-
Name
4-Bromomethyl-2-cyanobiphenyl
- EINECS 425-280-5
- CAS No. 114772-54-2
- Article Data65
- CAS DataBase
- Density 1.43 g/cm3
- Solubility Insoluble in water
- Melting Point 125-128 °C(lit.)
- Formula C14H10BrN
- Boiling Point 413.2°Cat760mmHg
- Molecular Weight 272.144
- Flash Point 203.7°C
- Transport Information
- Appearance Cream powder
- Safety 26-36/37/39
- Risk Codes 20/21/22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 2-(4-Bromomethylphenyl)benzonitrile;2-Cyano-4'-(bromomethyl)biphenyl;2'-(Cyano)biphenyl-4-ylmethyl bromide;2'-Cyano-4-(bromomethyl)-1,1'-biphenyl;4-(2-Cyanophenyl)benzyl bromide;4-(Bromomethyl)-2'-cyanobiphenyl;4-Bromomethyl-2'-cyano-1,1'-biphenyl;4'-(Bromomethyl)-2-cyanobiphenyl;4'-(Bromomethyl)[1,1'-biphenyl]-2-carbonitrile;4'-Bromomethylbiphenyl-2-carbonitrile;2-Cyano-4'-Bromomethylbiphenyl;2-Cyano-4'-Bromomethyl Biphenyl;
- PSA 23.79000
- LogP 4.12018
Synthetic route
-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; dibenzoyl peroxide In tetrachloromethane | 96% |
| With bromine In water; 1,2-dichloro-ethane at 84℃; for 0.25h; Temperature; Solvent; Irradiation; Flow reactor; | 96.3% |
| With bromine In 1,2-dichloro-ethane at 70℃; for 0.5h; Reagent/catalyst; Temperature; Time; Irradiation; | 94% |
-
-
64113-85-5
2-cyano-4-methylbiphenyl

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With 5,5-dibromohydantoin In dichloromethane at 50℃; for 0.5h; Temperature; Reagent/catalyst; Irradiation; | 94% |
| With N-Bromosuccinimide; dibenzoyl peroxide In tetrachloromethane at 100℃; for 2h; |
-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
-
107-06-2
1,2-dichloro-ethane

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With bromine In water | 89.4% |
| With N-Bromosuccinimide In water | 79.6% |

-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
A
-
209911-63-7
2-(4,4-dibromomethylphenyl)benzonitrile

-
B
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; Azo-bis(2,4-dimethyl-valeronitril) In dichloromethane at 45 - 50℃; for 2 - 5h; Industry scale; | A n/a B 82% |
| With sodium bromate; hydrogen bromide In dichloromethane; water at 0 - 20℃; UV-irradiation; | A 10% B 80% |
| With sodium bromate; azobis(2-cyanobutane); bromine In water; chlorobenzene at 70 - 80℃; for 6h; | A n/a B 69% |
-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
-
4419-11-8, 108346-39-0, 108346-40-3
2,2'-azobis (2,4-dimethyl valeronitrile)

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In dichloromethane; water | 80.5% |

-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
-
108-90-7
chlorobenzene

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With bromine | 75% |

-
-
3839-22-3
2-cyanobenzoic acid

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 71 percent / copper(II) oxide; potassium carbonate; potassium fluoride / palladium(II) bromide; 1,10-phenanthroline / quinoline / 24 h / 170 °C 2: 45 percent / N-bromosuccinimide; azobis(isobutyronitrile) / CCl4; cyclohexane / 18 h / 75 °C View Scheme |
-
-
131-63-5
ammonium 2-(aminocarbonyl)benzoate

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 72 percent / acetic anhydride; pyridine 2: 71 percent / copper(II) oxide; potassium carbonate; potassium fluoride / palladium(II) bromide; 1,10-phenanthroline / quinoline / 24 h / 170 °C 3: 45 percent / N-bromosuccinimide; azobis(isobutyronitrile) / CCl4; cyclohexane / 18 h / 75 °C View Scheme |
-
-
57598-33-1
2-(2-methoxyphenyl)-4,4-dimethyl-2-oxazoline

