hangzhou verychem science and technology co.ltd
Product name: Pinacolone Cas No: 75-97-8 Assay: 99%min Hangzhou Verychem Science And Technology Co. Ltd. was set up in year 2004, it’s a young but fast growing company. In the twelve years histo
Cas:75-97-8
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developm
Cas:75-97-8
Min.Order:1 Metric Ton
FOB Price: $1.0 / 3.0
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
Unique advantages for Pinacolone Cas 75-97-8 Guaranteed the purity High quality & competitive price Quality control Fast feedback Prompt shipment Appearance:Liquid Storage:Flammables area Package:200kg/drum Application: Agrochemical Inter
Cas:75-97-8
Min.Order:25 Kilogram
FOB Price: $1.0 / 60.0
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Appearance:White or off-white Solid Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
Product Name: Pinacolone Synonyms: ALPHA,ALPHA,ALPHA-TRIMETHYLACETONE;AKOS BBS-00004455;3,3-DIMETHYLBUTAN-2-ONE;3,3-DIMETHYL-2-BUTANONE;1,1,1-TRIMETHYLACETONE;METHYL TERT-BUTYL KETONE;TERT-BUTYL METHYL KETONE;PINACOLIN CAS: 75-97
Cas:75-97-8
Min.Order:1 Gram
FOB Price: $8900.0
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Chemical Name: Pinacolone Other Name: 3,3-DIMETHYL-2-BUTANONE Molecular formula :C6H12O Molecular weight : 100.16 CAS : 75-97-8 EINECS: 200-920-4 Usage: It is mainly used in agriculture intermediate, pesticide, solvents and organic i
Baoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At presen
Cas:75-97-8
Min.Order:1 Kilogram
FOB Price: $35.0 / 50.0
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:75-97-8
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Hangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
HANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:Colorless transparent liquid Storage:sealed in a cool place. Package:170kg/plastic drum; or as per your request Application:mainly used in agriculture intermediate, pesticide, solven
Hubei Vanz Pharm Co.,Ltd
ISO/factory/goodqualityAppearance:off white Storage:Dry,cool place Package:drum Application:active pharmaceutical ingredients Transportation:by air/sea/express Port:shenzhen/shanghai
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city?in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new api,?intermediat
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:75-97-8
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Xiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hubei Langyou International Trading Co., Ltd
phosalone CAS. NO.75-97-8 Application:phosalone CAS. NO.75-97-8
Luyunjia Chemistry Xiamen Limited
Xiamen Luyunjia Trading Co.,Ltd Package:1kg/bag; 4378kg/drum, or as customer's request. Application:Xiamen Luyunjia Trading Co.,Ltd Transportation:DHL, EMS, FedEx, TNT, AIR, SEA Port:Beijing,Shanghai,Guangzhou ,China main port
Hunan Longxianng Runhui Trading Co.,Ltd
phosalone CAS. NO.75-97-8Appearance:ask jude for details Storage:Dry and ventilated Package:in foil bag or customized Application:Pharmaceutical intermediates/Food additive Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small a
Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Synthetic route

| Conditions | Yield |
|---|---|
| With aluminium phosphate aluminium oxide catalyst Heating; | 100% |
| With silica-supported orthophosphoric acid In neat (no solvent) at 180℃; for 0.5h; Catalytic behavior; Temperature; Time; Pinacol Rearrangement; Green chemistry; | 100% |
| With [Fe3O(OOCC6H5)6(H2O)3]4[α-SiW12O40] In toluene at 99.84℃; for 6h; | 80% |
-
-
133619-37-1
C6H12OS

-
-
75-97-8
3,3-dimethyl-butan-2-one

| Conditions | Yield |
|---|---|
| Ambient temperature; | 100% |
| Ambient temperature; Yield given; |
-
-
5076-20-0
2,3-dimethyl-2,3-epoxybutane

-
A
-
75-97-8
3,3-dimethyl-butan-2-one

-
B
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
C
-
10473-13-9
2,3-dimethyl-1-buten-3-ol

| Conditions | Yield |
|---|---|
| With perchloric acid; sodium perchlorate at 25℃; Yields of byproduct given; | A n/a B 99.93% C n/a |
| With perchloric acid; sodium perchlorate at 25℃; Product distribution; heterogeneous reaction; |

