-
Name
Tobramycin
- EINECS 251-322-5
- CAS No. 32986-56-4
- Article Data10
- CAS DataBase
- Density 1.52 g/cm3
- Solubility 50 mg/mL in water
- Melting Point 178 °C
- Formula C18H37N5O9
- Boiling Point 775.436 °C at 760 mmHg
- Molecular Weight 467.52
- Flash Point 422.8 °C
- Transport Information
- Appearance White powder
- Safety 26-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms D-Streptamine, O-3-amino-3-deoxy-alpha-D-glucopyranosyl-(1-6)-O-(2,6-diamino-2,3,6-trideoxy-alpha-D-ribo-hexopyranosyl-(1-4))-2-deoxy- ; Deoxykanamycin B ; Gotabiotic ; Nebramycin ; O-3-Amino-3-deoxy-alpha-D-glucopyranosyl-(1-4)-O-(2,6-diamino-2,3,6-trideoxy-alpha-D-ribo-hexopyranosyl-(1-6))-2-deoxy-L-streptamine;O-3-Amino-3-deoxy-alpha-D-lucopyranosyl-(1-6)-O-(2,6-diamino-2,3,6-trideoxy-alpha-D-ibohexopyranosyl-(1-4))-2-deoxy-D-streptamine;Tobamycin Base;Sulfate sterile;
- PSA 268.17000
- LogP -2.79430
Synthetic route
-
-
23155-02-4
fosfomycin

-
-
32986-56-4
tobramycin

-
-
32986-56-4
tobramycin

| Conditions | Yield |
|---|---|
| Stage #1: tobramycin sulfate Stage #2: With trifluoroacetic acid In water |
-
-
32986-56-4
tobramycin

-
-
207857-15-6
1,3-di(tert-butyloxycarbonyl)-2-(trifluoromethylsulfonyl)guanidine

| Conditions | Yield |
|---|---|
| With triethylamine In 1,4-dioxane; water at 20℃; for 72h; | 100% |
-
-
32986-56-4
tobramycin

-
-
207857-15-6
1,3-di(tert-butyloxycarbonyl)-2-(trifluoromethylsulfonyl)guanidine

-
-
290360-48-4
C73H127N15O29

| Conditions | Yield |
|---|---|
| With triethylamine In 1,4-dioxane; water at 20℃; for 72h; | 100% |
-
-
32986-56-4
tobramycin

-
-
468065-22-7
O-3-azido-3-deoxy-α-D-glucopyranosyl-(1→6)-O-[2,6-diazido-2,3,6-trideoxy-α-D-ribo-hexopyranosyl-(1→4)]-1,3-diazido-1,2,3-trideoxy-D-myo-inositol

| Conditions | Yield |
|---|---|
| With TEA; triflic azide; copper(II) sulfate In methanol; dichloromethane | 100% |
| With triflic azide; triethylamine; zinc(II) chloride In methanol; dichloromethane; water at 20℃; | 92% |
| With triflic azide; copper(II) sulfate; triethylamine | 83% |
| With triflic azide; triethylamine; zinc(II) chloride In methanol; dichloromethane; water Inert atmosphere; | |
| With triflic azide; zinc(II) chloride In methanol; dichloromethane; water Inert atmosphere; |
-
-
32986-56-4
tobramycin

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
172950-21-9
di-tert-butyl ((1S,3R,4S,5S,6R)-4-(((2S,3R,4S,5S,6R)-4-((tert-butoxycarbonyl)amino)-3,5-dihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-6-(((2R,3R,5S,6R)-3-((tert-butoxycarbonyl)amino)-6-(((tert-butoxycarbonyl)methyl)-5-hydroxytetrahydro-2H-pyran-2-yl)oxy)-5-hydroxycyclohexane-1,3-diyl)dicarbamate)

| Conditions | Yield |
|---|---|
| With triethylamine In water at 60℃; for 0.666667h; | 98% |
| With triethylamine In methanol; water at 55℃; | 98% |
| With triethylamine In methanol; water at 55℃; | 97% |
-
-
32986-56-4
tobramycin

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 20℃; for 24h; Solvent; | 92% |
-
-
32986-56-4
tobramycin

-
-
501-53-1
benzyl chloroformate

-
-
61093-69-4
1,3,2',6',3''-pentakis(N-benzyloxycarbonyl)-3'-deoxykanamycin B

| Conditions | Yield |
|---|---|
| With sodium carbonate | 90% |
| With sodium carbonate In water | |
| With sodium carbonate In acetone at 0℃; |
-
-
32986-56-4
tobramycin

-
-
65162-83-6
N-(benzyloxycarbonyloxy)phthalimide

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol; water at 20℃; for 18h; | 64% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol; water for 20h; | 63% |

| Conditions | Yield |
|---|---|
| With Tmed buffer; ATP In water for 12h; aminoglycoside phosphotransferase; | 62% |
-
-
32986-56-4
tobramycin

