-
Name
n-Butanoic acid
- EINECS 203-532-3
- CAS No. 107-92-6
- Article Data1027
- CAS DataBase
- Density 0.987 g/cm3
- Solubility miscible with water
- Melting Point -6 - -3 °C
- Formula C4H8O2
- Boiling Point 164.3 °C at 760 mmHg
- Molecular Weight 88.1063
- Flash Point 69 °C
- Transport Information UN 2820 8/PG 3
- Appearance colourless liquid
- Safety 26-36-45
- Risk Codes 34
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, C
C
- Synonyms n-Butanoic acid;n-Butyric acid;(3R,4S)-1-Benzoyl-3-(1-methoxy-1-methylethoxy)-4-phenyl-2-azetidinone;Butyricacid (6CI,7CI,8CI);1-Propanecarboxylic acid;Ethylacetic acid;Honey robber;NSC 8415;Propylformic acid;
- PSA 37.30000
- LogP 0.87110
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium hydroxide; hydrogen; [RhCl(Ph3P)2]; Ph2PO2CCH=CMe2 In acetone at 22℃; under 2280 Torr; for 17h; | 100% |
| With sodium tetrahydroborate; sodium hydroxide In water at 20 - 60℃; | 90% |
| With sodium hydroxide; hydrogen; nickel In water hydrogen generated in situ electrochemically on Raney nickel electrode; | 60% |

| Conditions | Yield |
|---|---|
| With 2,2,2-trichloroethylperoxycarbonic acid; dihydrogen peroxide In dichloromethane Ambient temperature; | 100% |
| With C4H11FeMo6NO24(3-)*3C16H36N(1+); water; oxygen; sodium carbonate at 50℃; under 760.051 Torr; for 8h; Green chemistry; | 99% |
| With 4H3N*4H(1+)*CuMo6O18(OH)6(4-); water; oxygen; sodium carbonate at 50℃; under 760.051 Torr; for 12h; | 98% |

| Conditions | Yield |
|---|---|
| With ammonium cerium (IV) nitrate; sodium trimethylsilylpropionate-d4; C18H22N4O2Ru(2+)*2F6P(1-); water at 20℃; for 0.5h; | 100% |
| With oxygen In water at 80℃; under 760.051 Torr; for 24h; | 99.7% |
| With potassium hydroxide at 50℃; electrolysis; | 98.8% |
-
-
13022-83-8
2-oxopentanoic acid sodium salt

-
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With calcium hypochlorite; acetic acid In dichloromethane; water; acetonitrile for 3h; Ambient temperature; | 96% |
-
-
199921-04-5
5-methyl-1-propyl-2,7,8-trioxabicyclo[3.2.1]octane

-
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| Stage #1: 5-methyl-1-propyl-2,7,8-trioxabicyclo[3.2.1]octane With pyridinium p-toluenesulfonate In methanol; water at 22℃; for 1.5h; Ring cleavage; Stage #2: With lithium hydroxide In tetrahydrofuran at 22℃; for 6h; Hydrolysis; | 96% |
-
-
3068-88-0, 32082-74-9, 36536-46-6, 65058-82-4
4-methyloxetan-2-one

-
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; W(OTf)6; hydrogen at 135℃; under 760.051 Torr; for 12h; | 96% |
| With palladium on activated carbon; W(OTf)6; hydrogen In neat (no solvent) at 135℃; under 760.051 Torr; for 12h; | 96% |
| With hydrogen at 200 - 247℃; under 15514.9 Torr; for 5.66667h; Inert atmosphere; | 94.2 %Chromat. |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; Na12[WZn3(H2O)2(ZnW9O34)2] at 75℃; for 7h; | A 95% B 4% |
| With Na12[WZn3(H2O)2(ZnW9O34)2]; dihydrogen peroxide at 85℃; for 7h; | |
| With SiW11Zn; dihydrogen peroxide In water at 89.85℃; for 9h; |
-
-
106-31-0
butanoic acid anhydride

-
-
691909-53-2
4-chloro-N-(2-fluoro-5-chlorophenyl)-N-(1R)-(2-hydroxy-1-methylpentyl)benzenesulfonamide

-
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane | 94% |

| Conditions | Yield |
|---|---|
| With benzyl(triethyl)ammoniumpermanganate In dichloromethane | A 93% B 5% |
-
-
352-93-2
diethyl sulphide