-
-
464-07-3, 20281-91-8
3,3-dimethyl-2-butanol

-
-
75-97-8
3,3-dimethyl-butan-2-one

| Conditions | Yield |
|---|---|
| With 2,2,6,6-tetramethyl-piperidine-N-oxyl; trichloroisocyanuric acid; sodium hydrogencarbonate; sodium bromide In water; acetone at 20℃; for 1h; | 98% |
| With dihydrogen peroxide In water; tert-butyl alcohol at 80℃; for 24h; | 97% |
| With Ni(NTf2)2·xH2O; 4-mercapto-4-oxide-dinaphtho<2,1-d:1',2'-f><1,3,2>dioxaphosphepin; 9-(2-mesityl)-10-methylacridinium perchlorate In dichloromethane at 27 - 29℃; for 20h; Inert atmosphere; Irradiation; | 91% |

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; 6C6H15P*4CF3O3S(1-)*2Ru(2+); acetone at 20℃; for 0.75h; | 98% |
-
-
10341-64-7, 20259-50-1, 2475-93-6
pinacolone oxime

-
-
75-97-8
3,3-dimethyl-butan-2-one

| Conditions | Yield |
|---|---|
| With silica gel; copper(II) nitrate In tetrachloromethane for 1h; Heating; | 97% |
| With dihydrogen peroxide; tripropylammonium fluorochromate (VI) In acetone at 0 - 10℃; for 1.5h; | 97% |
| With sodium tungstate; dihydrogen peroxide In water; acetone at -5 - 20℃; for 5h; | 70% |
-
-
39263-43-9
3,3-dimethyl-butan-2-one-phenylhydrazone

-
-
75-97-8
3,3-dimethyl-butan-2-one

| Conditions | Yield |
|---|---|
| With baker's yeast; phosphate buffer In ethanol at 37℃; for 14h; | 93% |
| With iron(II) sulfate In chloroform at 20℃; for 1h; Hydrolysis; | 91% |
-
-
26785-73-9
2-(t-butyl)-2-methyl-1,3-dithiolane

-
-
75-97-8
3,3-dimethyl-butan-2-one

| Conditions | Yield |
|---|---|
| With silica gel; copper(II) nitrate In tetrachloromethane for 0.25h; Ambient temperature; | 93% |
-
-
35856-82-7
3,3,4,4-tetramethyl-1,2-dioxetane

-
-
1486-28-8
diphenyl(methyl)phosphine

-
A
-
75-97-8
3,3-dimethyl-butan-2-one

-
B
-
88635-85-2
2,2-dihydro-4,4,5,5-tetramethyl-2-methyl-2,2-diphenyl-1,3,2-dioxaphospholane

| Conditions | Yield |
|---|---|
| In benzene-d6 at 19.5℃; Kinetics; other temperatures; | A 7% B 92% |
| In benzene-d6 at 19.5℃; Kinetics; Rate constant; Thermodynamic data; other temperatures, different concentrations, isotope effects; Ea, ΔS(excit); | A 8% B 92% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydroquinone In diphenylether at 75℃; for 6h; Temperature; Solvent; Concentration; Reagent/catalyst; | 91.1% |
-
-
35856-82-7
3,3,4,4-tetramethyl-1,2-dioxetane

-
-
88635-84-1
dimethyl-d6-phenylphosphine

-
A
-
75-97-8
3,3-dimethyl-butan-2-one

-
B
-
88635-88-5
2,2-dihydro-4,4,5,5-tetramethyl-2,2-d6-dimethyl-2-phenyl-1,3,2-dioxaphospholane

| Conditions | Yield |
|---|---|
| In benzene-d6 at 19.5℃; Kinetics; Rate constant; Thermodynamic data; other temperatures, Ea, ΔS(excit); | A 9% B 91% |
| In benzene-d6 at 19.5℃; | A 9% B 91% |

