-
Name
2,4,7-TRINITRO-9-FLUORENONE
- EINECS 204-965-0
- CAS No. 129-79-3
- Article Data19
- CAS DataBase
- Density 1.747 g/cm3
- Solubility
- Melting Point 176-178°C
- Formula C13H5 N3 O7
- Boiling Point 561.7 °C at 760 mmHg
- Molecular Weight 315.199
- Flash Point 292.3 °C
- Transport Information 387
- Appearance
-
Safety
Suspected carcinogen with experimental tumorigenic data. Mildly toxic by ingestion. Human mutation data reported. A skin and eye irritant. When heated to decomposition it emits highly toxic fumes of NOx. See also NITRO COMPOUNDS of AROMATIC HYDROCARBONS and KETONES.
Analytical Methods:
For occupational chemical analysis use NIOSH: see 2,4,7-TRINITROFLUOREN-9-ONE.
- Risk Codes 23/24/25-33
- Molecular Structure
- Hazard Symbols Suspected carcinogen. Mildly toxic by ingestion. A skin and eye irritant.
- Synonyms Fluoren-9-one,2,4,7-trinitro- (6CI,7CI,8CI); 2,4,7-Trinitro-9H-fluoren-9-one;2,4,7-Trinitrofluoren-9-one; 2,4,7-Trinitrofluorenone; 2,4,7-Trinitrofluorone;9-Oxo-2,4,7-trinitrofluorene; NSC 12367; TNF; TNF (ketone)
- PSA 154.53000
- LogP 4.19220
Synthetic route
Conditions
Conditions Yield With sulfuric acid; nitric acid In water for 2h; Reflux; regioselective reaction; 94% With sulfuric acid; nitric acid at 70℃; 93% With sulfuric acid; nitric acid In water for 2h; Reflux; 82% -
-
34263-36-0
2,4,7-trinitrofluorene

-
-
129-79-3
2,4,7-trinitrofluorene-9-one
Conditions
Conditions Yield With sodium dichromate; acetic acid In water at 95℃; for 2h; 79% -
-
47430-58-0
2,4,7-tri-nitro-9,10-phenanthrenequinone

-
-
129-79-3
2,4,7-trinitrofluorene-9-one
Conditions
Conditions Yield With copper(l) chloride In N,N-dimethyl-formamide at 100℃; for 5h; Reagent/catalyst; 76% -
-
1172-02-7
9-dicyanomethylene-2,4,7-trinitrofluorene

-
-
74959-64-1
2-(4-methoxybenzylidene)-N-phenylhydrazinecarbothioamide

-
A
-
1093399-53-1
4-amino-1-(4-methoxybenzoyl)-2,7-dinitro-4'-phenylspiro(fluoren-9,3'-[1,2,4]triazolidine)-5'-thione

-
B
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
C
-
84409-01-8
4-amino-2,7-dinitro-9-fluorenone

-
D
-
74959-57-2
phenyl-(5-(4-methoxyphenyl)-3H-[1,3,4]thiadiazol-2-ylidene)amine
Conditions
Conditions Yield With pyridine at 100℃; for 5h; Further byproducts.; A 56%
B n/a
C n/a
D 7%...Expand
-
-
1172-02-7
9-dicyanomethylene-2,4,7-trinitrofluorene

-
-
13431-41-9
4-benzylthiosemicarbazide

-
C
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
D
-
84409-01-8
4-amino-2,7-dinitro-9-fluorenone
Conditions
Conditions Yield In pyridine for 3h; Heating; Further byproducts given; A 23%
B 23%
C n/a
D n/a...Expand
-
-
72391-95-8
4-nitro-9-fluorenone

-
-
129-79-3
2,4,7-trinitrofluorene-9-one
Conditions
Conditions Yield With nitric acid at 100℃; 
-
-
129-79-3
2,4,7-trinitrofluorene-9-one
Conditions
Conditions Yield With nitric acid 
-
-
129-79-3
2,4,7-trinitrofluorene-9-one
Conditions
Conditions Yield With nitric acid -
-
486-25-9
9-fluorenone

-
-
7664-93-9
sulfuric acid

-
-
7697-37-2
nitric acid

-
-
129-79-3
2,4,7-trinitrofluorene-9-one
...Expand
-
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
90053-09-1
4-(9H-9-carbazolyl)butanoic acid

-
-
102737-76-8
2,7-dihydroxy-3,6,10,11-tetrakis(pentyloxy)triphenylene
Conditions
Conditions Yield Stage #1: 4-(9H-9-carbazolyl)butanoic acid With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0℃; for 0.5h;
Stage #2: 2,4,7-trinitrofluorene-9-one; 2,7-dihydroxy-3,6,10,11-tetrakis(pentyloxy)triphenylene In dichloromethane at 20℃; for 24h;100% ...Expand
-
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
109-77-3
malononitrile

