-
Name
3,4,6-Trichloropyridazine
- EINECS 629-902-8
- CAS No. 6082-66-2
- Article Data31
- CAS DataBase
- Density 1.642 g/cm3
- Solubility
- Melting Point 55.0 to 59.0 °C
- Formula C4HCl3N2
- Boiling Point 315.521 °C at 760 mmHg
- Molecular Weight 183.424
- Flash Point 173.901 °C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms T6NNJ CG DG FG;NSC 19451;NSC 51080;
- PSA 25.78000
- LogP 2.43680
Synthetic route
-
-
15456-86-7
4-bromo-1,2-dihydropyridazine-3,6-dione

-
-
6082-66-2
3,4,6-trichloropyridazine

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 110℃; for 5h; | 92% |
| With trichlorophosphate at 100℃; for 3h; | 86% |
| With trichlorophosphate for 4h; Heating; | 84% |

| Conditions | Yield |
|---|---|
| Stage #1: bromomaleic anhydride With hydrazine In water for 4h; Reflux; Stage #2: With trichlorophosphate for 3.5h; Reflux; | 87% |
-
-
15456-86-7
4-Brom-pyridazin-3,6-diol

-
-
6082-66-2
3,4,6-trichloropyridazine

| Conditions | Yield |
|---|---|
| With trichlorophosphate Reflux; | 82% |

| Conditions | Yield |
|---|---|
| With chlorine; aluminum (III) chloride at 120℃; for 3h; Neat (no solvent); | 55% |
| With aluminum (III) chloride; chlorine at 140℃; for 4h; | |
| With chlorine; aluminium trichloride | |
| With aluminum (III) chloride; chlorine at 140℃; for 4h; | 44.1 g |

| Conditions | Yield |
|---|---|
| With trichlorophosphate | |
| With trichlorophosphate; phosphorus trichloride | |
| With trichlorophosphate for 10h; Heating / reflux; | |
| With trichlorophosphate |
-
-
15456-86-7
4-bromo-1,2-dihydropyridazine-3,6-dione

-
A
-
6082-66-2
3,4,6-trichloropyridazine

-
B
-
10344-42-0
4-bromo-3,6-dichloro-pyridazine

| Conditions | Yield |
|---|---|
| With trichlorophosphate for 3.5h; Heating / reflux; |
-
-
15456-86-7
4-bromo-1,2-dihydropyridazine-3,6-dione

-
A
-
6082-66-2
3,4,6-trichloropyridazine

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1,2-dihydropyridazine-3,6-dione With trichlorophosphate for 3.5h; Heating / reflux; Stage #2: With sodium hydrogencarbonate In water |
-
-
15456-86-7
4-bromo-1,2-dihydropyridazine-3,6-dione

-
A
-
6082-66-2
3,4,6-trichloropyridazine

-
B
-
1003944-29-3
C4HBrCl2N2

-
C
-
10344-42-0
4-bromo-3,6-dichloro-pyridazine

| Conditions | Yield |
|---|---|
| With trichlorophosphate for 3.5h; Heating / reflux; |

-
-
30480-35-4
pyridazine-3,4,6-triol

-
-
6082-66-2
3,4,6-trichloropyridazine

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 100℃; |

| Conditions | Yield |
|---|---|
| Stage #1: Cyclopropylmethanol With sodium hydride In tetrahydrofuran at 0℃; for 0.166667h; Stage #2: 3,4,6-trichloropyridazine In tetrahydrofuran at 0 - 20℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With ammonia In 1,4-dioxane Reflux; | 97% |
| With ammonium hydroxide In 1,4-dioxane at 90℃; for 12h; regioselective reaction; | 86% |
| With ammonium hydroxide In water at 75℃; for 16h; | 76% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 50℃; for 0.5h; | 97% |
| In isopropyl alcohol at 120℃; for 0.666667h; Microwave irradiation; Sealed tube; regioselective reaction; | 87% |
| In tetrahydrofuran at 50℃; for 2h; Sealed tube; | 62% |
| With potassium carbonate In acetonitrile at 95℃; for 4h; Inert atmosphere; |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
57260-71-6
1-t-Butoxycarbonylpiperazine

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 0 - 20℃; | 94% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
124-41-4
sodium methylate

-
-
70952-62-4
3,6-dichloro-4-methoxy-1,2-dihydropyridazine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 1h; | 94% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
74-89-5
methylamine

-
-
17645-06-6
3,6-dichloro-N-methyl-pyridazin-4-amine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 50℃; for 2h; Sealed tube; | 93.7% |
| In 1,4-dioxane; water at 20℃; for 0.166667h; regioselective reaction; | 88% |
| In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | |
| In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
124-40-3
dimethyl amine