-
-
106-44-5
p-cresol

-
A
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
125847-62-3, 131235-33-1
(P(CH3)3)4Ru(OC6H4CH3)2

| Conditions | Yield |
|---|---|
| In benzene-d6 inert gas, p-cresol added as solid; not isolated; NMR; | A >99 B 91% |

-
-
35856-82-7
3,3,4,4-tetramethyl-1,2-dioxetane

-
-
672-66-2
Dimethyl(phenyl)phosphine

-
A
-
75-97-8
3,3-dimethyl-butan-2-one

-
B
-
88635-87-4
2,2-dihydro-4,4,5,5-tetramethyl-2,2-dimethyl-2-phenyl-1,3,2-dioxaphospholane

| Conditions | Yield |
|---|---|
| In benzene-d6 at 19.5℃; Kinetics; Rate constant; Thermodynamic data; other temperatures, Ea, ΔS(excit); | A 10% B 90% |
| In benzene-d6 at 19.5℃; | A 10% B 90% |


| Conditions | Yield |
|---|---|
| With aluminium trichloride; copper(l) chloride; lithium chloride In tetrahydrofuran at 10℃; for 0.166667h; | 87% |
-
-
76-09-5
2,3-dimethyl-2,3-butane diol

-
A
-
75-97-8
3,3-dimethyl-butan-2-one

-
B
-
513-81-5
2,3-dimethyl-buta-1,3-diene

| Conditions | Yield |
|---|---|
| With H6S2V2W16O62 at 149.84℃; for 0.5h; Reagent/catalyst; Pinacol Rearrangement; | A 82% B 16% |
| With SBA-15 supported zirconium In toluene at 110℃; for 2h; Pinacol rearrangement; | A 81% B 19% |
| With Cu2+-montmorillonit In solid at 100℃; for 1h; Product distribution; effects of homoionic-montmorillonits contains various metal ions; | A 78% B 3% |

| Conditions | Yield |
|---|---|
| With lead(IV) acetate; oxygen; ozone In dichloromethane at 0℃; for 1.5h; | 82% |
-
-
98720-65-1
trans-t-BuOIr(CO)(PPh3)2

-
-
24762-44-5
triphenylphosphine-d15

-
A
-
75-97-8
3,3-dimethyl-butan-2-one

| Conditions | Yield |
|---|---|
| In (2)H8-toluene Kinetics; toluene-d7, PPh3, 95°C; not sepd., detected by (1)H-NMR and mass-spectra; | A 81% B n/a |


-
A
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
134002-31-6, 76171-49-8
dihydridotetrakis(trimethylphosphino)ruthenium(II)

| Conditions | Yield |
|---|---|
| With H2 In benzene-d6 inert gas, soln. of Ru-compound degassed, immersed in liquid nitrogen, exposed to H2 (450 Torr), sealed, heated to 45°C for 8 h; not isolated; NMR; | A 61% B 77% |

| Conditions | Yield |
|---|---|
| With bismuth oxide perchlorate In dichloromethane at 20℃; for 1h; Rearrangement; | 75% |
| at 600℃; | 13% |
| With tetrabutylammonium perchlorate In dichloromethane electrolysis; Yield given; |
-
-
10341-64-7, 20259-50-1, 2475-93-6
pinacolone oxime

-
-
74-86-2
acetylene

-
A
-
75-97-8
3,3-dimethyl-butan-2-one

-
B
-
5398-58-3
2-tert-butyl-1H-pyrrole

-
C
-
255376-82-0
O-Vinylpinacolonoxime

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide; pentane at 70℃; under 12160 Torr; for 0.1h; Addition; Cyclisation; Rearrangement; | A 1.9% B 1.9% C 74.8% |

-
-
2259-30-5
tert-butylmagnesium bromide

-
-
185245-65-2
3-methylthio-3-N-piperidino-1-phenyl-2-propen-1-one

-
A
-
75-97-8
3,3-dimethyl-butan-2-one

-
B
-
938-16-9
2,2-dimethylpropiophenone

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether for 4h; Ambient temperature; | A n/a B 66% |

-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
464-07-3, 20281-91-8
3,3-dimethyl-2-butanol

