-
Name
alpha-Chloralose
- EINECS 240-016-7
- CAS No. 15879-93-3
- Article Data11
- CAS DataBase
- Density 1.773 g/cm3
- Solubility 0.44 g/100 mL (15 °C) in water
- Melting Point 178-182 °C
- Formula C8H11Cl3O6
- Boiling Point 504.4 °C at 760 mmHg
- Molecular Weight 309.531
- Flash Point 258.9 °C
- Transport Information UN 3249
- Appearance needle-like crystals or powder
- Safety 16-24/25-28-28A
- Risk Codes 20/22
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms Chloralose(6CI);Chloralose, a- (8CI);a-D-Glucofuranose,1,2-O-(2,2,2-trichloroethylidene)-, (R)-;AGC;AGC(carbohydrate);Alfa 4;Alpha 4;Alphakil;Anhydroglucochloral;Aphosal;Chloralosane;Dorcalm;Dulcidor;Glucochloral;Glucochloralose;Kalmettumsomniferum;Murex;Murex (carbohydrate);Somio;a-Chloralose;a-D-Chloralose;a-Monotrichloroethylidene-D-glucose;
- PSA 88.38000
- LogP -0.46290
Synthetic route


| Conditions | Yield |
|---|---|
| With sulfuric acid |


-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| With methanol; sodium methylate |
-
-
39598-39-5
α-D-chloralose

-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: pyridine 2: MeONa; MeOH View Scheme |

| Conditions | Yield |
|---|---|
| In chloroform |

-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| With 4,4-dimethyl-5-methylene-1,3-dioxolan-2-one; 1,8-diazabicyclo[5.4.0]undec-7-ene In N,N-dimethyl-formamide at 25℃; for 24h; | 90% |
-
-
77-76-9
2,2-dimethoxy-propane

-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In N,N-dimethyl-formamide at 20℃; for 24h; | 84% |

| Conditions | Yield |
|---|---|
| With dmap In N,N-dimethyl-formamide at 25℃; for 24h; | 80% |
-
-
15879-93-3
α-chloralose

-
-
124595-32-0
(R)-1,2-O-(2,2,2-trichloroethylidene)-α-D-xylo-1,4-furanodialdose

| Conditions | Yield |
|---|---|
| With sodium periodate In methanol; water at 20℃; for 2h; | 78% |
| With sodium periodate In methanol; water at 20℃; for 4h; | 76% |
| With periodic acid | |
| With sodium periodate In methanol; water |
-
-
15879-93-3
α-chloralose

-
-
187029-11-4
1,2-O-R-(2,2,2-trichloroethylidene)-α-D-glucofuranose 3,5,6-phosphite

| Conditions | Yield |
|---|---|
| With hexaethylphosphorous triamide at 100 - 110℃; for 2h; | 74% |
-
-
67-64-1
acetone

-
-
15879-93-3
α-chloralose

-
-
88390-57-2
5,6-O-isopropylidene-1,2-O-trichloroethylidene-α-D-glucofuranose

| Conditions | Yield |
|---|---|
| With hydrogenchloride In N,N-dimethyl-formamide for 24h; Ambient temperature; | 71% |

| Conditions | Yield |
|---|---|
| In nitromethane at 40℃; for 0.333333h; | A 22.2% B 11% |

-
-
100-52-7
benzaldehyde

-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| With zinc(II) chloride |
-
-
75-87-6
chloral

-
-
15879-93-3
α-chloralose

-
-
4096-55-3
O1,O2;O5,O6-[(R,S)-bis-(2,2,2-trichloro-ethylidene)]-α-D-glucofuranose

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| With dinitrogen tetraoxide |
-
-
7732-18-5
water

-
-
15879-93-3
α-chloralose

-
A
-
50-00-0
formaldehyd

-
B
-
124595-32-0
(R)-1,2-O-(2,2,2-trichloroethylidene)-α-D-xylo-1,4-furanodialdose

