-
Name
Prostaglandin E1
- EINECS 212-017-2
- CAS No. 745-65-3
- Article Data30
- CAS DataBase
- Density 1.131 g/cm3
- Solubility insoluble in water
- Melting Point 115-116 °C
- Formula C20H34O5
- Boiling Point 529.3 °C at 760 mmHg
- Molecular Weight 354.487
- Flash Point 288 °C
- Transport Information UN 2811 6.1/PG 3
- Appearance Crystalline solid
- Safety 36-26
- Risk Codes 22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms Cyclopentaneheptanoicacid, 3-hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-, (-)- (8CI);Cyclopentaneheptanoicacid, 3a-hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-(7CI);(-)-Prostaglandin E1;11a,15(S)-Dihydroxy-9-oxo-13-trans-prostenoic acid;11a,15a-Dihydroxy-9-oxo-13-trans-prostenoic acid;Alprox TD;Caveject;Caverject;Eglandin;Liple;Lipoprost;Liprostin;Minprog;NSC 165559;ONO 1608;Palux;Prostandin 500;Prostin VR Pediatric;Prostivas;SEPA-PGE1;SEPA-alprostadil;Topiglan;Vasaprostan;l-PGE1;l-Prostaglandin E1;Prostaglandin E1;
- PSA 94.83000
- LogP 3.47510
Synthetic route
-
-
1251949-12-8
C32H64O5Si2

-
-
745-65-3
ALPROSTADIL

| Conditions | Yield |
|---|---|
| With hydrogen fluoride; water In acetonitrile at 20℃; for 6h; stereoselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| With porcine pancreatic lipase; sodium chloride; calcium chloride In tetrahydrofuran; water for 0.5h; Hydrolysis; Enzymatic reaction; pH 7.1; | 89% |
| With pig liver esterase In water for 4h; Ambient temperature; | 86% |
| With porcine liver esterase; phosphate buffer solution In acetone for 4h; Ambient temperature; | 86% |
| With phosphate buffer Baker's yeast, pH 7.0; | 72% |
| (microbiological transformation); |
-
-
87007-31-6
7-{(1R,2R,3R)-3-(tert-Butyl-dimethyl-silanyloxy)-2-[(E)-(S)-3-(tert-butyl-dimethyl-silanyloxy)-oct-1-enyl]-5-oxo-cyclopentyl}-heptanoic acid

-
-
745-65-3
ALPROSTADIL

| Conditions | Yield |
|---|---|
| With hydrogen fluoride In acetonitrile Yield given; |
-
-
1012104-26-5
(8R,11R,12R,15S)-11,15-Bis-(2-trimethylsilylethoxymethoxy)-9-oxo-prost-13-en-1-oic acid 2-trimethylsilylethoxymethyl ester

-
-
745-65-3
ALPROSTADIL

| Conditions | Yield |
|---|---|
| With nitromethane; n-butanethiol; magnesium bromide In diethyl ether at 20℃; for 1h; |
-
-
1012104-24-3
(S)-E-3-(2-trimethylsilylethoxymethoxy)-1-tributylstannyl-1-octene

-
-
745-65-3
ALPROSTADIL

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: n-BuLi / tetrahydrofuran; hexane / 0.5 h / -78 °C 1.2: 2-thienyl(cyano)copper lithium / tetrahydrofuran; hexane / 0.5 h / -78 °C 1.3: 67 percent / tetrahydrofuran; hexane / 0.5 h / -78 °C 2.1: magnesium bromide; nitromethane; n-butanethiol / diethyl ether / 1 h / 20 °C View Scheme |
-
-
1012104-25-4
2-trimethylsilylethoxymethyl-(R)-3-(2-trimethylsilylethoxymethoxy)-5-oxo-1-cyclopentene-1-heptenoate

