-
Name
cis-1,2-Diaminocyclohexane
- EINECS 205-754-6
- CAS No. 1436-59-5
- Article Data30
- CAS DataBase
- Density 0.939 g/cm3
- Solubility Completely miscible in water
- Melting Point 8 °C
- Formula C6H14N2
- Boiling Point 193.643 °C at 760 mmHg
- Molecular Weight 114.191
- Flash Point 75 °C
- Transport Information UN 2735 8/PG 2
- Appearance Clear colorless liquid
- Safety 26-36/37/39-45
- Risk Codes 34
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms 1,2-Cyclohexanediamine,cis- (8CI);(R)(S)-1,2-Diaminocyclohexane;cis-(1S,2R)-1,2-Diaminocyclohexane;cis-1,2-Cyclohexanediamine;cis-1,2-Diaminocyclohexane;meso-1,2-Cyclohexanediamine;meso-1,2-Diaminocyclohexane;
- PSA 52.04000
- LogP 1.61560
Synthetic route
-
-
65433-80-9
cis-cyclohexane-1,2-diammonium sulfate

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water for 1.16667h; | 67% |
-
-
1460-57-7
trans-1,2-cyclohexandiol

-
A
-
4006-50-2
1,2,3,4,6,7,8,9-octahydrophenazine

-
B
-
6850-38-0
2-aminocyclohexanol

-
C
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| With dodecacarbonyl-triangulo-triruthenium; 4,5-bis((diisopropylphosphanyl)methyl)acridine; ammonia In tert-Amyl alcohol at 170℃; for 21h; Temperature; Inert atmosphere; Schlenk technique; Autoclave; | A 44.8% B n/a C 45.9% |


| Conditions | Yield |
|---|---|
| With DL-tartaric acid In ethanol at 20℃; Reflux; Large scale; | 19.2% |

| Conditions | Yield |
|---|---|
| With chloroform; tris-(2-chloro-ethyl)-amine at 40℃; Erhitzen des Reaktionsprodukts mit methanol.KOH bis auf 130grad; | |
| With sodium azide; chloroform; sulfuric acid at 40℃; Erhitzen des Reaktionsprodukts mit methanol.KOH bis auf 130grad; |
-
-
18886-70-9
cis-cyclohexane-1,2-dicarboxylic acid dihydrazide

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium nitrite Erhitzen des Reaktionsprodukts mit methanol.KOH bis auf 130grad; |
-
-
2802-07-5
1,3-cyclohexanedionedioxime

-
A
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
3385-21-5, 26772-34-9, 26883-70-5, 115092-57-4
trans-cyclohexane-1,3-diamine

| Conditions | Yield |
|---|---|
| With acetic acid; platinum Hydrogenation; |
-
-
75730-13-1, 86694-16-8, 116836-89-6
diethyl (1RS,2SR)-cyclohexane-1,2-diylbiscarbamate

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 120℃; |
-
-
875923-28-7
(3aRS,7aSR)-octahydro-2H-benzimidazol-2-one

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| With sulfuric acid at 105℃; |
-
-
10027-79-9
(1R,2S)-1,2-diazidocyclohexane

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| With hydrogen; platinum(IV) oxide In ethanol |
-
-
85782-28-1, 95647-96-4
(3aR,7aS)-2-Ethoxy-3a,4,5,6,7,7a-hexahydro-1H-benzoimidazole

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| With barium dihydroxide at 120℃; for 18h; Yield given; |
-
-
85782-31-6
(1R,6S)-7-Aza-bicyclo[4.1.0]heptane-7-carboximidic acid ethyl ester

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 90 percent / NaI / dimethylformamide / 48 h 2: Ba(OH)2 / 18 h / 120 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) N-bromosuccinimide, 2.) HCl, 3.) NaOH 2: 90 percent / NaI / dimethylformamide / 48 h 3: Ba(OH)2 / 18 h / 120 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 3,6,9-trioxaundecane 2: ethanol; nickel / 135 - 145 °C / 88260.9 - 91938.4 Torr / Hydrogenation 3: sulfuric acid / 105 °C View Scheme |