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: tetrahydrofuran / 2 h / 20 °C 2: 96 percent / pyridine, phosphorus oxychloride / 3 h / 100 °C 3: N-bromosuccinimide, dibenzoyl peroxide / CCl4 / 3 h / Heating View Scheme | |
| Multi-step reaction with 4 steps 1: magnesium / tetrahydrofuran 2: hydrogenchloride 3: thionyl chloride / water; toluene 4: dibenzoyl peroxide View Scheme |
-
-
74201-13-1
N-(2-hydroxy-1,1-dimethyl)-2-methoxybenzamide

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: thionyl chloride / 1 h / 25 °C 2: tetrahydrofuran / 2 h / 20 °C 3: 96 percent / pyridine, phosphorus oxychloride / 3 h / 100 °C 4: N-bromosuccinimide, dibenzoyl peroxide / CCl4 / 3 h / Heating View Scheme |
-
-
84392-32-5
4,4-dimethyl-2-(4'-methyl-biphenyl-2-yl)-4,5-dihydrooxazole

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 96 percent / pyridine, phosphorus oxychloride / 3 h / 100 °C 2: N-bromosuccinimide, dibenzoyl peroxide / CCl4 / 3 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: hydrogenchloride 2: thionyl chloride / water; toluene 3: dibenzoyl peroxide View Scheme |
-
-
579-75-9
2-Methoxybenzoic acid

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 95 percent / thionyl chloride / 18 h / 20 °C 2: CH2Cl2 / 2 h / 20 °C 3: thionyl chloride / 1 h / 25 °C 4: tetrahydrofuran / 2 h / 20 °C 5: 96 percent / pyridine, phosphorus oxychloride / 3 h / 100 °C 6: N-bromosuccinimide, dibenzoyl peroxide / CCl4 / 3 h / Heating View Scheme |
-
-
21615-34-9
2-Methoxybenzoyl chloride

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: CH2Cl2 / 2 h / 20 °C 2: thionyl chloride / 1 h / 25 °C 3: tetrahydrofuran / 2 h / 20 °C 4: 96 percent / pyridine, phosphorus oxychloride / 3 h / 100 °C 5: N-bromosuccinimide, dibenzoyl peroxide / CCl4 / 3 h / Heating View Scheme |
-
-
108-90-7
chlorobenzene

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| In water |

-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| In water; chlorobenzene | |
| In water; chlorobenzene |


-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile
-
-
56-23-5
tetrachloromethane

-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
-
94-36-0
dibenzoyl peroxide

-
B
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
34241-39-9
azobisisobutyronitrile

-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In water; chlorobenzene | |
| With N-Bromosuccinimide In water; chlorobenzene | |
| With N-Bromosuccinimide In tetrachloromethane | |
| With N-Bromosuccinimide In tetrachloromethane | |
| With N-Bromosuccinimide In tetrachloromethane |
-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
-
94-36-0
dibenzoyl peroxide

-
B
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In tetrachloromethane |

-
-
114772-53-1
2-Cyano-4'-methylbiphenyl

-
-
94-36-0
dibenzoyl peroxide

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In tetrachloromethane | |
| With N-Bromosuccinimide In tetrachloromethane |
-
-
2042-37-7
o-cyanobromobenzene

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrakis(triphenylphosphine) palladium(0); sodium carbonate / ethanol; water; toluene / 80 °C / Inert atmosphere 2: N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) / tetrachloromethane / 40 h / 80 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: bis-triphenylphosphine-palladium(II) chloride; lithium chloride / N,N-dimethyl-formamide / Inert atmosphere 2: N-Bromosuccinimide; dibenzoyl peroxide / tetrachloromethane View Scheme | |
| Multi-step reaction with 2 steps 1.1: palladium dichloride / methanol; toluene / 0.17 h / 20 °C / Inert atmosphere 1.2: 12 h / 110 °C / Inert atmosphere 2.1: 2,2'-azobis(isobutyronitrile); N-Bromosuccinimide / chlorobenzene / 6 h / 70 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: bis-triphenylphosphine-palladium(II) chloride; lithium chloride / toluene / 6 h / 110 °C / Inert atmosphere 2: sodium bromate; sodium metabisulfite / ethyl acetate; water / 5 h / 0 - 25 °C View Scheme |
-
-
5720-05-8
4-methylphenylboronic acid

