-
Name
8-Nitroquinoline
- EINECS 210-135-9
- CAS No. 607-35-2
- Article Data37
- CAS DataBase
- Density 1.354 g/cm3
- Solubility Soluble in ethanol, ethyl ether and benzene, slightly soluble in cold water.
- Melting Point 89-91 °C(lit.)
- Formula C9H6N2O2
- Boiling Point 323.1 °C at 760 mmHg
- Molecular Weight 174.159
- Flash Point 149.2 °C
- Transport Information
- Appearance white to light yellow crystal powder
- Safety 26-27-36/37/39-36
- Risk Codes 20/21/22-36/37/38-40-68
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms 8-Nitroqunioline;quinoline, 8-nitro-;5-20-07-00327 (Beilstein Handbook Reference);
- PSA 58.71000
- LogP 2.66620
Synthetic route

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; [bis(trifluoroacetoxy)iodo]benzene; sodium nitrite In acetonitrile at 20℃; for 3h; regioselective reaction; | 84% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-nitro-aniline; glycerol With sodium iodide at 20℃; for 0.0833333h; Stage #2: With sulfuric acid for 0.166667h; Cooling with ice; Stage #3: at 140℃; for 2h; | 81% |
| Stage #1: 2-nitro-aniline With sulfuric acid at 70 - 80℃; Stage #2: glycerol at 70 - 80℃; Temperature; Further stages; | 79% |
| With sulfuric acid In water for 4h; Reflux; | 72% |
-
-
39217-93-1
8-nitro-1,2,3,4-tetrahydroquinoline

-
-
607-35-2
8-nitroquinoline

| Conditions | Yield |
|---|---|
| With iodine; oxygen In 1,2-dichloro-benzene; toluene at 160℃; for 30h; | 77% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In water; glycerol Reflux; | 44% |

| Conditions | Yield |
|---|---|
| Stage #1: quinoline With sulfuric acid; nitric acid at 20℃; for 1h; Stage #2: With sodium carbonate In water | A 34% B 38% |
| With sulfuric acid; nitric acid In water at 0 - 20℃; for 1h; | A 35% B n/a |
| With sulfuric acid; nitric acid |

| Conditions | Yield |
|---|---|
| durch Nitrieren; |

-
-
607-35-2
8-nitroquinoline

| Conditions | Yield |
|---|---|
| With ammonia at 130 - 140℃; im Rohr; |

| Conditions | Yield |
|---|---|
| With phosphoric acid; water; orthoarsenic acid | |
| With hydrogenchloride; phosphoric acid In water at 80 - 95℃; for 7h; |