-
-
1172-02-7
9-dicyanomethylene-2,4,7-trinitrofluorene
Conditions
Conditions Yield In methanol for 2.5h; Knoevenagel Condensation; Reflux; 98% With piperidine In methanol Conditions
Conditions Yield With sulfuric acid; bromine; nitric acid at 80 - 85℃; 96% With sulfuric acid; bromine; nitric acid at 80 - 85℃; for 2h; 96% With sulfuric acid; bromine; nitric acid at 55 - 60℃; for 2h; 58% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
121213-96-5
2,6-diisopropyl-3-nitrobenzeneamine
Conditions
Conditions Yield With zinc(II) chloride at 150 - 170℃; 92% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
81090-34-8
4-bromo-2-isopropyl-aniline
Conditions
Conditions Yield With zinc(II) chloride at 150 - 170℃; 92% Conditions
Conditions Yield With zinc(II) chloride at 150 - 170℃; 92% Conditions
Conditions Yield With zinc(II) chloride at 135 - 140℃; for 1.5h; 90% Conditions
Conditions Yield With zinc(II) chloride at 195 - 200℃; for 6h; 88% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
109-79-5
1-butanethiol

-
-
90685-68-0
9,9-bis(n-butylthio)-2,4,7-trinitrofluorene
Conditions
Conditions Yield With aluminium trichloride In nitromethane for 0.5h; Ambient temperature; 87% -
-
129-79-3
2,4,7-trinitrofluorene-9-one
Conditions
Conditions Yield With zinc(II) chloride at 160 - 165℃; for 3h; 87% Conditions
Conditions Yield With zinc(II) chloride at 160 - 165℃; for 4h; 87% Conditions
Conditions Yield With zinc(II) chloride at 150 - 170℃; 87% Conditions
Conditions Yield With zinc(II) chloride at 200 - 205℃; for 2h; 86% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
340017-16-5
[Au3(MeN=COMe)3]*[2,4,7-trinitro-9-fluorenone]
Conditions
Conditions Yield In dichloromethane adding soln. of fluorenone deriv. in CH2Cl2 to soln. of Au complex in CH2Cl2, shaking for a few minutes; filtration, concn. under reduced pressure, crystn. by layering with EtOHand standing for several days at room temp., washing crystals with Et2O , drying in vac.; 86% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
24544-04-5
2,6-diisopropylbenzenamine
Conditions
Conditions Yield With zinc(II) chloride at 150 - 170℃; 85% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
129012-68-6
4-(1'-methylethylideneaminooxy)butanoic acid
Conditions
Conditions Yield With sulfuric acid In acetic acid at 70℃; for 18h; Dean-Stark; 82% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
84408-99-1
4-hydroxy-2,7-dinitrofluorenone
Conditions
Conditions Yield With water In N,N,N,N,N,N-hexamethylphosphoric triamide three month at r.t.; 80% With water In N,N,N,N,N,N-hexamethylphosphoric triamide Conditions
Conditions Yield With zinc(II) chloride at 190 - 195℃; for 1.5h; 80% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
340017-22-3
[Au3(CH3NCOC2H5)3]

-
-
340017-26-7
[Au3(MeN=COEt)3]2*[2,4,7-trinitro-9-fluorenone]
Conditions
Conditions Yield In dichloromethane adding soln. of fluorenone deriv. in CH2Cl2 to soln. of Au complex in CH2Cl2; filtration, concn. under reduced pressure, crystn. by layering with EtOHand standing for several days at room temp., washing crystals with hexa ne, drying in vac.; 78% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
82766-04-9
3,8,10-trinitro-6H-dibenzopyran-6-one
Conditions
Conditions Yield With sulfuric acid; dihydrogen peroxide 76% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
21524-36-7
2,4,6-triisopropylaniline
Conditions
Conditions Yield With zinc(II) chloride at 150 - 170℃; 75% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
6673-15-0
2-(4-dimethylaminophenyl)ethylene-1,1,2-tricarbonitrile

-
-
72558-73-7
2,4,7-Trinitro-fluoren-9-one; compound with 2-cyano-3-(4-dimethylamino-phenyl)-but-2-enedinitrile
Conditions
Conditions Yield In ethanol for 15h; 74% Conditions
Conditions Yield With zinc(II) chloride at 175 - 180℃; for 6h; 73% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
62-53-3
aniline

-
-
6632-95-7
N-(2,4,7-trinitrofluorenylidene)aniline
Conditions
Conditions Yield With zinc(II) chloride at 185 - 190℃; for 1h; 73% Conditions
Conditions Yield With zinc(II) chloride at 150 - 170℃; 73% -
-
129-79-3
2,4,7-trinitrofluorene-9-one

