-
Name
Propyl gallate
- EINECS 204-498-2
- CAS No. 121-79-9
- Article Data41
- CAS DataBase
- Density 1.363 g/cm3
- Solubility ethanol: 50 mg/mL
- Melting Point 146-149 °C(lit.)
- Formula C10H12O5
- Boiling Point 448.6 °C at 760 mmHg
- Molecular Weight 212.202
- Flash Point 181.3 °C
- Transport Information
- Appearance white to light beige crystalline powder
- Safety 24-37
- Risk Codes 22-43
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms Tenox PG;Nipagallin P;Benzoic acid,3,4,5-trihydroxy-,esters,propyl ester;n-Propyl 3,4,5-trihydroxybenzoate;3,4, 5-Trihydroxybenzoic acid, propyl ester;Benzoic acid, 3,4, 5-trihydroxy-, propyl ester;Progallin P;Propyl 3,4,5-trihydroxybenzoate;n-Propyl ester of 3,4,5-trihydroxybenzoic acid;Nipa 49;Nipanox S 1;Gallic acid, propyl ester;3,4,5-Trihydroxybenzene-1-propylcarboxylate;n-Propyl gallate;Propyl Gallate BP/EP;Propyl Gallate (Tech.);propyl gallate;propyl 3,4,5-trihydroxybenzoate;
- PSA 86.99000
- LogP 1.37020
Synthetic route

| Conditions | Yield |
|---|---|
| With brominated modified sulfonic acid resin at 100℃; for 5h; | 98% |
| With toluene-4-sulfonic acid at 107℃; under 9308.91 Torr; for 0.133333h; Temperature; Wavelength; Sealed tube; Microwave irradiation; | 94% |
| With thionyl chloride at 35 - 65℃; for 1h; | 91.6% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In propan-1-ol; 2-methyl-propan-1-ol; water | 75% |
-
-
71-23-8
propan-1-ol

-
-
99-24-1
methyl galloate

-
A
-
121-79-9
Propyl gallate

-
B
-
149-91-7
3,4,5-trihydroxybenzoic acid

| Conditions | Yield |
|---|---|
| With tannase from Lactobacillus plantarum immobilised on glyoxyl-agarose at 25℃; pH=5; aq. acetate buffer; Enzymatic reaction; | A 55% B n/a |


| Conditions | Yield |
|---|---|
| With dipotassium hydrogenphosphate; tannase; phosphoric acid; water at 20 - 35℃; for 24h; Equilibrium constant; Thermodynamic data; ΔrHmo, ΔrGmo, ΔrSmo, other reagents: CH3COONa, CH3COOH; NaOH, 2-(N-morpholino)ethanesulfonic acid; various pH, other reaction time; |
-
-
71-23-8
propan-1-ol

-
-
18483-17-5, 23140-70-7, 135029-93-5
1,3,6-tri-O-galloyl-β-D-glucose

-
A
-
121-79-9
Propyl gallate

-
B
-
149-91-7
3,4,5-trihydroxybenzoic acid

| Conditions | Yield |
|---|---|
| With tannase from Lactobacillus plantarum immobilised on glyoxyl-agarose at 25℃; pH=5; aq. acetate buffer; Enzymatic reaction; |


| Conditions | Yield |
|---|---|
| With bio-imprinted tannase immobilized on ceolite In hexane; water at 40℃; for 24h; Enzymatic reaction; | |
| With tannase from Aspergillus oryzae In hexane; water at 40℃; Kinetics; Microbiological reaction; Enzymatic reaction; | |
| Stage #1: tannic acid With Aspergillus oryzae tannase at 20℃; for 0.0833333h; pH=6.0; Stage #2: propan-1-ol With D-Mannose; magnesium sulfate In hexane at 40℃; for 24h; Catalytic behavior; Reagent/catalyst; Enzymatic reaction; | |
| With immobilized tannase In hexane; water at 40℃; for 24h; Solvent; |

| Conditions | Yield |
|---|---|
| With Aspergillus fumigatus CAS-21 tannase In aq. buffer at 30℃; pH=6; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 48h; Williamson ether synthesis; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 75℃; for 5h; | 99% |
| With potassium carbonate In N,N-dimethyl-formamide for 4h; Reflux; | 73% |
-
-
121-79-9
Propyl gallate

-
-
200112-75-0
3-(perfluorooctyl)propyl iodide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone Inert atmosphere; Reflux; | 98% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In toluene for 5h; Reflux; | 98% |
-
-
121-79-9
Propyl gallate

-
-
1185733-70-3
2,2,2-trichloroethoxysulfuryl 2-methyl-3-methylimidazolium triflate

-
-
1313434-35-3
C16H15Cl9O14S3

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane at 20℃; for 0.266667h; | 96% |
-
-
121-79-9
Propyl gallate