-
-
10544-72-6
dinitrogen tetraoxide

-
-
15879-93-3
α-chloralose
-
-
7664-93-9
sulfuric acid

-
-
75-87-6
chloral

-
-
15879-93-3
α-chloralose

-
-
4096-55-3
O1,O2;O5,O6-[(R,S)-bis-(2,2,2-trichloro-ethylidene)]-α-D-glucofuranose

-
-
5435-44-9, 22243-66-9
(E)-3-Ureido-but-2-enoic acid ethyl ester

-
-
75-36-5
acetyl chloride

-
-
15879-93-3
α-chloralose

-
-
75-36-5
acetyl chloride

-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| With zinc(II) chloride D&r-1tF,2ξF,3rF,4-tetraacetoxy-1catF-<(2R)-5ξ-acetoxy-2r-trichloromethyl-<1,3>dioxolan-4c-yl>-butane of mp: 151.5 degree; |

-
-
75-36-5
acetyl chloride

-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| With zinc(II) chloride higher-melting-Dr-1tF,2ξF,3rF,4-tetraacetoxy-1catF-<(2R)-5ξ-acetoxy-2r-trichloromethyl-<1,3>dioxolan-4c-yl>-butane; |

-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 78 percent / NaIO4 / H2O; methanol / 2 h / 20 °C 2: dimethylformamide / 2 h / 100 °C View Scheme |
-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 78 percent / NaIO4 / H2O; methanol / 2 h / 20 °C 2: dimethylformamide / 2 h / 100 °C View Scheme |
-
-
15879-93-3
α-chloralose

-
-
620593-16-0
3-O-acetyl-(R)-1,2-O-trichloroethylidene-5,6,8-trideoxy-α-D-xylo-oct-5(E)-eno-1,4-furano-7-ulose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 78 percent / NaIO4 / H2O; methanol / 2 h / 20 °C 2: dimethylformamide / 2 h / 100 °C 3: 0.99 g / pyridine / 20 °C View Scheme |
-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 78 percent / NaIO4 / H2O; methanol / 2 h / 20 °C 2: dimethylformamide / 2 h / 100 °C 3: 0.98 g / pyridine / 20 °C View Scheme |
-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 74 percent / hexaethyl phosphoroustriamide / 2 h / 100 - 110 °C 2: 83 percent / Cl2 / CH2Cl2 / -25 °C 3: 58 percent / acetonitrile / 0.33 h / Ambient temperature View Scheme |
-
-
15879-93-3
α-chloralose

-
-
187029-12-5
(2R,4S,4aS,5aR,7R,8aR,8bS)-2-Chloro-4-chloromethyl-7-trichloromethyl-tetrahydro-[1,3]dioxolo[4,5]furo[3,2-d][1,3,2]dioxaphosphinine 2-oxide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 74 percent / hexaethyl phosphoroustriamide / 2 h / 100 - 110 °C 2: 83 percent / Cl2 / CH2Cl2 / -25 °C View Scheme |
-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 74 percent / hexaethyl phosphoroustriamide / 2 h / 100 - 110 °C 2: 83 percent / Cl2 / CH2Cl2 / -25 °C 3: 61 percent / acetonitrile / 1 h / Ambient temperature View Scheme |
-
-
15879-93-3
α-chloralose