-
-
17258-35-4
3,6-dichloro-N,N-dimethylpyridazin-4-amine

| Conditions | Yield |
|---|---|
| In 1,4-dioxane; water at 0℃; for 0.166667h; regioselective reaction; | 93% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 50℃; for 3h; Sealed tube; | 91.1% |
| In tetrahydrofuran at 50℃; for 3h; | 89% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
60-24-2
2-hydroxyethanethiol

-
-
943026-17-3
2-[(3,6-dichloro-4-pyridazinyl)thio]ethanol

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 20℃; for 72h; | 91% |
| With triethylamine In tetrahydrofuran at 0 - 20℃; for 70.5833h; | 91% |
| With triethylamine In tetrahydrofuran at 0 - 20℃; for 72.0833h; | 91% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
144-55-8
sodium hydrogencarbonate

-
-
60-24-2
2-hydroxyethanethiol

-
-
943026-17-3
2-[(3,6-dichloro-4-pyridazinyl)thio]ethanol

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran | 91% |

-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
75-04-7
ethylamine

-
-
29049-49-8
4-ethylamino-3,6-dichloropyridazine

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran; acetonitrile at 0 - 20℃; for 16h; Inert atmosphere; | 91% |
| In water for 2h; |

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 120℃; for 0.666667h; Microwave irradiation; Sealed tube; regioselective reaction; | 90% |

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 120℃; for 0.333333h; Microwave irradiation; Sealed tube; regioselective reaction; | 89% |
-
-
19816-92-3
3,3-dimethyl azetidine

-
-
6082-66-2
3,4,6-trichloropyridazine

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 40℃; for 3h; | 88.15% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
349-88-2
4-Fluorobenzenesulfonyl chloride

-
-
1162681-01-7
N-(3,6-dichloropyridazin-4-yl)-4-fluorobenzenesulfonamide

| Conditions | Yield |
|---|---|
| Stage #1: 3,4,6-trichloropyridazine With sodium hexamethyldisilazane In tetrahydrofuran at 0℃; for 0.5h; Stage #2: 4-Fluorobenzenesulfonyl chloride In tetrahydrofuran at 0℃; for 1h; Stage #3: With water; ammonium chloride In tetrahydrofuran | 86.7% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
897732-75-1
2-methyl-6-(methylsulfonyl)pyridin-3-amine

-
-
1236355-11-5
3,6-dichloro-N-(2-methyl-6-(methylsulfonyl)pyridin-3-yl)pyridazin-4-amine

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 20℃; for 16h; | 84% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
10344-42-0
4-bromo-3,6-dichloro-pyridazine

-
-
17284-80-9
2-[(3,6-dichloropyridazin-4-yl)oxy]ethan-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: ethylene glycol With sodium hydride In tetrahydrofuran at 0℃; for 0.666667h; Stage #2: 3,4,6-trichloropyridazine; 4-bromo-3,6-dichloro-pyridazine In tetrahydrofuran at 0 - 20℃; | 83% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
17284-80-9
2-[(3,6-dichloropyridazin-4-yl)oxy]ethan-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: ethylene glycol With sodium hydride In tetrahydrofuran at 0℃; for 0.666667h; Stage #2: 3,4,6-trichloropyridazine In tetrahydrofuran at 0 - 20℃; | 83% |
| Stage #1: ethylene glycol With sodium hydride In tetrahydrofuran at 0℃; for 0.666667h; Stage #2: 3,4,6-trichloropyridazine In tetrahydrofuran at 0 - 20℃; | |
| Stage #1: ethylene glycol With sodium hydride In tetrahydrofuran at 0℃; for 0.666667h; Stage #2: 3,4,6-trichloropyridazine In tetrahydrofuran at 0 - 20℃; |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
107-21-1
ethylene glycol

-
-
17284-80-9
2-[(3,6-dichloropyridazin-4-yl)oxy]ethan-1-ol

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 0 - 20℃; | 83% |
| Stage #1: ethylene glycol With sodium hydride In tetrahydrofuran at 0℃; for 0.666667h; Stage #2: 3,4,6-trichloropyridazine In tetrahydrofuran at 0 - 20℃; | 83% |
| Stage #1: ethylene glycol With sodium hydride In tetrahydrofuran at 0℃; for 0.666667h; Stage #2: 3,4,6-trichloropyridazine In tetrahydrofuran at 0 - 20℃; | 83% |
-
-
175277-74-4
(3-chloro-5-trifluoromethyl-pyridin-2-yl)methylamine

-
-
6082-66-2
3,4,6-trichloropyridazine

| Conditions | Yield |
|---|---|
| With triethylamine In methanol at 20℃; | 82.2% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 20 - 65℃; | 80% |
| With sodium hydride |

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 130℃; for 12h; Microwave irradiation; Sealed tube; regioselective reaction; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: benzene-1,2-diol With sodium hydride In 1,2-dimethoxyethane at 0℃; for 0.5h; Stage #2: 3,4,6-trichloropyridazine In 1,2-dimethoxyethane for 2h; Reflux; | 80% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
144222-22-0
tert-butyl 4-(aminomethyl)piperidine-1-carboxylate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; | 79% |
| In tetrahydrofuran at 100℃; | 26% |
-
-
6457-49-4
4-piperidinemethanol