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Py / 0 - 5 °C 2: NaN3 / dimethylformamide; H2O 3: H2 / PtO2 / ethanol View Scheme |
-
-
10039-14-2, 28141-32-4, 105230-11-3, 116051-21-9, 122673-84-1, 122673-91-0
2-iodo-1-cyclohexanol

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: NaN3 / ethanol / 24 h / Heating 2: Py / 0 - 5 °C 3: NaN3 / dimethylformamide; H2O 4: H2 / PtO2 / ethanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: NaN3, NH4Cl / aq. ethanol / 24 h / Heating 2: Py / 0 - 5 °C 3: NaN3 / dimethylformamide; H2O 4: H2 / PtO2 / ethanol View Scheme | |
| Multi-step reaction with 5 steps 1: aq. HI / 0 °C 2: NaN3 / ethanol / 24 h / Heating 3: Py / 0 - 5 °C 4: NaN3 / dimethylformamide; H2O 5: H2 / PtO2 / ethanol View Scheme |

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaN3 / dimethylformamide; H2O 2: H2 / PtO2 / ethanol View Scheme |
-
-
694-83-7
1,2-diaminocyclohexane

-
A
-
20439-47-8
(1R,2R)-1,2-diaminocyclohexane

-
B
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
C
-
21436-03-3
(S,S)-1,2-diaminocyclohexane

| Conditions | Yield |
|---|---|
| separation by salt formation with (+)-L-tartaric acid; | A 2.0 g B n/a C 0.9 g |

-
-
488-31-3, 608-54-8, 608-55-9, 6703-05-5, 10158-64-2, 20869-04-9, 33012-62-3
(2S,4S)-xylaric acid

-
-
694-83-7
1,2-diaminocyclohexane

-
B
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| In methanol at 80℃; for 3h; | A 177.3 g B n/a |

-
-
6850-38-0
2-aminocyclohexanol

-
A
-
20439-47-8
(1R,2R)-1,2-diaminocyclohexane

-
B
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| Stage #1: 2-aminocyclohexanol In ethanol for 1h; Stage #2: With ammonia; hydrogen In ethanol at 160℃; under 3000.3 - 11251.1 Torr; for 16h; Pressure; Reagent/catalyst; Solvent; Temperature; Overall yield = 98 %; |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
108-24-7
acetic anhydride

-
-
70924-77-5
cis-1,2-diacetamidocyclohexane

| Conditions | Yield |
|---|---|
| In chloroform for 1h; | 100% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; | 100% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
70197-13-6
methyltrioxorhenium(VII)

| Conditions | Yield |
|---|---|
| In toluene equimolar organic ligand added to Re-compound in toluene at room temp.; filtrated; washed with n-hexane; elem. anal.; | 100% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| In toluene for 1h; Reflux; | 98% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 150 - 170℃; for 3.5h; Microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 24h; Inert atmosphere; Reflux; regioselective reaction; | 97% |

| Conditions | Yield |
|---|---|
| In ethanol at 75℃; for 14h; Inert atmosphere; | 97% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
786712-01-4
cis-N,N'-bis-(p-toluenesulfonyl)-1,2-diaminocyclohexane

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 20℃; for 15h; | 96% |
-
-
12354-84-6, 12354-85-7
bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.5h; | 96% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| In diethylamine at 25℃; for 5h; | 96% |
-
-
504-64-3
carbon suboxide