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrakis(triphenylphosphine) palladium(0); sodium carbonate / ethanol; water; toluene / 80 °C / Inert atmosphere 2: N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) / tetrachloromethane / 40 h / 80 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1.1: palladium dichloride / methanol; toluene / 0.17 h / 20 °C / Inert atmosphere 1.2: 12 h / 110 °C / Inert atmosphere 2.1: 2,2'-azobis(isobutyronitrile); N-Bromosuccinimide / chlorobenzene / 6 h / 70 °C / Inert atmosphere View Scheme |
-
-
1075-70-3
benzaldehyde N,N-dimethylhydrazone

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 4-(dimethylamino)picolinaldehyde N,N-dibenzylhydrazone; (1,5-cyclooctadiene)(methoxy)iridium(I) dimer / tetrahydrofuran / 24 h / 80 °C / Inert atmosphere 2: palladium bis[bis(diphenylphosphino)ferrocene] dichloride; potassium phosphate / N,N-dimethyl-formamide / 80 °C / Inert atmosphere 3: magnesium bis(monoperoxyphthalate)hexahydrate / methanol / 0 h / 20 °C 4: sodium metabisulfite; sodium bromate / water; ethyl acetate / 20 °C View Scheme | |
| Multi-step reaction with 4 steps 1: 4-(dimethylamino)picolinaldehyde N,N-dibenzylhydrazone; (1,5-cyclooctadiene)(methoxy)iridium(I) dimer / tetrahydrofuran / 24 h / 80 °C / Inert atmosphere 2: palladium bis[bis(diphenylphosphino)ferrocene] dichloride; potassium phosphate / N,N-dimethyl-formamide / 80 °C / Inert atmosphere 3: magnesium bis(monoperoxyphthalate)hexahydrate / methanol / 0 h / 20 °C 4: sodium metabisulfite; sodium bromate / water; ethyl acetate / 20 °C View Scheme |
-
-
1360607-13-1
2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzaldehyde N,N-dimethylhydrazone

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: palladium bis[bis(diphenylphosphino)ferrocene] dichloride; potassium phosphate / N,N-dimethyl-formamide / 80 °C / Inert atmosphere 2: magnesium bis(monoperoxyphthalate)hexahydrate / methanol / 0 h / 20 °C 3: sodium metabisulfite; sodium bromate / water; ethyl acetate / 20 °C View Scheme |
-
-
1404116-53-5
4′-methyl-[1,1′-biphenyl]-2-carbaldehyde N,N-dimethylhydrazone

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: magnesium bis(monoperoxyphthalate)hexahydrate / methanol / 0 h / 20 °C 2: sodium metabisulfite; sodium bromate / water; ethyl acetate / 20 °C View Scheme |
-
-
31614-66-1
tributyl(p-tolyl)stannane

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: bis-triphenylphosphine-palladium(II) chloride; lithium chloride / N,N-dimethyl-formamide / Inert atmosphere 2: N-Bromosuccinimide; dibenzoyl peroxide / tetrachloromethane View Scheme |
-
-
873-32-5
2-Chlorobenzonitrile

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: manganese(ll) chloride; chloro-trimethyl-silane / tetrahydrofuran / -5 - 0 °C / Inert atmosphere; Green chemistry 1.2: 9 h / -5 - 25 °C / Inert atmosphere; Green chemistry 2.1: N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) / tetrachloromethane / 3 h / 80 °C / Green chemistry View Scheme |
-
-
696-61-7
4-tolylmagnesium chloride

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: manganese(ll) chloride; chloro-trimethyl-silane / tetrahydrofuran / -5 - 0 °C / Inert atmosphere; Green chemistry 1.2: 9 h / -5 - 25 °C / Inert atmosphere; Green chemistry 2.1: N-Bromosuccinimide; 2,2'-azobis(isobutyronitrile) / tetrachloromethane / 3 h / 80 °C / Green chemistry View Scheme |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
127007-35-6
Methyl 3-nitro-4-valerylaminobenzoate

-
-
136285-45-5
4-[(2'-Cyano-biphenyl-4-ylmethyl)-pentanoyl-amino]-3-nitro-benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 15h; Ambient temperature; | 100% |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
3249-68-1
ethyl butyroyl acetate