-
-
62210-73-5
N-<<(benzyloxy)carbonyl>oxy>-5-norbornene-2,3-dicarboximide

-
-
24424-99-5
di-tert-butyl dicarbonate

| Conditions | Yield |
|---|---|
| Stage #1: tobramycin With zinc(II) acetate dihydrate In dimethyl sulfoxide at 21℃; Stage #2: N-<<(benzyloxy)carbonyl>oxy>-5-norbornene-2,3-dicarboximide In dimethyl sulfoxide at 21℃; for 24h; Stage #3: di-tert-butyl dicarbonate In dimethyl sulfoxide at 21℃; for 4h; | 57% |


| Conditions | Yield |
|---|---|
| Stage #1: tobramycin With zinc(II) acetate dihydrate In dimethyl sulfoxide at 21℃; Stage #2: di-tert-butyl dicarbonate In dimethyl sulfoxide at 21℃; for 5h; | 53% |
-
-
32986-56-4
tobramycin

-
-
40371-52-6
N-[(S)-4-benzyloxycarbonylamino-2-hydroxy-butyryloxy]succinimide

-
-
1312471-07-0
6'-N-CbzAHB tobramycin

| Conditions | Yield |
|---|---|
| Stage #1: tobramycin With zinc diacetate In water; N,N-dimethyl-formamide at 20℃; for 16h; Stage #2: N-[(S)-4-benzyloxycarbonylamino-2-hydroxy-butyryloxy]succinimide In water; N,N-dimethyl-formamide for 24h; | 31% |
-
-
32986-56-4
tobramycin

-
-
93801-70-8
2,4-dinitrophenylthioglycolic acid N-hydroxy succinimide ester

-
-
93801-67-3
N-{(2R,3S,5R,6R)-5-Amino-6-[(1R,2S,3S,4R,6S)-4,6-diamino-3-((2S,3R,4S,5S,6R)-4-amino-3,5-dihydroxy-6-hydroxymethyl-tetrahydro-pyran-2-yloxy)-2-hydroxy-cyclohexyloxy]-3-hydroxy-tetrahydro-pyran-2-ylmethyl}-2-(2,4-dinitro-phenylsulfanyl)-acetamide

| Conditions | Yield |
|---|---|
| In water; N,N-dimethyl-formamide at 0℃; for 2h; pH=10.76; | 28.3% |

| Conditions | Yield |
|---|---|
| In methanol for 0.166667h; Ambient temperature; Yields of byproduct given; | A 26.9% B n/a C n/a D n/a |
| In methanol for 0.166667h; Ambient temperature; Yields of byproduct given; | A n/a B 7.4% C n/a D n/a |
| In methanol for 0.166667h; Ambient temperature; Yield given. Yields of byproduct given; |

-
-
32986-56-4
tobramycin

-
-
13139-17-8
N-(Benzyloxycarbonyloxy)succinimide

| Conditions | Yield |
|---|---|
| Stage #1: tobramycin With zinc diacetate In dimethyl sulfoxide for 12h; Stage #2: N-(Benzyloxycarbonyloxy)succinimide In dimethyl sulfoxide for 2h; | 25% |
-
-
32986-56-4
tobramycin


| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide Darkness; | 17% |
-
-
32986-56-4
tobramycin

-
-
1195-16-0, 768351-04-8
N-acetyl-DL-homocysteinethiolactone

| Conditions | Yield |
|---|---|
| In water for 30h; pH=10.12-10.25; Yield given; |
-
-
32986-56-4
tobramycin

-
-
1195-16-0, 768351-04-8
N-acetyl-DL-homocysteinethiolactone

-
-
70-11-1
α-bromoacetophenone

| Conditions | Yield |
|---|---|
| With hydrogenchloride 1.) 20110.12-10.25, 30 min; 2.) pH=7.5, water, THF, 0 deg C, 2 h; Yield given. Multistep reaction; |


| Conditions | Yield |
|---|---|
| With pyridine |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; triethylamine 1.) dioxane, RT, 30 min, 2.) dioxane, water, RT, 20 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| at 37℃; aminoglycoside-4'-nucleotidyltransferaze of Bacillus brevis, MgCl2, potassium phosphate buffer (pH 6.0); |
-
-
32986-56-4
tobramycin

| Conditions | Yield |
|---|---|
| With triflic azide; triethylamine; zinc(II) chloride In methanol; dichloromethane; water |
-
-
32986-56-4
tobramycin

-
-
103842-08-6
3,2',6'-tris(N-benzyloxycarbonyl)-3'-deoxy-3''-N-(trifluoroacetyl)kanamycin B