-
-
1821-02-9
2-oxopentanoic acid

-
A
-
70-29-1
diethyl sulphide

-
B
-
124-38-9
carbon dioxide

-
C
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With oxygen; methylene blue In pyridine; acetonitrile for 2h; Irradiation; | A 92% B n/a C 19% |


| Conditions | Yield |
|---|---|
| With octadecyltriethoxysilane-treated high-silica Hβ-20 zeolite In water at 130℃; for 24h; Reagent/catalyst; | 92% |
| With PIPES buffer; Candida rugosa lipase In water; toluene Rate constant; other enzymes, other solvent; | |
| With Candida antarctica lipase B; 4-nitro-phenol; MOPS buffer In water at 25℃; pH=7.2; Enzyme kinetics; Further Variations:; Reagents; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With sodium bromate; Ru2(dmnapy)Cl4 for 0.25h; Ambient temperature; | A 91.5% B 2.5% |
| With C30H24N2O7W; dihydrogen peroxide In water; acetonitrile for 14h; Reflux; | A 67% B 23% |
| With tert.-butylhydroperoxide; chromium tetra(tert-butoxide) In benzene at 20℃; for 24h; | A 16% B 53% |

| Conditions | Yield |
|---|---|
| With oxygen In neat (no solvent) at 30 - 40℃; with solvent (benzene); | A 0.8% B 90.5% |
| With oxygen; palladium diacetate; tetrabutyl-ammonium chloride In chlorobenzene at 10℃; Rate constant; Product distribution; other catalysts; | |
| With tetrachloromethane; oxygen; ozone at -20 - -10℃; |

| Conditions | Yield |
|---|---|
| enzyme from Synechocystis sp. PCC 6803 In phosphate buffer at 30℃; for 12h; pH=7.2; | 90% |
| With benzene-1,2-dicarboxylic acid for 0.666667h; microwave irradiation; | 83% |
| With chaetomium globosum (Q2GR86); water Reagent/catalyst; Enzymatic reaction; | |
| With nitrilase from Gordonia terrae In aq. phosphate buffer at 35℃; for 1h; pH=8; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| rhodium(II) acetate In chloroform | 90% |

| Conditions | Yield |
|---|---|
| A 90% B n/a |
-
-
110-15-6
succinic acid

-
A
-
109-99-9
tetrahydrofuran

-
B
-
96-48-0
4-butanolide

-
C
-
110-63-4
Butane-1,4-diol

-
D
-
107-92-6
butyric acid

-
E
-
106-97-8
n-butane

-
F
-
71-36-3
butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen In 1,4-dioxane at 139.84℃; under 60006 Torr; for 24h; Catalytic behavior; Reagent/catalyst; Time; Autoclave; Overall yield = > 99 %; | A 0.2% B 3.1% C 89% D n/a E n/a F 7.6% |

-
-
1009-14-9
phenyl butyl ketone

-
A
-
123-38-6
propionaldehyde

-
B
-
123-72-8
butyraldehyde

-
C
-
802294-64-0
propionic acid

-
D
-
65-85-0
benzoic acid

-
E
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With 5% active carbon-supported ruthenium; water; oxygen; calcium oxide at 100℃; for 12h; Reagent/catalyst; Time; | A n/a B n/a C n/a D 89% E n/a |


| Conditions | Yield |
|---|---|
| With sodium hypochlorite In acetonitrile at 20℃; for 1h; | 88% |
| With dihydrogen peroxide; tris(cetylpyridinium) 12-tungstophosphate In chloroform for 24h; Heating; | 9% |
-
-
22607-10-9
octane-4,5-diol

-
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With sodium hypochlorite In acetonitrile at 20℃; for 1.5h; | 88% |

| Conditions | Yield |
|---|---|
| In water for 0.25h; Decarboxylation; microwave irradiation; | 87% |
| In neat (no solvent) at 141℃; Kinetics; Thermodynamic data; ΔH(excit.), ΔS(excit.), ΔF(excit.); |

| Conditions | Yield |
|---|---|
| With water; 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In chloroform at 20℃; for 1h; | 87% |
| With sulfuric acid at 70 - 80℃; Equilibrium constant; Kinetics; | |
| With sulfuric acid In water at 55℃; Mechanism; Rate constant; | |
| With perchloric acid; water; potassium hexacyanoferrate(III) at 30℃; Rate constant; Thermodynamic data; Mechanism; Ea, ΔH(excit.), ΔG(excit.), ΔS(excit.), also in the presence of NaClO4, NaNO3, NaCl, KCl or LiCl, also in methanol, effect of substrate, K4Fe(CN)6 and HClO4 concentrations; |

| Conditions | Yield |
|---|---|
| copper(l) chloride at 0℃; for 0.25h; | 87% |

| Conditions | Yield |
|---|---|
| With copper(l) chloride In tetrahydrofuran at 0℃; for 0.25h; | 87% |

| Conditions | Yield |
|---|---|
| With dihydridotetrakis(triphenylphosphine)ruthenium; water; 1-Phenylbut-1-en-3-one In 1,2-dimethoxyethane at 180℃; for 24h; Product distribution; Mechanism; in the absence of hydrogen acceptor (benzalacetone); other aldehydes; | A n/a B 85% C n/a |