-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
1349874-35-6
[1-(3,6-dichloropyridazin-4-yl)piperidin-4-yl]methanol

| Conditions | Yield |
|---|---|
| With triethylamine In dimethyl sulfoxide at 100℃; | 75.99% |
-
-
103-76-4
1-(2-hydroxyethyl)piperazine

-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
921769-61-1
2-(4-(3,6-dichloropyridazin-4-yl)piperazin-1-yl)ethanol

| Conditions | Yield |
|---|---|
| With sodium carbonate In N,N-dimethyl acetamide at 20℃; for 12h; Inert atmosphere; | 73.6% |
| With sodium carbonate In N,N-dimethyl acetamide at 20℃; | 73.6% |
-
-
6082-66-2
3,4,6-trichloropyridazine

-
-
42302-19-2
2-(thiomorpholino)ethanethiol

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 20h; Heating; | 72% |
3,4,6-Trichloropyridazine Specification
The 3,4,6-Trichloropyridazine, with the CAS registry number 6082-66-2, is also known as Pyridazine, 3,4,6-trichloro-. This chemical's molecular formula is C4HCl3N2 and molecular weight is 183.42. What's more, its IUPAC name is the same with product name.
Physical properties about 3,4,6-Trichloropyridazine are: (1)ACD/LogP: 1.359; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.36; (4)ACD/LogD (pH 7.4): 1.36; (5)ACD/BCF (pH 5.5): 6.35; (6)ACD/BCF (pH 7.4): 6.35; (7)ACD/KOC (pH 5.5): 130.74; (8)ACD/KOC (pH 7.4): 130.74; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 25.78 Å2; (13)Index of Refraction: 1.579; (14)Molar Refractivity: 37.12 cm3; (15)Molar Volume: 111.733 cm3; (16)Surface Tension: 54.21 dyne/cm; (17)Density: 1.642 g/cm3; (18)Flash Point: 173.901 °C; (19)Enthalpy of Vaporization: 53.459 kJ/mol; (20)Boiling Point: 315.521 °C at 760 mmHg; (21)Vapour Pressure: 0.0010 mmHg at 25 °C.
Preparation of 3,4,6-Trichloropyridazine: this chemical can be prepared by 4-Bromo-1,2-dihydro-pyridazine-3,6-dione. The reaction occurs with reagent POCl3 and other condition of heating for 4 hours. The yield is 84 %.
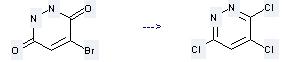
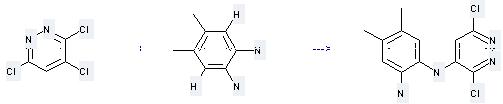
Uses of 3,4,6-Trichloropyridazine: it is used to produce other chemicals. For example, it can react with 4,5-Dimethyl-benzene-1,2-diamine to get N-(3,6-Dichloro-pyridazin-4-yl)-4,5-dimethyl-benzene-1,2-diamine. This reaction needs reagent anhydrous sodium acetate and solvent ethanol. The yield is 55 %.
You can still convert the following datas into molecular structure:
(1) SMILES: Clc1nnc(Cl)cc1Cl
(2) InChI: InChI=1S/C4HCl3N2/c5-2-1-3(6)8-9-4(2)7/h1H
(3) InChIKey: LJDQXQOPXOLCHL-UHFFFAOYSA-N
Related Products
- 3,10-Diaminotricyclo(5.2.1.0(sup 2,6))decane
- 3,10-Dinitrophenanthrene
- 3-((10-ETHYL-11-(p-HYDROXYPHENYL)DIBENZ-(B,F)OXEPIN-3-YL)OXY)-1,2-PROPANEDIOL HYDRATE (4:1)
- 3-(1,1,2,2-Tetrafluoroethoxy)aniline
- 3-(1,1,2,2-Tetrafluoroethoxy)benzaldehyde
- 3-(1,1,2,2-Tetrafluoroethoxy)bromobenzene
- 3-(1,1,2,2-Tetrafluoroethoxy)toluene
- 3-[1,1'-Biphenyl]-4-yl-1,2,3,4-tetrahydro-1-naphthol
- 3,11-Dichloro-6,11-dihydro-6-methyldibenzo[c,f][1,2]thiazepine 5,5-dioxide
- 3-[(1,1-Dimethyl-2-hydroxyethyl)amino]-2-hydroxypropanesulfonicacid
- 60827-45-4
- 608-27-5
- 608-28-6
- 608-30-0
- 608-31-1
- 60831-31-4
- 60832-72-6
- 608-33-3
- 60833-84-3
- 608-34-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View