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: Zn(OAc)2*2H2O / dimethylsulfoxide / 12 h 1.2: 25 percent / dimethylsulfoxide / 2 h 2.1: 60 percent / dimethylsulfoxide / 2 h View Scheme |
-
-
32986-56-4
tobramycin

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: Zn(OAc)2*2H2O / dimethylsulfoxide / 12 h 1.2: 25 percent / dimethylsulfoxide / 2 h 2.1: 60 percent / dimethylsulfoxide / 2 h 3.1: 11 mg / tetrahydrofuran; H2O / 96 h / 20 °C 4.1: aq. NaOH / methanol / 20 h 5.1: 4.5 mg / H2 / 10 percent Pd/C / dioxane; H2O; acetic acid / 0.67 h View Scheme |
-
-
32986-56-4
tobramycin

-
-
533923-31-8
C53H67N7O19

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: Zn(OAc)2*2H2O / dimethylsulfoxide / 12 h 1.2: 25 percent / dimethylsulfoxide / 2 h 2.1: 60 percent / dimethylsulfoxide / 2 h 3.1: 11 mg / tetrahydrofuran; H2O / 96 h / 20 °C 4.1: aq. NaOH / methanol / 20 h View Scheme |
-
-
32986-56-4
tobramycin

-
-
533923-30-7
C62H70F3N7O21

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: Zn(OAc)2*2H2O / dimethylsulfoxide / 12 h 1.2: 25 percent / dimethylsulfoxide / 2 h 2.1: 60 percent / dimethylsulfoxide / 2 h 3.1: 11 mg / tetrahydrofuran; H2O / 96 h / 20 °C View Scheme |
Tobramycin Chemical Properties
Molecular structure of Tobramycin (CAS NO.32986-56-4) is:
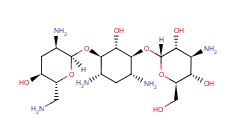
Product Name: Tobramycin
CAS Registry Number: 32986-56-4
IUPAC Name: 4-amino-2-[4,6-diamino-3-[3-amino-6-(aminomethyl)-5-hydroxyoxan-2-yl]oxy-2-hydroxycyclohexyl]oxy-6-(hydroxymethyl)oxane-3,5-diol
Molecular Weight: 467.51448 [g/mol]
Molecular Formula: C18H37N5O9
XLogP3-AA: -6.2
H-Bond Donor: 10
H-Bond Acceptor: 14
EINECS: 251-322-5
Refractive index: 143 ° (C=4, H2O)
Storage temp.: 2-8°C
Solubility: H2O: 50 mg/mL, clear, faintly yellow
Melting Point: 178 °C
Surface Tension: 92.6 dyne/cm
Density: 1.52 g/cm3
Flash Point: 422.8 °C
Enthalpy of Vaporization: 128.66 kJ/mol
Boiling Point: 775.4 °C at 760 mmHg
Vapour Pressure: 9.05E-28 mmHg at 25°C
Product Categories: Oligosaccharide Compounds; Aminoglycosides (Antibiotics for Research and Experimental Use); Antibiotics for Research and Experimental Use; Biochemistry; Intermediates & Fine Chemicals; Oligosaccharides; Pharmaceuticals; Peptide Synthesis/Antibiotics
Tobramycin Uses
Tobramycin (CAS NO.32986-56-4) is used to treat various types of bacterial infections, particularly Gram-negative infections. It is also combined with dexamethasone as an ophthalmic solution.
Tobramycin Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| man | TDLo | intravenous | 37714ug/kg (37.714mg/kg) | KIDNEY, URETER, AND BLADDER: URINE VOLUME INCREASED | American Journal of Kidney Diseases. Vol. 23, Pg. 118, 1994. |
| mouse | LD50 | intramuscular | 440mg/kg (440mg/kg) | Yakkyoku. Pharmacy. Vol. 27, Pg. 1467, 1976. | |
| mouse | LD50 | intraperitoneal | 445mg/kg (445mg/kg) | Journal of International Medical Research. Vol. 2, Pg. 100, 1974. | |
| mouse | LD50 | intravenous | 72500ug/kg (72.5mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD BEHAVIORAL: ATAXIA LUNGS, THORAX, OR RESPIRATION: DYSPNEA | Antibiotiki. Vol. 29, Pg. 361, 1984. |
| mouse | LD50 | oral | > 11500mg/kg (11500mg/kg) | Yakkyoku. Pharmacy. Vol. 27, Pg. 1467, 1976. | |
| mouse | LD50 | subcutaneous | 367mg/kg (367mg/kg) | SENSE ORGANS AND SPECIAL SENSES: PTOSIS: EYE BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION | Toxicology and Applied Pharmacology. Vol. 25, Pg. 398, 1973. |
