-
Name
2-(1H-Benzotriazole-1-yl)-1,1,3,3-tetramethyluronium tetrafluoroborate
- EINECS 423-040-4
- CAS No. 125700-67-6
- Article Data5
- CAS DataBase
- Density
- Solubility acetonitrile: 0.1 g/mL, clear
- Melting Point 205°C (dec.)(lit.)
- Formula C11H16N5O.BF4
- Boiling Point
- Molecular Weight 321.086
- Flash Point STABILITY
- Transport Information
- Appearance white or off-white crystalline powder
- Safety 26-36-37/39-36/37/39
- Risk Codes 36/37/38-5
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, F
F
- Synonyms (1H-Benzotriazol-1-yl)-1,1,3,3-tetramethyluronium tetrafluoroborate;Btttfb;Methanaminium, N-((1H-benzotriazol-1-yloxy)(dimethylamino)methylene)-N-methyl-, tetrafluoroborate(1-);O-(Benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate;
- PSA 46.19000
- LogP 1.34960
Synthetic route
-
-
150-75-4
N-methyl-p-aminophenol

-
-
164646-22-4
(R)-N2-[(4-amino-3,5-dichlorophenyl)sulphonyl]-N5-[amino(nitroimino)methyl]-ornithine

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| 58% |


-
-
164648-71-9
(R)-N5-[amino(nitroimino)methyl]-N2-[(2-naphthyl)carbonyl]-ornithine

-
-
2243-83-6
2-naphthaloyl chloride

-
A
-
164647-69-2
(R)-N5-[Amino(nitroimino)methyl]-N-[(4-hydroxyphenyl)-methyl]-N2-(2-naphthoyl)-ornithinamide

-
B
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| A n/a B 36% |

-
-
1871-76-7
diphenylacetic acid chloride

-
A
-
164644-76-2
(R,S)-N5-(Aminocarbonyl)-N2-(diphenylacetyl)-N-[(4-hydroxyphenyl)methyl]-ornithinamide

-
B
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| A n/a B 34% |
-
-
56043-45-9, 13829-06-6
N,N,N',N'-tetramethylchlorformamidinium chloride

-
-
62244-77-3
1-Hydroxy-1,2,3-benzotriazole potassium salt

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With potassium chloride; potassium tetrafluoroborate |
-
-
632-22-4
tetramethylurea

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: oxalyl chloride 2: KBF4,KCl View Scheme |
-
-
108-30-5
succinic acid anhydride

-
B
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With dmap; benzotriazol-1-ol In ethyl acetate; N,N-dimethyl-formamide |
-
-
123-75-1
pyrrolidine

-
-
68837-59-2
4-bromo-2-methylbenzoic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
276678-35-4
(4-bromo-2-methylphenyl)(pyrrolidin-1-yl)methanone

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; water | 98% |


-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
57260-71-6
1-t-Butoxycarbonylpiperazine

| Conditions | Yield |
|---|---|
| With triethylamine In water; ethyl acetate; N,N-dimethyl-formamide | 97% |

-
-
861359-74-2
diisopropylethyl amine


-
-
222640-99-5
N-ethyl-N-[1-oxo-4-[1-[(phenylmethoxy)carbonyl]-4-piperidinyl]butyl]glycine

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In N-methyl-acetamide; N,N-dimethyl-formamide | 95% |

-
-
508-02-1
Oleanolic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
1596377-01-3
1-benzotriazolyl 3β-hydroxyolean-12-en-28-oate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 12h; | 95% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 5h; Inert atmosphere; | 93% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 5h; Inert atmosphere; | 93% |
-
-
77-52-1
ursolic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 5h; Inert atmosphere; | 93% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 5h; Inert atmosphere; | 93% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; | |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran | |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 4h; |
-
-
472-15-1
Betulinic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 5h; Inert atmosphere; | 93% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 5h; Inert atmosphere; | 93% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; | |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran |

-
-
1180495-95-7
(2S,4S)-1-(tert-butoxycarbonyl)-4-(8-chloro-2-(4-isopropylthiazol-2-yl)-7-methoxyquinolin-4-yloxy)pyrrolidine-2-carboxylic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide | 92% |

-
-
109-02-4
4-methyl-morpholine

-
-
68076-36-8
1-amino-4-[(tert-butyloxycarbonyl)amino]butane

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
1315605-54-9
{4-[3-({2-[(4-Cyano-phenylamino)-methyl]-1-methyl-1H-benzoimidazole-5-carbonyl}-phenyl-amino)-propionylamino]-butyl}-carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide | 92% |

-
-
465-99-6
hedaragenin

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; | 92% |

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine at 20℃; for 12h; | 92% |
-
-
4339-72-4
oleanolic acid 3-acetate

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine at 20℃; for 12h; | 91% |

-
-
1180495-88-8
(2S,4S)-1-(tert-butoxycarbonyl)-4-(7-methoxy-8-methyl-2-(4-(trifluoromethyl)thiazol-2-yl)quinolin-4-yloxy)pyrrolidine-2-carboxylic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide | 90% |

-
-
6089-92-5
(2α,3β)-2,3-diacetyloxy-olean-12-en-28-oic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine at 20℃; for 12h; | 90% |
-
-
471-53-4
enoxolone

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
1612838-71-7
1H‐benzo[d][1,2,3]triazol‐1‐yl‐3β‐hydroxy‐11‐oxo‐olean‐12‐en‐30‐oate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 24h; | 90% |
-
-
510-30-5
echinocystic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
1438766-88-1
1-benzotriazolyl 3β,16α-dihydroxyolean-12-en-28-oate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 4h; | 89% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; | 86% |
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; Inert atmosphere; | 86% |
-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
58-85-5
biotin