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; hydrogen; [RuCl2(Ph2PCH2CH2NH2)2] at 20℃; under 2280 Torr; for 12h; | 100% |
| With hydrogen; Ru((R,R)-cyP2N2)HCl In benzene-d6 at 20℃; under 2280.15 Torr; for 12h; Product distribution / selectivity; Alkaline conditions; Cooling with liquid nitrogen; | 100% |
| With hydrogen; Ru((R,R)-cyP2(NH)2)HCl In benzene-d6 at 20℃; under 2280.15 Torr; for 12h; Product distribution / selectivity; Alkaline conditions; Cooling with liquid nitrogen; | 100% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; hydrogen; [dichloro((S)-2,2'-bis(di-4-tolylphosphino)-1,1'-binaphthyl)(α-picolylamino)ruthenium(II)] In ethanol at 25℃; under 3800.26 - 15201 Torr; for 5 - 24h; Product distribution / selectivity; | 100% |
| With 4-methyl-morpholine; 6C6H15P*4CF3O3S(1-)*2Ru(2+); isopropyl alcohol In 2,2,2-trifluoroethanol at 45℃; for 3h; | 99% |
| With [dichloro((S)-2,2'-bis(di-4-tolylphosphino)-1,1'-binaphthyl)(α-picolylamino)ruthenium(II)]; potassium tert-butylate; hydrogen In ethanol at 25℃; under 3800 Torr; for 5h; | 98% |

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide In 1,4-dioxane; water Heating; | 100% |
| With selenium(IV) oxide at 50℃; for 5h; Temperature; | 45.5% |
| With selenium(IV) oxide; water In methanol for 20h; Reflux; | 21% |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
4114-31-2
ethylhydrazine carboxylate

-
-
78259-24-2
(1,2,2-trimethyl-propylidene)-carbazic acid ethyl ester

| Conditions | Yield |
|---|---|
| 100% | |
| With ethanol; acetic acid | |
| With acetic acid In ethanol |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
529-28-2
4-iodoanisol

-
-
75508-73-5
1-(2-methoxyphenyl)-3,3-dimethyl-2-butanone

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In ammonia at -33℃; Irradiation; | 100% |
| With potassium tert-butylate In water; dimethyl sulfoxide | |
| With hydrogenchloride; sodium amide In water; dimethyl sulfoxide |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
29586-83-2
(2Z)-3-chloro-4,4-dimethylpent-2-enenitrile

| Conditions | Yield |
|---|---|
| Stage #1: 3,3-dimethyl-butan-2-one; N,N-dimethyl-formamide With trichlorophosphate In 1,2-dichloro-ethane at 75℃; for 4h; Stage #2: With hydroxylamine sulfate In 1,2-dichloro-ethane at 85℃; for 1h; Further stages.; | 100% |
| Stage #1: 3,3-dimethyl-butan-2-one; N,N-dimethyl-formamide With trichlorophosphate In 1,2-dichloro-ethane at 0 - 75℃; for 4h; Inert atmosphere; Stage #2: With hydroxylamine hydrochloride In 1,2-dichloro-ethane at 80℃; for 2h; Inert atmosphere; | 56% |
| Stage #1: 3,3-dimethyl-butan-2-one; N,N-dimethyl-formamide With trichlorophosphate In 1,2-dichloro-ethane at 0 - 75℃; for 4h; Stage #2: With hydroxylamine hydrochloride In 1,2-dichloro-ethane at 80℃; for 2h; | 56% |
| With hydroxylamine sulfate; trichlorophosphate 1.) 1,2-dichloroethane, 50 - 55 deg C, 2 h; 70 - 75 deg C, 2 h, 2.) 85 deg C, 1 h; Yield given. Multistep reaction; |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
69739-34-0
t-butyldimethylsiyl triflate

-
-
84850-50-0
3,3-dimethyl-2-{[(1,1-dimethylethyl)dimethylsilyl]oxy}-1-butene

| Conditions | Yield |
|---|---|
| With triethylamine In benzene for 4h; Heating; | 100% |
| With 2,6-dimethylpyridine In dichloromethane at 0 - 20℃; for 5h; Sealed tube; Inert atmosphere; |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
241475-84-3
(meso-5,10,15,20-tetra-p-tolylporphyrinato)Hf=N-2,6-diisopropylphenyl