| Conditions | Yield |
|---|---|
| With nitric acid at 160℃; im Rohr; |

-
-
607-35-2
8-nitroquinoline

| Conditions | Yield |
|---|---|
| durch Erhitzen ueber den Schmelzpunkt; |

| Conditions | Yield |
|---|---|
| at 160℃; |
-
-
91-22-5
quinoline

-
-
7664-93-9
sulfuric acid

-
-
7697-37-2
nitric acid

-
A
-
607-35-2
8-nitroquinoline

-
B
-
607-34-1
5-nitroquinoline

| Conditions | Yield |
|---|---|
| unter Kuehlung; | |
| at 0℃; |

-
-
91-22-5
quinoline

-
-
7697-37-2
nitric acid

-
-
108-24-7
acetic anhydride

-
A
-
607-35-2
8-nitroquinoline

-
B
-
17576-53-3
3-nitroquinoline

-
C
-
613-50-3
6-nitroquinoline

-
-
91-22-5
quinoline

-
-
108-24-7
acetic anhydride

-
A
-
607-35-2
8-nitroquinoline

-
B
-
17576-53-3
3-nitroquinoline

-
C
-
613-50-3
6-nitroquinoline

-
-
91-22-5
quinoline

-
-
7664-93-9
sulfuric acid

-
-
7697-37-2
nitric acid

-
A
-
607-35-2
8-nitroquinoline

-
B
-
607-34-1
5-nitroquinoline

-
C
-
88609-20-5
6,8-dinitroquinoline

-
D
-
62163-05-7
5,7-dinitroquinoline

| Conditions | Yield |
|---|---|
| in der Hitze; |


| Conditions | Yield |
|---|---|
| beim Eintragen des Chinolin-nitrats; |

-
-
7664-93-9
sulfuric acid

-
-
7778-39-4, 121471-47-4
orthoarsenic acid

-
-
88-74-4
2-nitro-aniline

-
-
56-81-5
glycerol

-
-
607-35-2
8-nitroquinoline

-
-
7664-93-9
sulfuric acid

-
-
98-95-3
nitrobenzene

-
-
88-74-4
2-nitro-aniline

-
-
56-81-5
glycerol

-
-
607-35-2
8-nitroquinoline


| Conditions | Yield |
|---|---|
| at 140℃; im geschlossenen Rohr; |

| Conditions | Yield |
|---|---|
| With Ni-modified Beta zeolite In neat (no solvent) for 0.0833333h; Microwave irradiation; Reflux; Green chemistry; | 19.4 %Chromat. |

| Conditions | Yield |
|---|---|
| With graphitic carbon nitride; hydrazine hydrate In water at 70℃; for 24h; Irradiation; Sealed tube; Green chemistry; chemoselective reaction; | 100% |
| With hydrogen In water at 60℃; under 760.051 Torr; for 3h; Green chemistry; | 99% |
| With hydrogen In tetrahydrofuran; water at 120℃; under 22502.3 Torr; for 6h; Autoclave; | 99% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In acetic acid for 2h; | 100% |
| With N-Bromosuccinimide In acetic acid at 110℃; for 3h; | 81.4% |
| With N-Bromosuccinimide In acetic acid at 65 - 70℃; for 6h; Inert atmosphere; | 75% |
-
-
607-35-2
8-nitroquinoline

-
-
57743-06-3
N-(quinolin-8-yl)hydroxylamine

| Conditions | Yield |
|---|---|
| With 5% rhodium-on-charcoal; hydrazine hydrate In tetrahydrofuran at 0℃; for 4h; Inert atmosphere; | 99% |
| With 5% rhodium-on-charcoal; hydrazine hydrate In tetrahydrofuran at 0℃; Inert atmosphere; | 99% |
| With ethyl acetate; platinum Hydrogenation; |
-
-
607-35-2
8-nitroquinoline

-
-
158117-55-6
3,6-dibromo-8-nitroquinoline

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In acetic acid for 336h; Ambient temperature; | 99% |
-
-
607-35-2
8-nitroquinoline

-
-
497084-46-5
3-iodo-8-nitroquinoline

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide; acetic acid at 100℃; for 3h; | 99% |
| With N-iodo-succinimide; acetic acid at 62℃; for 22h; | 97% |
| With N-iodo-succinimide In acetic acid at 62℃; for 22.1667h; | 97% |
-
-
607-35-2
8-nitroquinoline

-
-
73868-16-3
3-chloro-8-nitroquinoline

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide In acetic acid for 2h; | 96% |
| With N-chloro-succinimide; acetic acid for 3.5h; Heating; | 34% |
| With N-chloro-succinimide; acetic acid at 110℃; for 1h; Inert atmosphere; | 30% |
| With sulfur dichloride |

| Conditions | Yield |
|---|---|
| In benzene for 5h; Reflux; | 95% |
| In dichloromethane at 0 - 35℃; Inert atmosphere; | 69% |
-
-
607-35-2
8-nitroquinoline

-
-
35427-68-0
1-chloro-N,N-dimethylmethane sulfonamide

-
-
105199-32-4
N,N-dimethyl-(8-nitro-7-quinolyl)methanesulfonamide

| Conditions | Yield |
|---|---|
| With sodium hydroxide In dimethyl sulfoxide at 20 - 25℃; for 0.5h; | 92% |

| Conditions | Yield |
|---|---|
| With formic acid; triethylamine In water; tert-butyl alcohol at 100℃; for 24h; | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: 8-nitroquinoline With sulfuric acid at 35℃; for 0.416667h; Stage #2: benzene at 65℃; for 0.616667h; Temperature; | 90% |

| Conditions | Yield |
|---|---|
| With tetrahydroxydiboron; 5%-palladium/activated carbon; water In acetonitrile at 80℃; for 24h; | 90% |
| With tetrahydroxydiboron; copper diacetate In acetonitrile at 80℃; for 24h; Schlenk technique; chemoselective reaction; | 68% |

| Conditions | Yield |
|---|---|
| With palladium; copper diacetate In N,N-dimethyl-formamide at 120℃; for 8h; Inert atmosphere; regioselective reaction; | 89% |

| Conditions | Yield |
|---|---|
| With palladium; copper diacetate In N,N-dimethyl-formamide at 120℃; for 8h; Inert atmosphere; regioselective reaction; | 89% |
-
-
607-35-2
8-nitroquinoline