-
-
137860-26-5
ethyl 2-<(2'-cyanobiphenyl-4-yl)methyl>-3-oxohexanoate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; lithium chloride In tetrahydrofuran for 15h; Heating; | 98% |
| With N-ethyl-N,N-diisopropylamine; lithium chloride In tetrahydrofuran for 15h; Heating; | |
| With N-ethyl-N,N-diisopropylamine; lithium chloride In tetrahydrofuran for 15h; Heating; Yield given; |
-
-
16652-76-9
L-valine benzyl ester p-toluenesulfonate salt

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
137864-24-5
(S)-2-[(2'-cyanobiphenyl-4-ylmethyl)amino]-3-methylbutyric acid benzyl ester hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: L-valine benzyl ester p-toluenesulfonate salt With sodium hydroxide In 5,5-dimethyl-1,3-cyclohexadiene; water at 20℃; pH=< 8.5 - 9; Large scale; Stage #2: 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile In 5,5-dimethyl-1,3-cyclohexadiene at 60℃; for 3.41667h; Large scale; Stage #3: With hydrogenchloride In 5,5-dimethyl-1,3-cyclohexadiene; water at 72℃; for 2h; Dean-Stark; Large scale; | 98% |
| Stage #1: L-valine benzyl ester p-toluenesulfonate salt; 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile With tetrabutylammomium bromide; potassium carbonate; potassium iodide In water; toluene at 50 - 55℃; for 25h; Stage #2: With hydrogenchloride In water; toluene pH=1 - 2; Product distribution / selectivity; | 90% |
| Stage #1: L-valine benzyl ester p-toluenesulfonate salt; 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile With tetrabutylammomium bromide; potassium carbonate; potassium iodide In water; toluene Stage #2: With hydrogenchloride In water; toluene pH=1 - 2; Product distribution / selectivity; | 90% |
-
-
10264-18-3
N-phenylpentanamide

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: N-phenylpentanamide With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.166667h; Inert atmosphere; Stage #2: 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile In N,N-dimethyl-formamide at 20℃; for 2h; Inert atmosphere; | 98% |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
143096-18-8
N-(2'-cyanobiphenyl-4-ylmethyl)-N-propyl-amine

| Conditions | Yield |
|---|---|
| 96% |
-
-
1206680-19-4
methyl 4-methyl-2-propyl-1H-benzo[d]imidazole-6-carboxylate hydrochloride

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
1098100-86-7
2-cyano-4'-[2"-n-propyl-4"-methyl-6"-methylcarboxylate-benzimidazole-1"-ylmethyl]biphenyl

| Conditions | Yield |
|---|---|
| Stage #1: methyl 4-methyl-2-propyl-1H-benzo[d]imidazole-6-carboxylate hydrochloride With sodium carbonate; sodium iodide In acetone Stage #2: 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile In acetone for 15h; Product distribution / selectivity; Reflux; | 95% |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
149-30-4
2-Mercaptobenzothiazole

-
-
721970-96-3
4'-(benzothiazol-2-ylsulfanylmethyl)-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In acetonitrile at 20℃; for 1.33333h; | 94.7% |
-
-
1429045-74-8
2-(4-fluoro-benzylsulfanyl)-3H-quinazolin-4-thione

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
1429045-99-7
C29H20FN3S2

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 12h; Inert atmosphere; | 94% |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
99-76-3
methyl 4-hydroxylbenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In butanone at 78 - 80℃; for 2h; | 93.5% |
-
-
1313233-70-3
2-butyl-4-(4-fluorophenyl)-1H-imidazole-5-carbaldehyde

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
1313233-71-4
4'-[{2-butyl-4-(4-fluorophenyl)-5-formyl-1H-imidazol-1-yl}methyl]biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 4h; | 93.2% |
-
-
7737-62-4
Ethyl 3-oxoheptanoate

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
137860-02-7
2-(2'-Cyano-biphenyl-4-ylmethyl)-3-oxo-heptanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; lithium chloride In tetrahydrofuran for 15h; Heating; | 93% |
| With N-ethyl-N,N-diisopropylamine; lithium chloride In tetrahydrofuran for 15h; Heating; | |
| With N-ethyl-N,N-diisopropylamine; lithium chloride In tetrahydrofuran for 15h; Heating; Yield given; |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
147876-57-1
(6-Butyl-2-oxo-1,2-dihydro-pyridin-3-ylmethyl)-carbamic acid tert-butyl ester