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 74 percent / hexaethyl phosphoroustriamide / 2 h / 100 - 110 °C 2: 72 percent / CH2Cl2 / 24 h / Ambient temperature View Scheme | |
| Multi-step reaction with 3 steps 1: 74 percent / hexaethyl phosphoroustriamide / 2 h / 100 - 110 °C 2: 83 percent / Cl2 / CH2Cl2 / -25 °C 3: acetonitrile / 0.33 h / Ambient temperature View Scheme |
alpha-Chloralose Chemical Properties
Chemical Name: alpha-Chloralose
IUPAC NAME: (1R)-1-[(2R,3aR,5R,6S,6aR)-6-Hydroxy-2-(trichloromethyl)-3a,5,6,6a-tetrahydrofuro[2,3-d][1,3]dioxol-5-yl]ethane-1,2-diol
CAS No.: 15879-93-3
EINECS: 240-016-7
RTECS: FM9450000
RTECS Class: Agricultural Chemical and Pesticide ; Tumorigen ; Human Data
EC Class: Harmful
Molecular Formula: C8H11Cl3O6
Molecular Weight: 309.53 g/mol
Melting Point: 178-182 °C
Density: 1.773 g/cm3
Flash Point: 258.9 °C
Boiling Point: 504.4 °C at 760 mmHg
Following is the structure of α-d-Glucochloralose (15879-93-3):
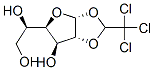
Product Categories about α-d-Glucochloralose (15879-93-3) are Biochemistry ; Glucose ; O-Substituted Sugars ; Sugars ; Dextrins、Sugar & Carbohydrates
The chemical synonymous of are (R)-1,2-O-(2,2,2-Trichloroethylidene)-alpha-D-glucofuranose ; 2-O-(2,2,2-Trichloroethylidene)-(R)-alpha-D-glucofuranos ; 2-O-(2,2,2-Trichloroethylidene)-(theta)-alpha-D-glucofuranos ; Alfamat ; Alpha-Chloralos ; Alphakil ; Aphosal ; Chloralosane
alpha-Chloralose Toxicity Data With Reference
| 1. | orl-rat LDLo:400 mg/kg | 85ESA3 Merck Index; an Encyclopedia of Chemicals, Drugs, and Biologicals. 9 (1976),260. | ||
| 2. | scu-rat LDLo:200 mg/kg | 27ZIAQ Drug Dosages in Laboratory Animals-A Handbook C.D. Barnes andL.G. Eltherington,Berkeley, CA.: Univ. of California Press,1973,70. | ||
| 3. | orl-mus LD50:200 mg/kg | FMCHA2 Farm Chemicals Handbook .(Meister Publishing,Willoughy, OH.: )1991,C68. | ||
| 4. | ipr-mus LD50:175 mg/kg | ARZNAD Arzneimittel-Forschung. Drug Research. 21 (1971),1727. | ||
| 5. | orl-dog LD50:250 mg/kg | RMVEAG Recueil de Medecine Veterinaire. 154 (1978),137. | ||
| 6. | ipr-dog LDLo:400 mg/kg | AIPTAK Archives Internationales de Pharmacodynamie et de Therapie. 3 (1897),191. | ||
| 7. | orl-cat LD50:250 mg/kg | RMVEAG Recueil de Medecine Veterinaire. 154 (1978),137. | ||
| 8. | ipr-cat LDLo:150 mg/kg | AIPTAK Archives Internationales de Pharmacodynamie et de Therapie. 3 (1897),191. |
alpha-Chloralose Consensus Reports
Reported in EPA TSCA Inventory.
alpha-Chloralose Safety Profile
Poison by ingestion, subcutaneous, and intraperitoneal routes. Questionable carcinogen with experimental tumorigenic data. When heated to decomposition it emits toxic fumes of Cl−.
Hazard Codes:
Xn: Harmful ![]()
Risk Statements about α-d-Glucochloralose (15879-93-3):
R20/22 Harmful by inhalation and if swallowed.
Safety Statements about α-d-Glucochloralose (15879-93-3):
S16 Keep away from sources of ignition - No smoking.
S24/25 Avoid contact with skin and eyes.
S28A After contact with skin, wash immediately with plenty of water.
Attention:
1. Storage: Store in a cool, dry place. Store in a tightly closed container.
2. Handling: Wash thoroughly after handling. Use with adequate ventilation. Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Do not ingest or inhale.
Related Products
- alpha-Chloralose
- 15880-03-2
- 158807-47-7
- 158816-55-8
- 158817-11-9
- 158817-45-9
- 158830-50-3
- 15883-20-2
- 1588-44-9
- 15884-65-8
- 158851-30-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View