-
-
35427-68-0
1-chloro-N,N-dimethylmethane sulfonamide

-
A
-
105199-33-5
5-(Dimethylaminosulfonylmethyl)-8-nitroquinoline

-
B
-
105199-32-4
N,N-dimethyl-(8-nitro-7-quinolyl)methanesulfonamide

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at -30 - -25℃; for 0.25h; | A 4% B 88% |


| Conditions | Yield |
|---|---|
| With tetrahydroxydiboron; 5%-palladium/activated carbon; water In acetonitrile at 80℃; for 24h; | 88% |
| With tetrahydroxydiboron; copper diacetate In acetonitrile at 80℃; for 24h; Schlenk technique; chemoselective reaction; | 70% |
-
-
607-35-2
8-nitroquinoline

-
-
42606-39-3
5-amino-8-nitroquinoline

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; potassium hydroxide In ethanol at 40 - 60℃; for 2.5h; | 87% |
| With potassium hydroxide; hydroxylamine hydrochloride In ethanol at 20℃; for 1h; | 55% |
| With hydrogenchloride; potassium hydroxide; ethanol; hydroxylamine |

| Conditions | Yield |
|---|---|
| With tetrahydroxydiboron; 5%-palladium/activated carbon; water In acetonitrile at 80℃; for 24h; | 87% |
| With tetrahydroxydiboron; copper diacetate In acetonitrile at 80℃; for 24h; Schlenk technique; chemoselective reaction; | 66% |

| Conditions | Yield |
|---|---|
| With tetrahydroxydiboron; 5%-palladium/activated carbon; water In acetonitrile at 80℃; for 24h; | 86% |
| With tetrahydroxydiboron; copper diacetate In acetonitrile at 80℃; for 24h; Schlenk technique; chemoselective reaction; | 82% |
| With hydrogen In tetrahydrofuran; water at 110℃; under 37503.8 Torr; for 24h; Autoclave; | 79 %Chromat. |
-
-
607-35-2
8-nitroquinoline

| Conditions | Yield |
|---|---|
| With palladium; copper diacetate In N,N-dimethyl-formamide at 120℃; for 8h; Inert atmosphere; regioselective reaction; | 86% |
-
-
607-35-2
8-nitroquinoline

-
-
7205-98-3
chloromethyl phenyl sulfone

-
A
-
105199-29-9
8-Nitro-7-(phenylsulfonylmethyl)quinoline

-
B
-
105199-30-2
8-Nitro-5-(phenylsulfonylmethyl)quinoline

| Conditions | Yield |
|---|---|
| With sodium hydroxide In dimethyl sulfoxide at 20 - 25℃; for 0.5h; | A 84% B 12% |

-
-
607-35-2
8-nitroquinoline

-
-
39217-93-1
8-nitro-1,2,3,4-tetrahydroquinoline

| Conditions | Yield |
|---|---|
| With iodine; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In dichloromethane at 20℃; Inert atmosphere; | 84% |
| With iron(II) triflate; C13H21N3O2 In chloroform at 40℃; for 4h; Schlenk technique; | 76% |
| With ammonia borane; C52H64N12Pd2S4(2+)*2F6P(1-) In dodecane; toluene at 100℃; for 24h; Schlenk technique; Inert atmosphere; regioselective reaction; | 58 %Chromat. |

| Conditions | Yield |
|---|---|
| With sodium hydride; dimethyl sulfoxide; potassium hexacyanoferrate(III) In paraffin oil at 20℃; for 6h; | 84% |
-
-
607-35-2
8-nitroquinoline

-
-
7205-98-3
chloromethyl phenyl sulfone

-
-
105199-29-9
8-Nitro-7-(phenylsulfonylmethyl)quinoline

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at -30 - -25℃; for 0.25h; | 83% |
-
-
607-35-2
8-nitroquinoline

-
-
124-40-3
dimethyl amine

-
-
868-85-9
Dimethyl phosphite

-
-
131214-36-3
dimethyl (9-aza-1-(dimethylamino)-5H-2-benzazepin-3-yl)phosphonate

