthalpy of Vaporization: 40.87 kJ/mol
Boiling Point: 189.9 °C at 760 mmHg
Vapour Pressure: 0.77 mmHg at 25°C
Propoxybenzene Uses
Propyl phenyl ether (CAS NO. 622-85-5) is used as intermediate in organic synthesis .
Propoxybenzene Toxicity Data With Reference
| 1. | orl-mus LD50:3400 mg/kg | JPETAB Journal of Pharmacology and Experimental Therapeutics. 88 (1946),400. |
Propoxybenzene Consensus Reports
Reported in EPA TSCA Inventory.
Propoxybenzene Safety Profile
Moderately toxic by ingestion. When heated to decomposition it emits acrid smoke and irritating fumes. See also ETHERS.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View