-
Name
Zinc sulfide (hexagonal)
- EINECS 215-251-3
- CAS No. 1314-98-3
- Density 4.1 g/mL at 25 °C(lit.)
- Solubility Insoluble in water
- Melting Point 1700 °C
- Formula ZnS
- Boiling Point 1185 °C(Press: 800 Torr)
- Molecular Weight 97.43
- Flash Point
- Transport Information
- Appearance white to yellow powder, clear to yellow crystals
- Safety 50A
- Risk Codes 31
-
Molecular Structure
- Hazard Symbols 31:;
- Synonyms LitoponeL;Pigment Black 17;Pigment White 7;RAK-LC;RAK-S;Sachtolith;Sachtolith HD;Sachtolith L;ZNI 08PB;Zinc monosulfide;zinc sulphide;Albalith;C.I. 77975;C.I. Pigment Black 17;C.I. Pigment White7;Cleartran;FV 40-1;HD-S;Irtran 2;KV 2;
- PSA 32.09000
- LogP 0.64570
Zinc sulfide Chemical Properties
Molecular Formula: ZnS
Formula Weight: 97.46
EINECS: 215-251-3
Density: 4.1 g/mL at 25 °C(lit.)
Melting point: 1700 °C
Storage tempreture: 2-8 °C
Appearance: White to yellow powder, clear to yellow crystals
Structure of Zinc sulfide (CAS NO.1314-98-3):
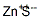
IUPAC Name: Sulfanylidenezinc
Canonical SMILES: S=[Zn]
InChI: InChI=1S/S.Zn
InChIKey: WGPCGCOKHWGKJJ-UHFFFAOYSA-N
Product Category of Zinc sulfide (CAS NO.1314-98-3): Inorganics;Chemical Synthesis;Catalysis and Inorganic Chemistry;Metal and Ceramic Science;Electronic Chemicals;Micro/Nanoelectronics
Zinc sulfide Uses
Zinc sulfide (CAS NO.1314-98-3) was used by Ernest Rutherford and others in the early years of nuclear physics as a scintillation detector, and is also used as an infrared optical material, transmitting from visible wavelengths to over 12 micrometres. It can be used planar as an optical window or shaped into a lens, and is used as phosphor in many applications, from cathode ray tubes through x-ray screens to glow in the dark products.
Zinc sulfide Production
Zinc sulfide (CAS NO.1314-98-3) is easily produced by mixing an amount of zinc and sulfur and then igniting it: Zn2+ + S2 → ZnS .This has formed the basis of a gravimetric analysis for zinc.
Zinc sulfide Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LC50 | inhalation | > 5040mg/m3/4H (5040mg/m3) | LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES SKIN AND APPENDAGES (SKIN): HAIR: OTHER | Research and Consulting Company, Technical Reports. Vol. NOTOX1072, Pg. 1989, |
| rat | LD50 | oral | > 2gm/kg (2000mg/kg) | Research and Consulting Company, Technical Reports. Vol. NOTOX1072/1332, Pg. 1989, | |
| rat | LD50 | skin | > 2gm/kg (2000mg/kg) | Research and Consulting Company, Technical Reports. Vol. NOTOX1072/1333, Pg. 1989, |
Zinc sulfide Consensus Reports
Reported in EPA TSCA Inventory.
Zinc sulfide Safety Profile
Risk Statements: 31
R31 :Contact with acids liberates toxic gas.
Safety Statements: 50A
S50:Do not mix with ...
WGK Germany: 3
RTECS: ZH5400000
Moderately toxic by ingestion, inhalation, and skin contact. When heated to decomposition it emits toxic vapors of Zn and SOx.
Zinc sulfide Specification
Zinc sulfide , its cas register number is 1314-98-3. It also can be called C.I. Pigment White 7 ; Cleartran ; and Zinc monosulfide . It is typically encountered in the more stable cubic form, known also as zinc blende or sphalerite. The first time its phosphorescence was observed by the French chemist Théodore Sidot in 1866. It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product: Wash mouth out with water, and get medical aid immediately. Notes to physician: Treat supportively and symptomatically.
In addition, Zinc sulfide (CAS NO.1314-98-3) could be stable under normal temperatures and pressures. It is not compatible with strong oxidizing agents, and you must not take it with incompatible materials. And also prevent it to broken down into hazardous decomposition products: Oxides of sulfur, hydrogen sulfide, zinc oxides.
Related Products
- Zinc
- Zinc chromate
- Zinc chromate hydroxides
- ZINC CHROMATE with ZINC HYDROXIDE and CHROMIUM OXIDE (9:1)
- Zinc citrate
- Zinc citrate dihydrate
- Zinc cyanide
- Zinc cyclohexanebutyrate
- Zinc dibenzoate
- Zinc dibenzyldithiocarbamate
- 13150-00-0
- 1315-01-1
- 1315-04-4
- 1315-05-5
- 1315-07-7
- 1315-09-9
- 13151-04-7
- 131511-13-2
- 1315-11-3
- 131512-74-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View