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 5 - 10℃; | 81% |

-
-
607-35-2
8-nitroquinoline

-
-
54012-92-9
8-amino-1,2,3,4-tetrahydroquinoline

| Conditions | Yield |
|---|---|
| With hexarhodium hexadecacarbonyl; carbon monoxide; water In 2-methoxy-ethanol at 150℃; under 41188.4 Torr; for 48h; | 81% |
| With carbon monoxide; water In 2-methoxy-ethanol at 150℃; under 41188.4 Torr; for 48h; | 81% |
| With hydrogen; acetic acid; platinum(IV) oxide at 20℃; under 1034.32 Torr; for 20h; | 79% |
-
-
607-35-2
8-nitroquinoline

-
-
72047-94-0
[2-(acetoxymethyl)prop-2-enyl]trimethylsilane

| Conditions | Yield |
|---|---|
| Stage #1: 8-nitroquinoline With N,N-bis(-1-phenylethyl)dinaphtho[2,1-d:1',2'-f][1,3,2]dioxaphosphepin-4-amine; bis(dibenzylideneacetone)-palladium(0) In toluene at 25℃; for 0.0833333h; Inert atmosphere; Sealed tube; Stage #2: [2-(acetoxymethyl)prop-2-enyl]trimethylsilane In toluene at 25℃; for 15h; Inert atmosphere; Sealed tube; | 81% |
-
-
607-35-2
8-nitroquinoline

-
-
3598-13-8
(4-chlorophenoxy)acetonitrile

-
-
105214-06-0
2-(8-nitroquinolin-7-yl)acetonitrile

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In dimethyl sulfoxide at -16 - 20℃; for 2h; | 80% |
| With potassium tert-butylate In tetrahydrofuran at -10℃; for 0.416667h; | 58% |
-
-
607-35-2
8-nitroquinoline

-
-
110-13-4
2,5-hexanedione

-
-
32668-78-3
8-(2,5-dimethyl-1H-pyrrol-1-yl)quinoline

| Conditions | Yield |
|---|---|
| With indium; acetic acid In toluene at 80℃; for 24h; Inert atmosphere; | 80% |
| With hydrogen In neat (no solvent) at 120℃; under 30003 Torr; for 24h; Autoclave; Green chemistry; chemoselective reaction; | 55% |
8-NITROQUINOLINE Chemical Properties
8-Nitroquinoline(607-35-2) has a melting point of 89-91 °C(lit.).
The chemical synonyms of 8-Nitroquinoline(607-35-2) are 8-nitro-quinolin;Quinoline, 8-nitro-;Quinoline,8-nitro-;AKOS 214-03;8-NITROQUINOLINE;8-Nitroqunioline;8-Nitroquinoline,98%;8-Nitrochinolin
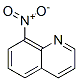
The molecular structure of 8-Nitroquinoline(607-35-2):
8-NITROQUINOLINE Toxicity Data With Reference
| 1. | mmo-sat 50 µg/plate | MUREAV Mutation Research. 39 (1977),285. | ||
| 2. | mma-sat 100 nmol/plate | MUREAV Mutation Research. 58 (1978),11. | ||
| 3. | dnr-sat 350 µg/disc | MUREAV Mutation Research. 39 (1977),285. | ||
| 4. | orl-rat TD:36,400 mg/kg/2Y-C:CAR,REP | CALEDQ Cancer Letters (Shannon, Ireland). 14 (1981),115. | ||
| 5. | ipr-mus LDLo:125 mg/kg | CBCCT* “Summary Tables of Biological Tests“ National Research Council Chemical-Biological Coordination Center. 6 (1954),149. |
8-NITROQUINOLINE Consensus Reports
8-NITROQUINOLINE Safety Profile
Hazard Codes Xn,Xi
Risk Statements 20/21/22-36/37/38-40-68
Safety Statements 26-27-36/37/39-36
WGK Germany 3
RTECS VC1925000
Hazard Note Harmful/Irritant
8-NITROQUINOLINE Specification
Conditions to Avoid: Incompatible materials.
Incompatibilities with Other Materials Strong oxidizing agents, strong acids, strong bases.
Hazardous Decomposition Products Nitrogen oxides, carbon monoxide, carbon dioxide.
Hazardous Polymerization Has not been reported.
Related Products
- 8-NITROQUINOLINE
- 607354-69-8
- 607366-20-1
- 607366-21-2
- 607371-03-9
- 607373-82-0
- 607373-83-1
- 607380-28-9
- 607-42-1
- 60748-07-4
- 6074-84-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View