-
-
364358-08-7
((Z,E)-2,4,7-trinitro-9-fluorenylideneaminooxy)undecan-11-ol
Conditions
Conditions Yield Stage #1: 2,4,7-trinitrofluorene-9-one; propane-2-one-O-(11-hydroxyundecyl)oxim With toluene-4-sulfonic acid In acetic acid Heating;
Stage #2: With toluene-4-sulfonic acid In ethanol for 16h; Heating;73% Conditions
Conditions Yield In chloroform at 20℃; for 0.166667h; 72% Trinitrofluorenone Chemical Properties
Molecular Structure of Trinitrofluorenone (CAS NO.129-79-3):
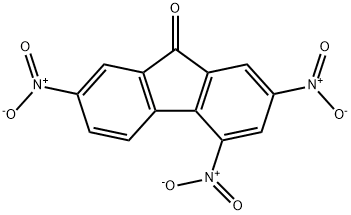
EINECS: 204-965-0
IUPAC Name: 2,4,7-Trinitrofluoren-9-one
Molecular Formula: C13H5N3O7
Molecular Weight: 315.194700 g/mol
XLogP3: 2.4
H-Bond Donor: 0
H-Bond Acceptor: 7
Canonical SMILES: C1=CC2=C(C=C1[N+](=O)[O-])C(=O)C3=CC(=CC(=C23)[N+](=O)[O-])[N+](=O)[O-]
InChI: InChI=1S/C13H5N3O7/c17-13-9-3-6(14(18)19)1-2-8(9)12-10(13)4-7(15(20)21)5-11(12)16(22)23/h1-5H
InChIKey: VHQGURIJMFPBKS-UHFFFAOYSA-N
Index of Refraction: 1.751
Molar Refractivity: 73.56 cm3
Molar Volume: 180.3 cm3
Surface Tension: 93.4 dyne/cm
Density: 1.747 g/cm3
Flash Point: 292.3 °C
Enthalpy of Vaporization: 84.47 kJ/mol
Boiling Point: 561.7 °C at 760 mmHg
Vapour Pressure: 1.2E-12 mmHg at 25 °C
Melting Point: 176-178°C
Water Solubility: 2.712 mg/L at 25 °CTrinitrofluorenone Toxicity Data With Reference
1. skn-rbt 500 mg/24H MLD
EPASR* United States Environmental Protection Agency, Office of Pesticides and Toxic Substances. (U.S. Environmental Protection Agency, 401M St., S.W., Washington, DC 20460) History Unknown 8EHQ-0480-0339 . 2. eye-rbt 100 mg MLD
EPASR* United States Environmental Protection Agency, Office of Pesticides and Toxic Substances. (U.S. Environmental Protection Agency, 401M St., S.W., Washington, DC 20460) History Unknown 8EHQ-0480-0339 . 3. mma-sat 10 µL/plate
EPASR* United States Environmental Protection Agency, Office of Pesticides and Toxic Substances. (U.S. Environmental Protection Agency, 401M St., S.W., Washington, DC 20460) History Unknown 8EHQ-0280-0333 . 4. sce-hmn:lym 3 mg/L
MUREAV Mutation Research. 138 (1984),181. 5. orl-rat LD50:9910 mg/kg
PESTC* Pesticide and Toxic Chemical News. 8 (1980),4. Trinitrofluorenone Consensus Reports
Reported in EPA TSCA Inventory.
Trinitrofluorenone Safety Profile
Safety Information of Trinitrofluorenone (CAS NO.129-79-3):
Risk Statements: 23/24/25-33
R23/24/25:Toxic by inhalation, in contact with skin and if swallowed
R33:Danger of cumulative effects
Safety Statements: 9-16-36/37/39
S9:Keep container in a well-ventilated place
S16:Keep away from sources of ignition
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection
RIDADR: 387
HazardClass: 1.1D
PackingGroup: II
Suspected carcinogen with experimental tumorigenic data. Mildly toxic by ingestion. Human mutation data reported. A skin and eye irritant. When heated to decomposition it emits highly toxic fumes of NOx. See also NITRO COMPOUNDS of AROMATIC HYDROCARBONS and KETONES.Trinitrofluorenone Standards and Recommendations
DFG MAK: Confirmed Animal Carcinogen with Unknown Relevance to Humans
Trinitrofluorenone Analytical Methods
For occupational chemical analysis use NIOSH: see 2,4,7-TRINITROFLUOREN-9-ONE.
Trinitrofluorenone Specification
Trinitrofluorenone is pale yellow needles or yellow powder with CAS Registry Number of 129-79-3. It is also called for 2,4,7-Trinitro-9H-fluoren-9-one ; 2,4,7-Trinitrofluorenone ; 2,4,7-Trinitrofluoren-9-one ; 2,4,7-Trinitrofluorenone ; 2,4,7-Trinitrofluorone ; 9H-Fluoren-9-one, 2,4,7-trinitro- ; AI3-17164 ; CCRIS 1298 ; Fluoren-9-one, 2,4,7-trinitro- ; HSDB 5408 ; NSC 12367 ; TNF ; TNF (ketone) ; 9H-Fluoren-9-one, 2,4,7-trinitro- . Trinitrofluorenone is often used as a chemical reagent.
Related Products
- Trinitrofluorenone
- 129799-08-2
- 129799-11-7
- 129799-15-1
- 129809-09-2
- 129811-26-3
- 129-81-7
- 129820-74-2
- 129822-48-6
- 129823-21-8
- 129825-11-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View