-
-
592-99-4, 7642-15-1, 14850-23-8
4-octene

-
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With potassium permanganate; iron(III) chloride In acetone at -78 - 20℃; for 29h; | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: allyl butyrate With sodium tetrahydroborate In dimethyl sulfoxide at 20℃; Stage #2: With hydrogenchloride; water In dimethyl sulfoxide for 0.5h; chemoselective reaction; | 85% |
-
-
192815-71-7
(5R)-5-phenylmorpholin-3-one

-
A
-
138713-44-7, 74572-03-5
(3R)-3-phenylmorpholine

-
B
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With [RuCl2(N-heterocyclic carbene)(bis[2-(diphenylphosphino)ethyl]amine)]; caesium carbonate In toluene; butan-1-ol at 150℃; under 37503.8 Torr; for 6h; Schlenk technique; Autoclave; | A 85% B 10% |

| Conditions | Yield |
|---|---|
| zirconium(IV) oxide at 200℃; in vapor-phase; | 100% |
| With HZSM-5 at 170℃; for 12h; Temperature; | 99% |
| With Thermomyces lanuginosus lipase immobilized in an ionic-exchange resin at 30℃; for 6h; Concentration; Temperature; Sonication; Molecular sieve; Enzymatic reaction; | 90% |

| Conditions | Yield |
|---|---|
| With [Al(H2O)6][MS]3 In cyclohexane for 1h; Reagent/catalyst; Dean-Stark; Reflux; | 100% |
| With Candida antarctica B lipase In 2,2,4-trimethylpentane at 40℃; for 3h; Enzymatic reaction; | 98% |
| With DOOl-AlCl3 superacid resin for 1.5h; Heating; | 97% |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B; ammonium carbamate In various solvent(s) at 25℃; for 408h; Substitution; | 100% |
| Stage #1: butyric acid With niobium pentachloride In dichloromethane Stage #2: With ammonia In dichloromethane at 45 - 50℃; for 1h; | 98% |
| With Candida antarctica lipase B; ammonium carbamate In various solvent(s) at 35℃; for 72h; Substitution; | 91% |
-
-
2043-61-0
cyclohexanecarbaldehyde

-
-
107-92-6
butyric acid

-
-
114049-45-5
2-(Cyclohexyl-hydroxy-methyl)-butyric acid

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran | 100% |
-
-
54107-24-3
3-(2-vinyloxyethoxy)-1,2-propylene carbonate

-
-
107-92-6
butyric acid

-
-
127827-84-3
Butyric acid 1-[2-(2-oxo-[1,3]dioxolan-4-ylmethoxy)-ethoxy]-ethyl ester

| Conditions | Yield |
|---|---|
| With heptafluorobutyric Acid at 75℃; for 3h; | 100% |
-
-
120-20-7
2-(3,4-dimethoxyphenyl)-ethylamine

-
-
107-92-6
butyric acid

-
-
81165-56-2
N-(3,4-Dimethoxyphenethyl)butyramide

| Conditions | Yield |
|---|---|
| Stage #1: 2-(3,4-dimethoxyphenyl)-ethylamine; 4-formyl-3,5-dimethoxyphenoxy-Merrifield resin With sodium tris(acetoxy)borohydride In acetic acid; N,N-dimethyl-formamide at 20℃; for 69h; Solid phase reaction; reductive amination; Stage #2: butyric acid With benzotriazol-1-ol; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; for 15h; Solid phase reaction; acylation; Stage #3: With dimethylsulfide; water; trifluoroacetic acid In dichloromethane at 20℃; for 15h; Solid phase reaction; cleavage of amide; | 100% |

-
-
2039-67-0
2-(3-methoxyphenyl)-1-ethanamine

-
-
107-92-6
butyric acid

-
-
138505-09-6
N-[2-(3-Methoxyphenyl)ethyl]butanamide

| Conditions | Yield |
|---|---|
| Stage #1: 2-(3-methoxyphenyl)-1-ethanamine; 4-formyl-3,5-dimethoxyphenoxy-Merrifield resin With sodium tris(acetoxy)borohydride In acetic acid; N,N-dimethyl-formamide at 20℃; for 69h; Solid phase reaction; reductive amination; Stage #2: butyric acid With benzotriazol-1-ol; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; for 15h; Solid phase reaction; acylation; Stage #3: With dimethylsulfide; water; trifluoroacetic acid In dichloromethane at 20℃; for 15h; Solid phase reaction; cleavage of amide; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: o-methoxy-2-phenylethylamine; 4-formyl-3,5-dimethoxyphenoxy-Merrifield resin With sodium tris(acetoxy)borohydride In acetic acid; N,N-dimethyl-formamide at 20℃; for 69h; Solid phase reaction; reductive amination; Stage #2: butyric acid With benzotriazol-1-ol; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; for 15h; Solid phase reaction; acylation; Stage #3: With dimethylsulfide; water; trifluoroacetic acid In dichloromethane at 20℃; for 15h; Solid phase reaction; cleavage of amide; | 100% |


