 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

| Conditions | Yield |
|---|---|
| In water | 98.75% |
| In not given | 80% |

| Conditions | Yield |
|---|---|
| In acetonitrile byproducts: H2; Electrolysis; electrolysis of AgClO4 in CH3CN, Pt anode, 200 mA, 5V;; cathodic deposition of Ag, current yield 70 %;; | A >99 B 97% |
| In acetonitrile byproducts: H2; Electrolysis; electrolysis of AgClO4 in CH3CN, Pt anode, 200 mA, 5V;; cathodic deposition of Ag, current yield 70 %;; | A >99 B 97% |
| In acetonitrile byproducts: H2; Electrolysis; electrolysis of AgClO4 in CH3CN, Pt anode, 30 mA, 4V;; cathodic deposition of Ag, current yield 94 %;; | A >99 B 90% |
| In acetonitrile byproducts: H2; Electrolysis; electrolysis of AgClO4 in CH3CN, Pt anode, 30 mA, 4V;; cathodic deposition of Ag, current yield 94 %;; | A >99 B 90% |

| Conditions | Yield |
|---|---|
| In not given concd. HCl is added to NaClO4; filtering; filtrate is heated to 135°C;; acid is free of Cl(1-);; | 95% |
| In water part of cyclic process;; | |
| In water byproducts: NaCl; addn. of concd. HCl to NaClO4 under formation of NaCl; concg. soln. under removing of HCl excess;; | |
| In not given byproducts: NaCl; aq. HClO4 is obtained;; | |
| In water part of cyclic process;; |

-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In hydrogenchloride addition of 20% HCl-solution to solid waterfree Ba(ClO4)2 forms HClO4;; | 95% |
| With HCl In hydrogenchloride addition of 20% HCl-solution to solid waterfree Ba(ClO4)2 forms HClO4;; | 95% |
| With hydrogenchloride In hydrogenchloride addition of concd. HCl-solution to solid waterfree Ba(ClO4)2 forms HClO4;; | 90% |
| With HCl In hydrogenchloride addition of concd. HCl-solution to solid waterfree Ba(ClO4)2 forms HClO4;; | 90% |

| Conditions | Yield |
|---|---|
| In water byproducts: K2SiF6; 30% aq. H2SiF6 is added to a boiling aq. soln. of KClO4; mixt. is boiled for 1 h;; K2SiF6 and KClO4 is filtered off; H2SiF6 and H2SO4 is removed by BaCl2; evapn.; diln.; purity of acid depends on purity of H2SiF6; chem. pure acid is obtained by distn. under reduced pressure;; | 80% |
| In water excess of H2SiF6 is removed by BaCl2 or Ba(OH)2; Fe is removed by K4Fe(CN)6; evapn. to remove other impurities; acid contains alkali and K;; |
-
-
106379-46-8
1-benzyl-5,7-dimethyl-4,6-dioxo-4,5,6,7-tetrahydropyrazolo<3,4-d>pyrimidine

-
A
-
7601-90-3
perchloric acid

-
B
-
4680-51-7
5,7-dimethylpyrazolo<3,4-d>pyrimidine-4,6(5H,7H)-dione

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; ethanol Hydrogenation; |

-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
-
7732-18-5
water

-
-
10028-15-6
ozone

-
A
-
7647-01-0
hydrogenchloride

-
B
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In water reaction of ClO2 and O3; addn. of water;; |

-
-
7732-18-5
water

-
-
7782-50-5
chlorine

-
-
10028-15-6
ozone

-
A
-
7601-90-3
perchloric acid

-
B
-
16887-00-6
chloride

| Conditions | Yield |
|---|---|
| With moisture In water reaction of O3 and Cl2 in presence of moisture; addn. of water;; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) Irradiation (UV/VIS); at 1800 - 4000 Å; condensation in liquid O2;; | |
| In neat (no solvent) Irradiation (UV/VIS); at 1800 - 4000 Å; condensation in liquid O2;; |


| Conditions | Yield |
|---|---|
| In water ozonized air is passed through an aq. soln. of HClO;; |

| Conditions | Yield |
|---|---|
| In not given distg. at a pressure below 100 Torr; steam is added to prevent formation of solid HClO4*H2O;; obtained HClO4 is 88 to 98% in H2O;; | |
| In not given distg. at a pressure below 100 Torr; steam is added to prevent formation of solid HClO4*H2O;; acid contains 20 to 25% H2SO4;; |