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
135039-13-3
2,6-Diaza-cis-bicyclo<5.4.0>undecan-3,5-dione

| Conditions | Yield |
|---|---|
| In chloroform for 1h; Ambient temperature; | 95% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
152523-69-8
3,3'-<<1,1'-binaphthalene>-2,2'-diylbis(oxy-2,1-ethanediyloxy-2,1-ethanediyloxy)>bis<2-hydroxybenzaldehyde>

| Conditions | Yield |
|---|---|
| With barium triflate In tetrahydrofuran refluxing (30 min); solvent evapn., dissoln. (CH2Cl2), washing (water, aq. Na2SO4), evapn., flash column chromy. (MeOH/CH2Cl2 3/97), pptn. (CH2Cl2/cyclohexane); elem. anal.; | 95% |

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
1363198-59-7
cis-1,2-dinitrocyclohexane

| Conditions | Yield |
|---|---|
| With water; fluorine In dichloromethane; acetonitrile at 20℃; for 0.5h; Inert atmosphere; Flow reactor; | 95% |

| Conditions | Yield |
|---|---|
| With triethylamine for 48h; Ambient temperature; | 94% |

| Conditions | Yield |
|---|---|
| In ethanol for 2h; Inert atmosphere; Reflux; | 94% |
-
-
4411-26-1
1-adamantylisothiocyanate

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
B
-
25348-94-1
N,N'-bis(tricyclo[3.3.1.1(3,7)]dec-1-yl)thiourea

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 18h; Reflux; Inert atmosphere; | A 93% B 4% |


-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 2h; | 93% |
-
-
590-92-1
3-Bromopropionic acid

-
-
931-53-3
Cyclohexyl isocyanide

-
-
1074-12-0
phenylglyoxal hydrate

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| Stage #1: phenylglyoxal hydrate; cis-cyclohexane-1,2-diamine In methanol at 20℃; for 0.166667h; Ugi Condensation; Stage #2: 3-Bromopropionic acid; Cyclohexyl isocyanide In methanol for 24h; diastereoselective reaction; | 93% |

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 24h; | 92% |

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| In ethanol Heating; | 92% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 150 - 170℃; for 3.5h; Microwave irradiation; | 92% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 24h; | 91% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 2h; | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: undec-10-enoyl chloride; cis-cyclohexane-1,2-diamine In 1,4-dioxane at 0 - 20℃; Stage #2: With boron trifluoride diethyl etherate In 1,4-dioxane at 130℃; | 90% |
-
-
15022-08-9
diallylcarbonate

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
1261082-38-5
allyl ((1R,2S)-2-aminocyclohexyl)carbamate

| Conditions | Yield |
|---|---|
| With Novozyme 435 In toluene at 20℃; for 96h; Enzymatic reaction; | 90% |
| With candida antarctica novozyme-435 In toluene at 20℃; for 96h; Enzymatic reaction; | 90% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
90-02-8
salicylaldehyde

-
-
41013-27-8
(1R,2S)-cis-N,N'-bis(salicylidene)-1,2-cyclohexanediamine

| Conditions | Yield |
|---|---|
| In ethanol | 89% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
152523-70-1
3,3'-<<1,1'-binaphthalene>-2,2'-diylbis(oxy-2,1-ethanediyloxy-2,1-ethanediyloxy-2,1-ethanediyloxy)>bis<2-hydroxybenzaldehyde>

| Conditions | Yield |
|---|---|
| With barium triflate In tetrahydrofuran refluxing (30 min); solvent evapn., dissoln. (CH2Cl2), washing (water, aq. Na2SO4), evapn., flash column chromy. (MeOH/CH2Cl2 3/97), pptn. (CH2Cl2/cyclohexane); elem. anal.; | 89% |

-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
154824-43-8
2-(3-formyl-2-hydroxyphenoxy)-N-<4-(n-octyloxy)phenyl>acetamide

| Conditions | Yield |
|---|---|
| With H2O In methanol under Ar; the aldehyde and the amine were refluxed in methanol for 1 h, UO2(OAc)2 in methanol was added, refluxed for 1 h; cooled, filtered, washed with methanol; elem. anal.; | 89% |


| Conditions | Yield |
|---|---|
| Stage #1: cis-cyclohexane-1,2-diamine; octadec-9-enoyl chloride In 1,4-dioxane at 0 - 20℃; Stage #2: With boron trifluoride diethyl etherate In 1,4-dioxane at 130℃; | 89% |
-
-
1436-59-5
cis-cyclohexane-1,2-diamine