-
-
745-65-3
ALPROSTADIL

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: n-BuLi / tetrahydrofuran; hexane / 0.5 h / -78 °C 1.2: 2-thienyl(cyano)copper lithium / tetrahydrofuran; hexane / 0.5 h / -78 °C 1.3: 67 percent / tetrahydrofuran; hexane / 0.5 h / -78 °C 2.1: magnesium bromide; nitromethane; n-butanethiol / diethyl ether / 1 h / 20 °C View Scheme |
-
-
54996-33-7
7-(3R-hydroxy-5-oxo-cyclopent-1-enyl)heptanoic acid

-
-
745-65-3
ALPROSTADIL

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: 31 percent / 4-(dimethylamino)pyridine; lithium carbonate / dimethylformamide / 20 h / 20 °C 2.1: n-BuLi / tetrahydrofuran; hexane / 0.5 h / -78 °C 2.2: 2-thienyl(cyano)copper lithium / tetrahydrofuran; hexane / 0.5 h / -78 °C 2.3: 67 percent / tetrahydrofuran; hexane / 0.5 h / -78 °C 3.1: magnesium bromide; nitromethane; n-butanethiol / diethyl ether / 1 h / 20 °C View Scheme |
-
-
39178-64-8
(E)-1-iodo-1-octen-3-one