| Conditions | Yield |
|---|---|
| In water Electrolysis; vacuum distillation under concg. of 2 n HClO4 soln.;; | |
| In water Electrolysis; vacuum distillation under concg. of 2 n HClO4 soln.;; |


| Conditions | Yield |
|---|---|
| In water Electrolysis; anodic oxidation;; | |
| In water Electrolysis; anodic oxidation;; |

| Conditions | Yield |
|---|---|
| In water Electrolysis; O2 and H2 are evolved on electrodes; after some time a vol. ratio O2:H2=1:2 has established; oxygen reacts with free chlorine to form perchlorate;; | |
| In water Electrolysis; | |
| In water Electrolysis; O2 and H2 are evolved on electrodes; after some time a vol. ratio O2:H2=1:2 has established; oxygen reacts with free chlorine to form perchlorate;; |

| Conditions | Yield |
|---|---|
| In gas common condensing of educts;; |

-
-
13444-99-0, 501680-61-1
perchloric acid monohydrate

-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In not given repeated freezing out;; | |
| In neat (no solvent) decompn. at 110°C into anhyd. HClO4 and a less volatile aq. acid;; | |
| In neat (no solvent) decompn. at 110°C into anhyd. HClO4 and a less volatile aq. acid;; |
-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
A
-
7647-01-0
hydrogenchloride

-
B
-
7601-90-3
perchloric acid

-
C
-
7790-93-4
chloric acid

| Conditions | Yield |
|---|---|
| In water Irradiation (UV/VIS); decompn. in sunlight;; | |
| In water Irradiation (UV/VIS); reaction in sunlight;; | |
| In water Irradiation (UV/VIS); reaction in sunlight;; |

-
-
10049-04-4, 25052-55-5
chlorine dioxide

-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In not given Irradiation (UV/VIS); at 20°C;; | |
| In not given Irradiation (UV/VIS); | |
| With PbO2 hydrate In not given |

-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In water byproducts: Fe(OH)3, H2O; thermal decompn.;; |

| Conditions | Yield |
|---|---|
| Pt apparatus, 6°C, equilibrium reaction; | |
| Pt apparatus, 6°C, equilibrium reaction; |


| Conditions | Yield |
|---|---|
| In water heating;; | 0% |


| Conditions | Yield |
|---|---|
| In hydrogen fluoride byproducts: H2O; aq. HF; |


| Conditions | Yield |
|---|---|
| In not given aq. acid is obtaind; HCl is removed by heating to 135°C;; | |
| In water aluminium shot is added; heating to 110°C for 10h; Cl(1-) is removed by Ag2O; distg. with H2SO4; anhyd. acid is obtained;; |

-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In water byproducts: Ca(OH)2, H2O; thermal decompn.;; |
-
-
16887-00-6
chloride

-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In not given Electrolysis; in accumulator;; |

| Conditions | Yield |
|---|---|
| With water In not given Electrolysis; | |
| With H2O In not given Electrolysis; |

| Conditions | Yield |
|---|---|
| In dichloromethane; water aq. HClO4 was added to mixt. of corazol and Fe-complex in CH2Cl2, 12 min, Et2O was added; filtered, washed with ether, dried; elem. anal.; | 100% |


| Conditions | Yield |
|---|---|
| With C57H42CoFe3N4 at 20℃; Catalytic behavior; Reagent/catalyst; Electrochemical reaction; | 100% |
| With catalyst: Ba/carbon In perchloric acid aq. HClO4; Electrochem. Process; electroreduced on Ba/carbon in 0.1 M HClO4 (1 bar, 21+/-0.5°C, 1.2 V vs. RHE); | |
| With catalyst: Ce/carbon In perchloric acid aq. HClO4; Electrochem. Process; electroreduced on Ce/carbon in 0.1 M HClO4 (1 bar, 21+/-0.5°C, 1.2 V vs. RHE); |
-
-
1129-30-2
2,6-Diacetylpyridine