-
-
273927-92-7
(meso-5,10,15,20-tetra-p-tolylporphyrinato)Hf(NH-2,6-diisopropylphenyl)[OC(tBu)(=CH2)]

| Conditions | Yield |
|---|---|
| With Ph3CH In benzene-d6 under N2 atm. (TTP)Hf=NAriPr, pinacolone, and Ph3CH reacted in C6D6 at 25°C for 17 h; not isolated, identified by NMR spectroscopy; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromo-6-methylpyridine With n-butyllithium; diisopropylamine In tetrahydrofuran; hexane at -78 - 0℃; for 0.5h; Stage #2: 3,3-dimethyl-butan-2-one In tetrahydrofuran; hexane at -78 - 20℃; for 0.5h; | 100% |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
100-52-7
benzaldehyde

-
-
538-44-3, 28435-47-4, 29569-91-3
(E)-benzylidenepinacolone

| Conditions | Yield |
|---|---|
| With sodium hydroxide; dodecyl-α,ω-bis(dodecyldimethylammonium bromide) In water at 25℃; for 1.16667h; Sonication; | 99% |
| With barium(II) hydroxide In ethanol at 20℃; | 99% |
| barium dihydroxide In ethanol for 1h; Heating; | 96% |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
75-56-9, 16033-71-9
methyloxirane

-
-
139367-18-3
2,2-dimethyl-6-hydroxy-3-heptanone

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane; yttrium(III) trifluoromethanesulfonate In hexane; toluene at 0℃; for 18h; | 99% |
| With lithium perchlorate; lithium hexamethyldisilazane 1.) THF, 0 deg C, 15 min, 2.) 25 deg C, 72 h; Yield given. Multistep reaction; |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
7677-24-9
trimethylsilyl cyanide

-
-
219860-94-3
3,3-dimethyl-2-<(trimethylsilyl)oxy>-2-cyanobutane

| Conditions | Yield |
|---|---|
| With potassium carbonate at 20℃; for 4.5h; | 99% |
| With bis(triphenylphosphine)iminium chloride at 25℃; for 1h; Inert atmosphere; Neat (no solvent); | 97% |
| 1,4-diaza-bicyclo[2.2.2]octane at 20℃; | 94% |
| With zinc(II) iodide In dichloromethane at 0 - 20℃; |

| Conditions | Yield |
|---|---|
| Stage #1: 4-chlorobenzoyl chloride With diisopropylamine; Rink-amide resin In tetrahydrofuran at 20℃; for 18h; Stage #2: 3,3-dimethyl-butan-2-one With tris(dibenzylideneacetone)dipalladium (0); 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; sodium t-butanolate In 1,4-dioxane at 70℃; Buchwald-Hartwig coupling; Stage #3: With trifluoroacetic acid In dichloromethane at 20℃; | 99% |
-
-
75-97-8
3,3-dimethyl-butan-2-one

| Conditions | Yield |
|---|---|
| With methane In toluene Heating at 80°C with a slight excess of Ti-complex in toluene in a sealed tube (or under a static Ar atmosphere) for 115 h.; (1)H NMR.; | 99% |
| With methane In toluene Heating at 100°C with a slight excess of Ti-complex in toluene in a sealed tube (or under a static Ar atmosphere) for 21 h.; (1)H NMR.; | 71% |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
28049-80-1
bis(diisopropylamino)chloroborane