| pig | LD50 | subcutaneous | 676mg/kg (676mg/kg) | SENSE ORGANS AND SPECIAL SENSES: PTOSIS: EYE LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) | Toxicology and Applied Pharmacology. Vol. 25, Pg. 398, 1973. |
| rat | LD50 | intramuscular | 913mg/kg (913mg/kg) | Iyakuhin Kenkyu. Study of Medical Supplies. Vol. 8, Pg. 107, 1977. | |
| rat | LD50 | intraperitoneal | 1030mg/kg (1030mg/kg) | Journal of International Medical Research. Vol. 2, Pg. 100, 1974. | |
| rat | LD50 | intravenous | 104mg/kg (104mg/kg) | Journal of International Medical Research. Vol. 2, Pg. 100, 1974. | |
| rat | LD50 | oral | > 7500mg/kg (7500mg/kg) | Yakkyoku. Pharmacy. Vol. 27, Pg. 1467, 1976. | |
| rat | LD50 | subcutaneous | 969mg/kg (969mg/kg) | SENSE ORGANS AND SPECIAL SENSES: OTHER CHANGES: OLFACTION | Toxicology and Applied Pharmacology. Vol. 22, Pg. 332, 1972. |
Tobramycin Consensus Reports
EPA Genetic Toxicology Program.
Tobramycin Safety Profile
Safty information about Tobramycin (CAS NO.32986-56-4) is:
Poison by intravenous and subcutaneous routes. Moderately toxic by intramuscular and intraperitoneal routes. An experimental teratogen. When heated to decomposition it emits toxic fumes of NOx.
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S37/39:Wear suitable gloves and eye/face protection.
WGK Germany: 2
RTECS: WK2100000
F: 3-10
Tobramycin Specification
Tobramycin , its cas register number is 32986-56-4. It also can be called Tobramycin [USAN:INN:BAN:JAN] ; Tobradex ; Tobrasone ; 1-Epitobramycin ; 3'-Deoxykanamycin B ; 4-(2,6-Diamino-2,3,6-trideoxy-alpha-D-glycopyranosyl)-6-(3-amino-3-deoxy-alpha-D-glycopyranosyl)-2-deoxystreptamine ; Aktob ; D-Streptamine, O-3-amino-3-deoxy-alpha-D-glucopyranosyl-(1-6)-O-(2,6-diamino-2,3,6-trideoxy-alpha-D-ribo-hexopyranosyl-(1-4))-2-deoxy- ; Deoxykanamycin B ; Gotabiotic ; Nebramycin ; O-3-Amino-3-deoxy-alpha-D-glucopyranosyl-(1-4)-O-(2,6-diamino-2,3,6-trideoxy-alpha-D-ribo-hexopyranosyl-(1-6))-2-deoxy-L-streptamine ; O-3-Amino-3-deoxy-alpha-D-lucopyranosyl-(1-6)-O-(2,6-diamino-2,3,6-trideoxy-alpha-D-ibohexopyranosyl-(1-4))-2-deoxy-D-streptamine ; Streptamine, O-3-amino-3-deoxy-alpha-D-glucopyranosyl-(1-4)-O-(2,6-diamino-2,3,6-tyrideoxy-alpha-D-ribohexopyranosyl-(1-6))-2-deoxy-, D- ; Tenebrimycin ; Tenemycin ; Tobacin ; Tobi ; Tobracin ; Tobradistin ; Tobralex ; Tobramaxin ; Tobramicin ; Tobramicina ; Tobramitsetin ; Tobramycetin ; Tobramycine ; Tobramycinum ; Tobrased ; Tobrex ; D-Streptamine, O-3-amino-3-deoxy-alpha-D-glucopyranosyl-(1-6)-O-(2,6-diamino-2,3,6-trideoxy-alpha-D-ribo-hexopyranosyl-(1-4))-2-deoxy- ; Streptamine, O-3-amino-3-deoxy-alpha-D-glucopyranosyl-(1-4)-O-(2,6-diamino-2,3,6-trideoxy-alpha-D-ribohexopyranosyl-(1-6))-2-deoxy-, D- . It is a white solid. Like other aminoglycosides, In genetically susceptible individuals, it can cause deafness or a loss of equilibrioception (vertigo) .It can also be highly toxic to the kidneys, particularly if multiple doses accumulate over a course of treatment. For these reasons, when it is given parenterally, it is usually dosed by body weight. Various formulae exist for calculating tobramycin dosage.
Related Products
- Tobramycin
- Tobramycin sulfate
- Tobramycin sulfate
- 32988-39-9
- 329-89-5
- 32990-47-9
- 329909-27-5
- 32991-58-5
- 329922-44-3
- 32993-05-8
- 329944-72-1
- 32996-16-0
- 32996-24-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View