-
-
7601-90-3
perchloric acid

-
-
3375-31-3
palladium diacetate

-
-
1372169-85-1
[Pd(O1,N1,C1-pyridine-2-acetyl-6-(C(O)CH2))(acetonitrile)]ClO4

| Conditions | Yield |
|---|---|
| Stage #1: 2,6-Diacetylpyridine; perchloric acid In diethyl ether; water for 2h; Inert atmosphere; Stage #2: palladium diacetate In diethyl ether; water; acetonitrile for 2h; Inert atmosphere; | 100% |

-
-
7601-90-3
perchloric acid

-
-
181575-42-8
N,N'-dimethyl-N,N'-ethylenebis(2-aminomethyl-4-bromo-6-formylphenolato)copper(II)

-
-
107-15-3
ethylenediamine

| Conditions | Yield |
|---|---|
| In water; N,N-dimethyl-formamide to soln. ethylenediamine in DMF Cu complex was added and stirred at roomtemp. for 2 h, suspn. was acidified with aq. HClO4; ppt. was collected, washed with Et2O and dried in vacuo; elem. anal.; | 99% |

-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In acetonitrile addn. of one drop concd. HClO4 to soln. of Cr-complex; pptn. on ether addn., washing (ether), drying in air; elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| In water crystn. on cooling, collection, washing (EtOH, ether), drying (air); | 99% |

-
-
7601-90-3
perchloric acid

-
-
449146-32-1
[CH2(CH2NHCH2CH2NHCH2)2NC3N3(NH2)2Cu(OClO3)2]*H2O

-
-
449728-76-1
Cu(CH2(CH2NHCH2CH2NHCH2)2NC3N3H(NH2)2)(ClO4)2(1+)*ClO4(1-)*H2O=[CH2(CH2NHCH2CH2NHCH2)2NC3N3H(NH2)2Cu(OClO3)2]ClO4*H2O

| Conditions | Yield |
|---|---|
| In perchloric acid aq. HClO4; 0.1 M HClO4; recrystn., elem. anal.; | 99% |
-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In methanol slow addn. of excess 1 M HClO4 to Mo-complex; crystn.; elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| In dichloromethane; water aq. HClO4 was added to mixt. of corazol and Fe-complex in CH2Cl2, 15 min, Et2O was added; filtered, washed with ether, dried; elem. anal.; | 99% |


| Conditions | Yield |
|---|---|
| In dichloromethane; water aq. HClO4 was added to mixt. of corazol and Fe-complex in CH2Cl2, 20 min, Et2O was added; filtered, washed with ether, dried; elem. anal.; | 99% |

-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| With air In not given Mn complex oxidized by air in presence of HClO4; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: copper (II) carbonate hydroxide; perchloric acid In isopropyl alcohol for 0.166667h; Heating; Stage #2: 1,10-Phenanthroline In isopropyl alcohol at 20℃; | 99% |

-
-
7601-90-3
perchloric acid

-
-
28755-93-3
chloro(2,3,7,8,12,13,17,18-octaethylporphyrinato-N,N',N'',N''')iron(III) complex

| Conditions | Yield |
|---|---|
| In methanol treating with 5% HClO4 for 2 d, stay for 2 d to crystn.; | 98% |
-
-
7601-90-3
perchloric acid

-
-
82595-77-5
di-μ-chloro-bis[chloro(η5-pentamethylcyclopentadienyl)cobalt]

| Conditions | Yield |
|---|---|
| evapn. at 0 °C in high vac.; | 98% |
-
-
7601-90-3
perchloric acid

-
-
223139-66-0
ferrocenyl(2,4,6-trimethoxyphenyl)methanol

-
-
223139-82-0
Ferrocenyl(2,4,6-trimethoxyphenyl)carbenium perchlorate

| Conditions | Yield |
|---|---|
| In perchloric acid; ethanol aq. HClO4; stirring Fe-complex suspn. (in EtOH) with 1.1 equiv. of 60% aq. HClO4 (0°C, 15 min); crystn. (-30°C); | 98% |
| In perchloric acid; diethyl ether aq. HClO4; stirring Fe-complex with slight excess of 60% HClO4 (0°C, 15 min); elem. anal.; | 94% |
| In methanol; perchloric acid aq. HClO4; stirring Fe-complex suspn. (in MeOH) with 1.1 equiv. of 60% aq. HClO4 (0°C, 15 min); crystn. (-30°C); | 86% |
-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In water pH=3; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: perchloric acid; C48H54N3O6P3Pd3 In tetrahydrofuran; water at 20℃; for 12h; Inert atmosphere; Stage #2: acetonitrile Inert atmosphere; | 98% |


| Conditions | Yield |
|---|---|
| Stage #1: copper (II) carbonate hydroxide; perchloric acid In isopropyl alcohol for 0.166667h; Heating; Stage #2: 1,10-Phenanthroline In isopropyl alcohol at 20℃; for 3h; | 98% |