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide for 4h; | 89% |
-
-
1159252-14-8
6-[3-(5-fluoro-pyridin-2-yl)-5-methyl-isoxazol-4-ylmethoxy]-pyridazine-3-carboxylic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
124-68-5
2-Amino-2-methyl-1-propanol

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
1159252-13-7
6-[3-(5-fluoro-pyridin-2-yl)-5-methyl-isoxazol-4-ylmethoxy]-pyridazine-3-carboxylic acid (2-hydroxy-1,1-dimethyl-ethyl)-amide

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide | 88% |

-
-
1613191-77-7
2-amino-6-fluoro-pyrazolo[1,5-a]pyrimidine-3-carboxylic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
1613191-75-5
1H-benzo[d][1,2,3]triazol-1-yl 2-amino-6-fluoropyrazolo[1,5-a]pyrimidine-3-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: 2-amino-6-fluoro-pyrazolo[1,5-a]pyrimidine-3-carboxylic acid With triethylamine In chloroform for 0.0833333h; Stage #2: O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate In chloroform at 60℃; for 1h; | 88% |
| Stage #1: 2-amino-6-fluoro-pyrazolo[1,5-a]pyrimidine-3-carboxylic acid With triethylamine In chloroform for 0.0833333h; Stage #2: O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate In chloroform at 60℃; for 1h; | 88% |
| Stage #1: 2-amino-6-fluoro-pyrazolo[1,5-a]pyrimidine-3-carboxylic acid With triethylamine In chloroform for 0.0833333h; Stage #2: O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate In chloroform at 60℃; for 1h; | 88% |
| With triethylamine In chloroform at 60℃; for 1h; | 88% |
-
-
1613191-81-3
2-amino-6-chloro-pyrazolo[1,5-a]pyrimidine-3-carboxylic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
1613191-79-9
1H-benzo[d][1,2,3]triazol-1-yl 2-amino-6-chloropyrazolo[1,5-a]pyrimidine-3-carboxylate

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform at 50℃; for 1h; | 88% |
| With triethylamine In chloroform at 50℃; for 1h; | 88% |
| With triethylamine In chloroform at 50℃; for 1h; | 88% |
-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
99-57-0
2-hydroxy-5-nitroaniline

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 10h; | 86.5% |

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform at 50℃; for 0.333333h; | 86% |
| In chloroform at 50℃; for 0.333333h; | 86% |
-
-
1108656-90-1
N-methylhex-5-en-1-amine tosylate salt

-
-
1265883-88-2
(1R,2R,4R)-4-(tert-butyldiphenylsilanyloxy)cyclohexane-1,2-dicarboxylic acid 2-tert-butyl ester

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide | 85% |

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
669065-21-8
N(1)-(4-chlorophenyl)-2-hydrazino-2-thioxoacetamide

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 10h; | 84.6% |
-
-
1613191-85-7
2-amino-6-(cyanomethyl)-pyrazolo[1,5-a]pyrimidine-3-carboxylic acid

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
1613191-82-4
1H-benzo[d][1,2,3]triazol-1-yl 2-amino-6-(cyanomethyl)pyrazolo[1,5-a]pyrimidine-3-carboxylate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 1h; | 84% |
| With triethylamine In dichloromethane at 20℃; for 1h; | 84% |
| With triethylamine In dichloromethane at 20℃; for 1h; | 84% |

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
53617-35-9
4-(piperidin-4-yl)morpholine

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In ethanol; ethyl acetate; N,N-dimethyl-formamide | 84% |

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
185563-95-5
5-[4-(1-triphenylmethylimidazol-2-ylamino)butyl]isoxazoline-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide | 83% |

-
-
125700-67-6
O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate

-
-
132800-13-6
1-cyclobutyl-piperazine

| Conditions | Yield |
|---|---|
| With triethylamine; trifluoroacetic acid In water; dimethyl sulfoxide; acetonitrile | 83% |

Benzotriazol-1-yl-tetramethyluronium tetrafluoroborate Chemical Properties
IUPAC Name of TBTU (125700-67-6): [benzotriazol-1-yloxy(dimethylamino)methylidene]-dimethylazanium; boron(3+); tetrafluoride
Synonyms:O-Benzotriazol-1-yl-N,N,N',N'-tetramethyluronium tetrafluoroborate; 2-(1H-Benzotriazole-1-yl)-1,1,3,3-tetramethyluronium tetrafluoroborate
Molecular Formula: C11H16N5O.BF4
Molecular Weight : 321.08 g/mol
EINECS: 423-040-4
storage temp.: 2-8 °C
Melting point: 200 °C
solubility:acetonitrile: 0.1 g/mL, clear
Appearance: white or off-white crystalline powder
Following is the molecular structure of TBTU (125700-67-6):
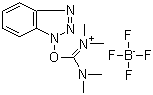
Benzotriazol-1-yl-tetramethyluronium tetrafluoroborate Safety Profile
Safty informations about TBTU (125700-67-6):
Hazard Symbols: Xi,
Xi, F
F
Risk Codes :
R36/37/38:Irritating to eyes, respiratory system and skin.
R5 :Heating may cause an explosion.
Safety Description :
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S37/39: Wear suitable gloves and eye/face protection.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
WGK Germany: 3
F: 8-10-21
Hazard Note: Irritant/Flammable
Related Products
- Benzotriazol-1-yl-tetramethyluronium tetrafluoroborate
- 125700-68-7
- 125700-69-8
- 125700-71-2
- 125700-72-3
- 125700-73-4
- 125706-19-6
- 1257-08-5
- 125-71-3
- 125713-88-4
- 125732-13-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View