-
-
147876-52-6
6-butyl-1-(2'-cyanobiphenyl-4-ylmethyl)-1,2-dihydro-2-oxo-3-tert-butoxycarbonylaminomethyl pyridine

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane for 72h; Heating; | 92% |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
124-40-3
dimethyl amine

-
-
1000930-96-0
4'-((dimethylamino)methyl)-[1,1'-biphenyl]-2-carbonitrile

| Conditions | Yield |
|---|---|
| In diethyl ether; water at 20℃; | 92% |
-
-
666816-98-4
8-bromo-7-(but-2-yn-1-yl)-3-methyl-2,,6-dihydro-1H-purine-2,6-dione

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 55℃; for 6h; | 92% |
-
-
51-17-2
benzoimidazole

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
136285-25-1
4′-((1H-benzo[d]imidazol-1-yl)methyl)-[1,1′-biphenyl]-2-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: benzoimidazole With potassium hydroxide In dimethyl sulfoxide at 100℃; for 2h; Stage #2: 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile In dimethyl sulfoxide for 12h; | 92% |
-
-
34036-16-3
5-methyl-3-oxo-hexanoic acid ethyl ester

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
137860-48-1
2-(2'-Cyano-biphenyl-4-ylmethyl)-5-methyl-3-oxo-hexanoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; lithium chloride In tetrahydrofuran for 15h; Heating; | 91% |
-
-
5465-29-2
2-propyl-1H-benzimidazole

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
136285-23-9
4’-((2-propyl-1H-benzo[d]imidazol-1-yl)methyl)-[1,1'-biphenyl]-2-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 2-propyl-1H-benzimidazole With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.5h; Stage #2: 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile In N,N-dimethyl-formamide at 0 - 20℃; | 91% |
| With sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 3h; Cooling with ice; | 70% |
| Stage #1: 2-propyl-1H-benzimidazole With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.5h; Inert atmosphere; Stage #2: 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile In N,N-dimethyl-formamide at 20℃; Inert atmosphere; |
-
-
3337-86-8
quinazoline-4(3H)-thione

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
1429045-94-2
C22H15N3S

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 12h; Inert atmosphere; | 91% |

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| In nitromethane for 7.5h; Heating; | 91% |
-
-
1186293-68-4
7-amino-4-(azidomethyl)-2H-chromen-2-one

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With sodium carbonate In acetone for 6h; Reflux; | 90.5% |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
138401-24-8
4'-[(2-butyl-4-oxo-1,3-diazaspiro[4.4]non-1-en-3-yl)methyl]-[1,1'-biphenyl]-2-carbonitrile

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 0.0208333h; microwave irradiation; | 90% |
| Stage #1: 2-butyl-1,3-diazaspiro[4.4]non-1-en-4-one hydrochloride With potassium carbonate; tetrabutylammomium bromide In water at 25 - 30℃; for 2h; Inert atmosphere; Stage #2: 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile In water at 55 - 60℃; Product distribution / selectivity; | 84.2% |
| With tetrabutylammomium bromide; sodium hydroxide In Isopropyl acetate at 0 - 30℃; for 12h; Solvent; | 39% |
-
-
123-30-8
4-amino-phenol

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide Reflux; | 90% |
-
-
455-14-1
4-trifluoromethylphenylamine

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide Reflux; | 90% |

-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 5-chloro-1,7’-dimethyl-2’-propyl-1H,3’H-2,5’-bibenzo[d]imidazole With sodium hydroxide In water; acetone for 0.5h; Stage #2: 4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile In water; acetone at 25℃; | 90% |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium methylate In tetrahydrofuran; methanol at 20 - 30℃; Solvent; Reagent/catalyst; Temperature; | 89.9% |
| With hydroxylamine hydrochloride; sodium carbonate In dimethyl sulfoxide at 25 - 80℃; Reagent/catalyst; Solvent; Temperature; | 85.4% |
-
-
114772-54-2
4'-(bromomethyl)-1,1'-biphenyl-2-carbonitrile