-
-
109-94-4
formic acid ethyl ester

-
-
75599-23-4
(1R,2S)-N1,N2- dimethylcyclohexane-1,2-diamine

| Conditions | Yield |
|---|---|
| Stage #1: cis-cyclohexane-1,2-diamine; formic acid ethyl ester Reflux; Stage #2: With lithium aluminium tetrahydride In tetrahydrofuran at 20℃; Reflux; Stage #3: With water; sodium hydroxide In tetrahydrofuran | 89% |
cis-1,2-Diaminocyclohexane Chemical Properties
Molecular Structure of cis-1,2-Diaminocyclohexane (CAS No.1436-59-5):
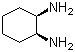
Molecular Formula: C6H14N2
Molecular Weight: 114.19
CAS No: 1436-59-5
XLogP3-AA: -0.3
H-Bond Donor: 2
H-Bond Acceptor: 2
Rotatable Bond Count: 0
Exact Mass: 114.115698
MonoIsotopic Mass: 114.115698
Topological Polar Surface Area: 52
Heavy Atom Count: 8
Formal Charge: 0
Complexity: 62.9
Isotope Atom Count: 0
Defined Atom StereoCenter Count: 0
Undefined Atom StereoCenter Count: 2
Defined Bond StereoCenter Count: 0
Undefined Bond StereoCenter Count: 0
Covalently-Bonded Unit Count: 1
Index of Refraction: 1.483
Molar Refractivity: 34.77 cm3
Molar Volume: 121.5 cm3
Surface Tension: 37 dyne/cm
Density: 0.939 g/cm3
Flash Point: 75 °C
Enthalpy of Vaporization: 42.98 kJ/mol
Boiling Point: 193.6 °C at 760 mmHg
Vapour Pressure: 0.46 mmHg at 25°C
Chemical Properties Clear colorless liquid
IUPAC Name: Cyclohexane-1,2-diamine
Canonical SMILES: C1CCC(C(C1)N)N
InChI: InChI=1S/C6H14N2/c7-5-3-1-2-4-6(5)8/h5-6H,1-4,7-8H2
InChIKey: SSJXIUAHEKJCMH-UHFFFAOYSA-N
cis-1,2-Diaminocyclohexane Safety Profile
Hazard Codes:  C
C
Risk Statements: 34
R34:Causes burns.
Safety Statements: 26-36/37/39-45
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
RIDADR: UN 2735 8/PG 2
WGK Germany: 3
F: 10-34
HazardClass: 8
PackingGroup: II
cis-1,2-Diaminocyclohexane Specification
cis-1,2-Diaminocyclohexane (CAS No.1436-59-5), it also can be called 1,2-Cyclohexanediamine ; Cyclohexane-1,2-diamine ; 1,2-Diaminocyclohexane, mixture of cis and trans .
Related Products
- cis-1,1-Dimethylethyl 4-amino-3-fluoro-1-piperidinecarboxylate
- cis-1,2,3,4-Tetrahydro-5-(oxiranylmethoxy)-2,3-naphthalenediol
- cis-1,2,3,6-Tetrahydrophthalic anhydride
- cis-1,2,3,6-Tetrahydrophthalimide
- cis-1,2,3,6-Tetrahydrophthalimide
- cis-1,2-Bis(diphenylphosphine)ethene
- cis-1,2-Cyclohexanedimethanamine
- cis-1,2-Cyclohexanediol
- cis-1,2-Cyclohexanediol diacetate
- cis-1,2-Cyclopentanediol
- 143664-11-3
- 143665-37-6
- 14366-74-6
- 143667-61-2
- 143-66-8
- 143673-66-9
- 143-67-9
- 143679-80-5
- 143683-20-9
- 14368-40-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View