-
-
959925-25-8
bis(diisopropylamino)boron enolate

| Conditions | Yield |
|---|---|
| With potassium hexamethyldisilazane In tetrahydrofuran byproducts: KCl; under Ar atm. to soln. KHMDS in THF ketone in THF was added dropwise at 0°C for 30 min, soln. chlorobis(dialkylamino)borane in THF was added and stirred at room temp. for 1 h; soln. was centrifugated, ppt. was filtered, volatiles were evapd. in vacuo; | 99% |
| With sodium hexamethyldisilazane In tetrahydrofuran byproducts: NaCl; under Ar atm. to soln. NaHMDS in THF ketone in THF was added dropwise at0°C for 30 min, soln. chlorobis(dialkylamino)borane in THF was a dded and stirred at room temp. for 1 h; soln. was centrifugated, ppt. was filtered, volatiles were evapd. in vacuo; | 92% |
| With diisopropylamine; n-butyllithium In tetrahydrofuran; hexane byproducts: LiCl; under Ar atm. to soln. i-Pr2NH in THF n-BuLi in hexane was added at -20°C and stirred for 30 min, soln. ketone in THF was added, stirred at -20°C for 30 min, chlorobis(dialkylamino)borane in THF was added, stirred at 60°C for 24 h; soln. was centrifugated, ppt. was filtered, volatiles were evapd. in vacuo; | 56% |

| Conditions | Yield |
|---|---|
| With magnesium sulphate trihydrate In dichloromethane for 2h; Reflux; | 99% |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
5469-26-1
1-Bromopinacolon

-
-
3205-30-9
5-hydroxy-2,2,5,6,6-pentamethylheptan-3-one

| Conditions | Yield |
|---|---|
| With samarium diiodide In tetrahydrofuran at -78℃; for 2.41667h; Reformatsky type reaction; Inert atmosphere; | 98% |
| With diethyl ether; magnesium; benzene | |
| (i) Mg, EtBr, Et2O, (ii) /BRN= 1209331/, Et2O, benzene; Multistep reaction; |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
100-51-6
benzyl alcohol

-
-
5195-24-4
4,4-dimethyl-1-phenylpentan-3-one

| Conditions | Yield |
|---|---|
| With cesium hydroxide; (1,4-dimethyl-5,7-diphenyl-1,2,3,4-tetrahydro-6H-cyclopenta[b]pyrazin-6-one)(triphenylphosphine)(dicarbonyl)iron In toluene at 90℃; for 16h; Schlenk technique; Inert atmosphere; | 98% |
| With potassium phosphate; aluminum oxyhydroxide; palladium In toluene at 110℃; for 3h; | 94% |
| With bis(μ-chloro)-bis[1,3-di(2-pyridyl)-4,6-dimethylbenzene-N,C(2'),N-iridium chloride]; caesium carbonate In tert-Amyl alcohol for 12h; Reflux; | 86% |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
37595-74-7
N,N-phenylbistrifluoromethane-sulfonimide

-
-
27701-32-2
3,3-dimethyl-1-butenyl-2-triflate

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane In tetrahydrofuran at -78 - 20℃; Inert atmosphere; | 98% |
| Stage #1: 3,3-dimethyl-butan-2-one In tetrahydrofuran at -78℃; Stage #2: N,N-phenylbistrifluoromethane-sulfonimide In tetrahydrofuran at -78℃; | 86% |
| Stage #1: 3,3-dimethyl-butan-2-one With lithium diisopropyl amide In tetrahydrofuran at -78℃; Stage #2: N,N-phenylbistrifluoromethane-sulfonimide In tetrahydrofuran at -78 - 20℃; | 86% |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
120-80-9
benzene-1,2-diol

-
-
105732-50-1
2-tert-butyl-2-methyl-1,3-benzodioxole

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene for 1h; Microwave irradiation; Reflux; | 98% |
| With toluene-4-sulfonic acid In toluene for 24h; Reflux; | 71% |
-
-
75-97-8
3,3-dimethyl-butan-2-one

-
-
994-30-9
triethylsilyl chloride

-
-
51440-17-6
1-triethylsiloxy-1-tert-butylethene

| Conditions | Yield |
|---|---|
| With n-butyllithium; 1,1,1,3,3,3-hexamethyl-disilazane In tetrahydrofuran; hexane at -78℃; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With sodium In methanol for 12h; Claisen Condensation; Inert atmosphere; Schlenk technique; Glovebox; | 98% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View


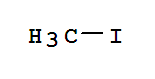



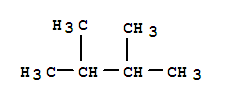









 F,
F,  Xn,
Xn,  Xi
Xi