-
-
88-74-4
2-nitro-aniline

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide Reflux; | 89% |
4-Bromomethyl-2-cyanobiphenyl Chemical Properties
Synonyms: 4'-BROMOMETHYLBIPHENYL-2-CARBONITRILE;[1,1'-BIPHENYL]-2-CARBONITRILE, 4'-(BROMOMETHYL)-;2-(4-BROMO-METHYLPHENYL) BENZONITRILE;OTBNBR ;2'-CYANO-4-BROMOMETHYL BIPHENYL;2-CYANO-4'-BROMOMETHYLBIPHENYL;4'-BROMOMETHYL-2-BIPHENYLCARBONITRILE;4'-BROMOMETHYL-2-CYANOBIPHENYL;
Molecular formula: C14H10BrN
Molecular formula: 272.14
EINECS: 425-280-5
Index of Refraction: 1.64
Molar Refractivity: 68.37 cm3
Molar Volume: 189.6 cm3
Surface Tension: 54.9 dyne/cm
Density: 1.43 g/cm3
Flash Point: 203.7 °C
Enthalpy of Vaporization: 66.6 kJ/mol
Boiling Point: 413.2 °C at 760 mmHg
Vapour Pressure: 4.87E-07 mmHg at 25 °C
Melting point: 125-128 °C(lit.)
Appearance: Cream powder
Structure of 2-Cyano-4'-bromomethylbiphenyl (CAS NO.114772-54-2):
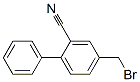
IUPAC Name: 2-[4-(Bromomethyl)phenyl]benzonitrile
Canonical SMILES: C1=CC=C(C(=C1)C#N)C2=CC=C(C=C2)CBr
InChI: InChI=1S/C14H10BrN/c15-9-11-5-7-12(8-6-11)14-4-2-1-3-13(14)10-16/h1-8H,9H2
InChIKey: LFFIEVAMVPCZNA-UHFFFAOYSA-N
Product Category of 2-Cyano-4'-bromomethylbiphenyl (CAS NO.114772-54-2): Chemical intermediate for Sartan;Biphenyl & Diphenyl ether;API intermediates;Sartan's serie;Hypertension;Intermediates of Irbesartan;Irbesartan
4-Bromomethyl-2-cyanobiphenyl Toxicity Data With Reference
2-Cyano-4'-bromomethylbiphenyl (CAS NO.114772-54-2) hasn't been listed as a carcinogen by NTP, IARC, ACGIH, or CA Prop 65. And the toxicological properties have not been fully investigated. You can see actual entry in RTECS for complete information.
4-Bromomethyl-2-cyanobiphenyl Safety Profile
Hazard Codes:  Xi,Xn
Xi,Xn
The Risk Statements information of 2-Cyano-4’-bromomethylbiphenyl:
20/21/22: Harmful by inhalation, in contact with skin and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
The Safety Statements information of 2-Cyano-4’-bromomethylbiphenyl:
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37: Wear suitable protective clothing and gloves
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
RIDADRP:3439
WGK Germany:3
Hazard Note:Irritant
4-Bromomethyl-2-cyanobiphenyl Specification
2-Cyano-4'-bromomethylbiphenyl , its cas register number is 114772-54-2. It also can be called 4-Bromomethyl-2-cyanobiphenyl ; and 4'-Bromomethyl-2-biphenylcarbonitrile . It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product: Wash mouth out with water, and get medical aid immediately. Notes to physician: Treat supportively and symptomatically.
In addition, 2-Cyano-4'-bromomethylbiphenyl (CAS NO.114772-54-2) could be stable under normal temperatures and pressures. It is not compatible with strong oxidizing agents, acids, strong bases, acid chlorides, carbon dioxide, acid anhydrides, and you must not take it with incompatible materials. And also prevent it to broken down into hazardous decomposition products: Hydrogen cyanide, nitrogen oxides, carbon monoxide, oxides of nitrogen, carbon dioxide, hydrogen bromide.
Related Products
- 4-Bromomethyl-2-cyanobiphenyl
- 114772-55-3
- 114776-15-7
- 1147-76-8
- 114780-06-2
- 1147872-22-7
- 114787-91-6
- 114788-05-5
- 114793-00-9
- 114798-26-4
- 114-80-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View