-
-
745-65-3
ALPROSTADIL

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: 95 percent / n-butylboronic complex of diphenyl D-prolinol; catecholborane / toluene / 36 h / -78 °C 2.1: 97 percent / Et3N; imidazole / dimethylformamide / 20 °C 3.1: n-BuLi; CuCN; MeLi / diethyl ether / 3.08 h / -78 - 0 °C 3.2: diethyl ether; hexane / 0.33 h / -78 °C 4.1: pyridinium p-toluenesulfonate / acetone; H2O / 4 h / 20 °C 5.1: 89 percent / porcine pancreatic lipase; NaCl; CaCl2 / H2O; tetrahydrofuran / 0.5 h / Enzymatic reaction; pH 7.1 View Scheme |
Prostaglandin E1 Specification
1. Introduction of Prostaglandin E1
Prostaglandin E1 is one kind of white powder. The IUPAC Name of this chemical is 7-[(1R,3R)-3-hydroxy-2-[(E,3S)-3-hydroxyoct-1-enyl]-5-oxocyclopentyl]heptanoic acid. Besides, Prostaglandin E1 belogns to Miscellaneous Biochemicals; Prostaglandins; Prostaglandin; Chiral Reagents; Intermediates & Fine Chemicals; Pharmaceuticals; Prostanoid receptor and related. In addition, Prostaglandin E1 is reported soluble in ethanol at >100 mg/ml. It is insoluble in water.
2. Properties of Prostaglandin E1
Physical properties about Prostaglandin E1 are:
(1)Melting Point: 115-116 °C; (2)Boiling point: 529.3 °C at 760 mmHg; (3)Storage Temperature: -20°C; (4)Flash Point: 288 °C; (5)Density: 1.131 g/cm3; (6)Surface Tension: 52.3 dyne/cm; (7)Enthalpy of Vaporization: 92.52 kJ/mol; (8)Vapour Pressure: 2.06E-13 mmHg at 25°C; (9)XLogP3-AA: 3.2; (10)H-Bond Donor: 3; (11)H-Bond Acceptor: 5; (12)Rotatable Bond Count: 13; (13)Tautomer Count: 3; (14)Exact Mass: 354.240624; (15)MonoIsotopic Mass: 354.240624; (16)Topological Polar Surface Area: 94.8; (17)Heavy Atom Count: 25; (18)Formal Charge: 0; (19)Complexity: 432; (20)Isotope Atom Count: 0; (21)Defined Atom Stereocenter Count: 0; (22)Undefined Atom Stereocenter Count: 4; (23)Defined Bond Stereocenter Count: 0; (24)Undefined Bond Stereocenter Count: 1; (25)Covalently-Bonded Unit Count: 1; (26)Feature 3D Acceptor Count: 5; (27)Feature 3D Donor Count: 2; (28)Feature 3D Anion Count: 1; (29)Feature 3D Hydrophobe Count: 4; (30)Feature 3D Ring Count: 1; (31)Effective Rotor Count: 13.8; (32)Conformer Sampling RMSD: 1.6; (33)CID Conformer Count: 88.
3. Structure Descriptors of Prostaglandin E1
(1)Canonical SMILES: CCCCCC(C=CC1C(CC(=O)C1CCCCCCC(=O)O)O)O
(2)Isomeric SMILES: CCCCC[C@@H](/C=C/C1[C@@H](CC(=O)[C@@H]1CCCCCCC(=O)O)O)O
(3)InChI: InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/b13-12+/t15-,16+,17?,19+/m0/s1
(4)InChIKey: GMVPRGQOIOIIMI-ZWLIRFKBSA-N
4. Toxicity information of Prostaglandin E1
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 19800ug/kg (19.8mg/kg) | Drugs in Japan Vol. 6, Pg. 47, 1982. | |
| mouse | LD50 | intravenous | 21mg/kg (21mg/kg) | Drugs in Japan Vol. 6, Pg. 47, 1982. | |
| mouse | LD50 | oral | 186mg/kg (186mg/kg) | Drugs in Japan Vol. 6, Pg. 47, 1982. | |
| mouse | LD50 | subcutaneous | 26400ug/kg (26.4mg/kg) | Drugs in Japan Vol. 6, Pg. 47, 1982. | |
| rat | LD50 | intraarterial | 21mg/kg (21mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) LUNGS, THORAX, OR RESPIRATION: DYSPNEA GASTROINTESTINAL: "HYPERMOTILITY, DIARRHEA" | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 25, Pg. 499, 1983. |
| rat | LD50 | intraperitoneal | 24900ug/kg (24.9mg/kg) | Drugs in Japan Vol. 6, Pg. 47, 1982. | |
| rat | LD50 | intravenous | 19200ug/kg (19.2mg/kg) | Drugs in Japan Vol. 6, Pg. 47, 1982. | |
| rat | LD50 | oral | 228mg/kg (228mg/kg) | Drugs in Japan Vol. 6, Pg. 47, 1982. | |
| rat | LD50 | subcutaneous | 18600ug/kg (18.6mg/kg) | Drugs in Japan Vol. 6, Pg. 47, 1982. |
5. Safety Information of Prostaglandin E1
Hazard Codes: Xn
 ,Xi
,Xi
Risk Statements: 22-36/37/38
22: Harmful if swallowed
36: Irritating to the eyes
37: Irritating to the respiratory system
38: Irritating to the skin
Safety Statements: 36-26
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
6. Uses of Prostaglandin E1
Prostaglandin E1 (CAS NO.745-65-3) is used as a drug in the treatment of erectile dysfunction and has vasodilatory properties. Aprostadil is also used in maintaining a patent ductus arteriosus in the newborn. This is primarily useful when there is threat of premature closure of the ductus arteriosus in an infant with ductal-dependent congenital heart disease, including cyanotic lesions (e.g., pulmonary atresia/stenosis, tricuspid atresia/stenosis, transposition of the great arteries) and acyanotic lesions (e.g., coarctation of the aorta, hypoplastic left heart syndrome, critical aortic stenosis, interrupted aortic arch).
7. Production of Prostaglandin E1
Prostaglandin E1 can be got from Methyl g-linolenate which is showed as follows:
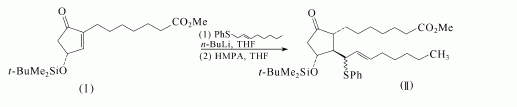
Related Products
- PROSTAGLANDIN A1
- Prostaglandin A2
- Prostaglandin D2
- Prostaglandin E1
- Prostaglandin E2
- Prostaglandin E2 sodium salt
- PROSTAGLANDIN F1ALPHA
- Prostaglandin F2 methyl ester
- Prostaglandin F2a
- Prostaglandin F2a tris salt
- 74568-07-3
- 74572-01-3
- 74572-03-5
- 74572-04-6
- 74572-05-7
- 74572-13-7
- 74572-14-8
- 74572-29-5
- 74575-17-0
- 74577-19-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View