-
-
112-89-0
1-Bromooctadecane

-
-
1167983-67-6
propyl 3,4,5-tris(octadecyloxy)benzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In tetrahydrofuran; acetonitrile at 85℃; for 6h; | 93% |
| With potassium carbonate |
-
-
121-79-9
Propyl gallate

-
-
23726-00-3
<2H9>trimethylsulphoxonium iodide

| Conditions | Yield |
|---|---|
| With potassium carbonate at 65℃; for 18h; Sealed tube; | 92.1% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 18h; Inert atmosphere; | 92% |
-
-
112-71-0
1-Bromotetradecane

-
-
121-79-9
Propyl gallate

-
-
402912-75-8
3,4,5-tristetradecyloxybenzoic acid propyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In N,N-dimethyl-formamide for 4h; Reflux; | 90% |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 3h; Inert atmosphere; | 80% |
| With potassium carbonate In acetone for 48h; Williamson ether synthesis; Reflux; | 69% |
| With potassium carbonate In dimethyl sulfoxide; acetone for 10h; Heating; | |
| With potassium carbonate In N,N-dimethyl-formamide at 90℃; |

| Conditions | Yield |
|---|---|
| With tris(pentafluorophenyl)borate In 5,5-dimethyl-1,3-cyclohexadiene Reflux; | 89.9% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In ethanol at 35℃; for 96h; Friedel-Crafts Alkylation; | 89.68% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In ethanol at 35℃; for 96h; Friedel-Crafts Alkylation; | 85.92% |
-
-
121-79-9
Propyl gallate

-
-
143-15-7
1-dodecylbromide

-
-
1228276-91-2
propyl 3,4,5-tris(n-dodecan-1-yloxy)benzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 20h; Williamson Ether Synthesis; | 84% |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 3h; Inert atmosphere; | 80% |
| With potassium carbonate | |
| With potassium carbonate In N,N-dimethyl-formamide at 90℃; |

| Conditions | Yield |
|---|---|
| Stage #1: Propyl gallate With lithium carbonate In N,N-dimethyl-formamide at 50℃; for 0.5h; Inert atmosphere; Stage #2: methyl iodide In N,N-dimethyl-formamide Inert atmosphere; | 83% |

-
-
121-79-9
Propyl gallate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane for 24h; Inert atmosphere; | 83% |
-
-
7766-49-6
1-iodo-10-undecene

-
-
121-79-9
Propyl gallate

-
-
894104-86-0
3,4,5-tris(10-undecenyloxy)benzoic acid n-propyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 48h; Heating; | 82% |

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In sulfuric acid at 80℃; for 12h; Solvent; Reagent/catalyst; | 82% |
-
-
150-46-9
triethyl borate

-
-
121-79-9
Propyl gallate

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
143259-85-2
Propyl 3-[(4-methyl-phenyl)-sulfonyloxy]-4,5-dihydroxy benzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran; dichloromethane | 81.7% |


| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 3h; Inert atmosphere; | 79% |
| With potassium carbonate In N,N-dimethyl-formamide at 90℃; |

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate In acetonitrile at 20℃; for 16h; Inert atmosphere; Cooling with ice; | 73.1% |

| Conditions | Yield |
|---|---|
| With lipozyme 435 at 55℃; for 120h; Temperature; Enzymatic reaction; | 69.1% |
| With recombinantlipase B from C. antarctica, expressed in Aspergillusniger, and immobilized on a macroporous hydrophobic resin In neat (no solvent) at 50℃; for 120h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Time; Green chemistry; Enzymatic reaction; | 67.1% |
-
-
10199-34-5, 14056-88-3, 15604-36-1
cis-dichlorobis(triphenylphosphine)platinum(II)

-
-
121-79-9
Propyl gallate

| Conditions | Yield |
|---|---|
| With triethylamine In methanol for 24h; Reflux; Inert atmosphere; | 67% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In butanone for 26h; Reflux; Inert atmosphere; | 65.8% |

| Conditions | Yield |
|---|---|
| Stage #1: Propyl gallate; diphenyltin(IV) dichloride In methanol at 20℃; for 4h; Stage #2: 1,10-Phenanthroline In methanol at 80℃; for 4h; | 58% |


| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In acetone for 72h; Inert atmosphere; Reflux; | 50% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In ethanol at 35℃; for 96h; Friedel-Crafts Alkylation; | 43.18% |
Propyl gallate Chemical Properties
Molecular Structure of Propyl gallate (CAS NO.121-79-9):
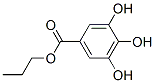
IUPAC Name: propyl 3,4,5-trihydroxybenzoate
Molecular Formula: C10H12O5
Formula Weight: 212.2
H bond acceptors: 5
H bond donors: 3
Freely Rotating Bonds: 7
Polar Surface Area: 53.99Å2
Index of Refraction: 1.595
Molar Refractivity: 52.93 cm3
Molar Volume: 155.6 cm3
Surface Tension: 62 dyne/cm
Density: 1.363 g/cm3
Flash Point: 181.3 °C
Enthalpy of Vaporization: 73.43 kJ/mol
Boiling Point: 448.6 °C at 760 mmHg
Vapour Pressure: 1.16E-08 mmHg at 25°C
Melting point: 146-149 °C(lit.)
Storage temp: 0-6°C
Solubility: ethanol: 50 mg/mL
Form: powder
Water Solubility: 0.35 g/100 mL (25 oC)
InChI
InChI=1/C10H12O5/c1-2-3-15-10(14)6-4-7(11)9(13)8(12)5-6/h4-5,11-13H,2-3H2,1H3
Smiles
c1(cc(c(O)c(c1)O)O)C(OCCC)=O
Propyl gallate Toxicity Data With Reference
| 1. | mmo-sat 200 µg/plate | SYSWAE Shiyan Shengwa Xuebao. Journal of Experimental Biology. (Guozi Shudian, China Publications Center, P.O. Box 399, Peking, Peoples Republic of China) V.1- 1953- 12 (1979),41. | ||
| 2. | cyt-ham:fbr 40 mg/L | ESKHA5 Eisei Shikenjo Hokoku. Bulletin of the National Hygiene Sciences. 96 (1978),55. | ||
| 3. | orl-rat LD50:2100 mg/kg | NTIS** National Technical Information Service. (Springfield, VA 22161) (Formerly U.S. Clearinghouse for Scientific and Technical Information) PB245-441 . | ||
| 4. | ipr-rat LD50:380 mg/kg | FOTEAO Food Technology. 2 (1948),308. | ||
| 5. | orl-mus LD50:1700 mg/kg | JAOCA7 Journal of the American Oil Chemists Society. 54 (1977),239. | ||
| 6. | orl-cat LD50:400 mg/kg | 14CYAT Industrial Hygiene and Toxicology. 2 (1963),1897. |
Propyl gallate Consensus Reports
NTP Carcinogenesis Bioassay (feed); No Evidence: mouse, rat NTPTR* National Toxicology Program Technical Report Series. (Research Triangle Park, NC 27709) No. NTP-TR-240 ,1982. . Reported in EPA TSCA Inventory.
Propyl gallate Safety Profile
Hazard Codes:  Xn
Xn
Risk Statements: 22-43-36/37/38 (Harmful if swallowed; May cause sensitization by skin contact; Irritating to the eyes; Irritating to the respiratory system; Irritating to the skin)
Safety Statements: 24-37-36-26 (Avoid contact with skin; Wear suitable gloves; Wear suitable protective clothing; In case of contact with eyes, rinse immediately with plenty of water and seek medical advice)
WGK Germany: 2
RTECS: LW8400000
HS Code: 29182950
Poison by ingestion and intraperitoneal routes. Experimental teratogenic and reproductive effects. Questionable carcinogen with experimental tumorigenic data. Mutation data reported. Combustible when exposed to heat or flame; can react with oxidizing materials. When heated to decomposition it emits acrid smoke and irritating fumes.
Propyl gallate Specification
Propyl gallate , with CAS number of 121-79-9, can be called 3,4,5-Trihydroxybenzene-1-propylcarboxylate ; Propyl Gallate BP/EP ; propyl gallate;propyl 3,4,5-trihydroxybenzoate ; n-Propyl ester of 3,4,5-trihydroxybenzoic acid ; Benzoic acid, 3,4, 5-trihydroxy-, propyl ester ; 3,4, 5-Trihydroxybenzoic acid, propyl ester . It is a white to light beige crystalline powder. Propyl gallate (CAS NO.121-79-9) is used as a triplet quencher in fluorescence microscopy. It is used as food and feed additives, and in foods, cosmetics, hair products, adhesives, and lubricants. n-PROPYL GALLATE is used to protect oils and fats in products from oxidation.
Related Products
- Propyl (<sup>2</sup>H)formate
- Propyl 1,4-benzodioxan-2-carboxylate
- Propyl 2-methyl-3-furyl disulfide
- Propyl 2-methylbutyrate
- Propyl 4-pyridyl ketone
- Propyl 6-aminopyridazine-3-carboxylate
- Propyl acetate
- Propyl beta-D-glucopyranoside
- Propyl bromoacetate
- Propyl butyrate
- 121801-61-4
- 121806-76-6
- 12180-80-2
- 121808-62-6
- 121809-54-9
- 121809-74-3
- 121-81-3
- 121816-84-0
- 121817-29-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View