-
-
7601-90-3
perchloric acid

-
-
133957-42-3
bis[3-(dimethylamino)propyl]tin dichloride

| Conditions | Yield |
|---|---|
| In water for 5h; | 98% |
-
-
7601-90-3
perchloric acid

| Conditions | Yield |
|---|---|
| In water for 1h; | 98% |

| Conditions | Yield |
|---|---|
| In dichloromethane Re-compd. and HClO4 suspended in MeOH at 0°C, addn. of CH2Cl2 until reaction had begun, addn. of diethyl ether, stirred for 5 min at 0°C; filtered (vac., N2), washed (diethyl ether); | 97% |

-
-
7601-90-3
perchloric acid

-
-
223139-72-8
ferrocenyl(2,6-dimethoxyphenyl)methanol

-
-
223139-96-6
Ferrocenyl(2,6-dimethoxyphenyl)carbenium perchlorate

| Conditions | Yield |
|---|---|
| In perchloric acid; diethyl ether aq. HClO4; stirring Fe-complex with slight excess of 60% HClO4 (0°C, 15 min); elem. anal.; | 97% |
-
-
7601-90-3
perchloric acid

-
-
127099-85-8, 780722-26-1
dicyandiamide

-
-
20667-12-3
silver(l) oxide

-
-
573987-19-6
[Ag(cyanogunidine)2]ClO4

| Conditions | Yield |
|---|---|
| In water Ag2O was dissolved in aq. HClO4 in a polypropylene beaker, 4 equiv. of N-compd. was added in H2O; standing for 20 min at room temp., crystals were filtered, washed with small vol. of ice-cold H2O, dried in a desiccator; filtrate waskept at 0 °C for 24 h, further amt. of compd. could be isolated, elem. anal.; | 97% |

-
-
7601-90-3
perchloric acid

-
-
3457-55-4
1-phenyl-1,2-butanedione

-
-
10444-89-0
2-amino-5-trifluoromethyl-1,3,4-thiadiazole

| Conditions | Yield |
|---|---|
| In ethanol in boiling ethanol ( 0.2 h ), with 20 % 1-phenylbutane-1,2-dione excess; | A 4% B 96% |
| In ethanol in boiling ethanol ( 0.2 h ), with 20 % 1-phenylbutane-1,2-dione excess; | A 4% B 96% |


| Conditions | Yield |
|---|---|
| In formic acid aq. formic acid; refluxing soln. of Cu-complex and HCHO (48 h), evapn. (reduced pressure), dissolving in hot H2O, pptn. on HClO4 addn.; filtration, washing (EtOH, Et2O), drying (vac. desiccator); elem. anal.; | 96% |

-
-
7601-90-3
perchloric acid

-
-
719-54-0
10-methyl-9, 10-dihydro-9-acridinone

-
-
576-83-0
2,4,6-trimethylphenyl bromide

| Conditions | Yield |
|---|---|
| Stage #1: 2,4,6-trimethylphenyl bromide With tert.-butyl lithium In diethyl ether; pentane at -78℃; for 0.5h; Cooling with acetone-dry ice; Stage #2: 10-methyl-9, 10-dihydro-9-acridinone In diethyl ether; pentane at -78 - 25℃; for 72h; Cooling with acetone-dry ice; Stage #3: perchloric acid In ethyl acetate | 96% |


| Conditions | Yield |
|---|---|
| Stage #1: perchloric acid; C13H11N2O2S(1-)*Cu(2+)*NO3(1-) In methanol; water for 4h; Stage #2: acetonitrile In methanol; dichloromethane | 96% |

Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View





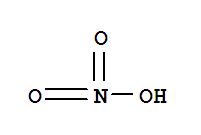

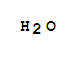







 C,
C, O,
O, Xi
Xi