| Conditions | Yield |
|---|---|
| Stage #1: 4-Methoxyphenethylamine; 4-formyl-3,5-dimethoxyphenoxy-Merrifield resin With sodium tris(acetoxy)borohydride In acetic acid; N,N-dimethyl-formamide at 20℃; for 69h; Solid phase reaction; reductive amination; Stage #2: butyric acid With benzotriazol-1-ol; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; for 15h; Solid phase reaction; acylation; Stage #3: With dimethylsulfide; water; trifluoroacetic acid In dichloromethane at 20℃; for 15h; Solid phase reaction; cleavage of amide; | 100% |

-
-
13078-79-0
2-(3-chlorophenyl)ethylamine

-
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-(3-chlorophenyl)ethylamine; 4-formyl-3,5-dimethoxyphenoxy-Merrifield resin With sodium tris(acetoxy)borohydride In acetic acid; N,N-dimethyl-formamide at 20℃; for 69h; Solid phase reaction; reductive amination; Stage #2: butyric acid With benzotriazol-1-ol; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; for 15h; Solid phase reaction; acylation; Stage #3: With dimethylsulfide; water; trifluoroacetic acid In dichloromethane at 20℃; for 15h; Solid phase reaction; cleavage of amide; | 100% |


| Conditions | Yield |
|---|---|
| In methanol for 18h; Joullie-Ugi coupling; | 100% |

-
-
380848-17-9
(S)-5-phenylpent-1-en-3-amine

-
-
107-92-6
butyric acid

-
-
380848-27-1
N-(1-phenethyl-allyl)-butyramide

| Conditions | Yield |
|---|---|
| Stage #1: butyric acid With 4-methyl-morpholine; isobutyl chloroformate In tetrahydrofuran at -20℃; for 0.75h; Stage #2: With pyridine; 4-hydroxy-3-nitrobenzophenone resin In tetrahydrofuran; N,N-dimethyl-formamide for 12h; Stage #3: (S)-5-phenylpent-1-en-3-amine In dichloromethane for 24h; Further stages.; | 100% |

| Conditions | Yield |
|---|---|
| In methanol for 18h; Ugi-type reaction; | 100% |

-
-
49715-04-0
chlorosulfuric acid chloromethyl ester

-
-
107-92-6
butyric acid

-
-
33657-49-7
chloromethyl n-butyrate

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium hydrogencarbonate In dichloromethane; water at 20℃; for 16h; | 100% |
| With tetra(n-butyl)ammonium hydrogensulfate; potassium carbonate In dichloromethane; water at 20℃; for 4h; Inert atmosphere; | 58% |
| With sodium hydrogencarbonate; tetra(n-butyl)ammonium hydrogen sulfate In dichloromethane; water | 41% |
| With tetra(n-butyl)ammonium hydrogensulfate; sodium hydrogencarbonate In dichloromethane; water at 0℃; for 1h; | 31% |

| Conditions | Yield |
|---|---|
| Zn4(OAc)6O In chlorobenzene for 12h; Product distribution / selectivity; Heating / reflux; | 100% |
| Zn4(OCOCF3)6O In chlorobenzene for 12h; Product distribution / selectivity; Heating / reflux; | 100% |
-
-
2033-24-1
cycl-isopropylidene malonate

-
-
107-92-6
butyric acid

-
-
72546-06-6
5-butanoyl-2,2-dimethyl-1,3-dioxane-4,6-dione

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; | 100% |
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 16h; Inert atmosphere; | 100% |
| With dmap In dichloromethane; N,N-dimethyl-formamide |
-
-
78-73-9
cholinium hydrogen carbonate

-
-
107-92-6
butyric acid

-
-
93458-02-7
2-hydroxy-N,N,N-trimethylethan-1-aminium butyrate

| Conditions | Yield |
|---|---|
| In water at 20℃; | 100% |
| In water at 20℃; | |
| at 75℃; for 12h; |
-
-
1381872-39-4
C15H21NO3

-
-
72155-45-4
Boc-L-phenylalaninal

-
-
107-92-6
butyric acid

-
-
1381872-97-4
C33H48N2O8

| Conditions | Yield |
|---|---|
| In dichloromethane Passerini reaction; | 100% |

-
-
501-94-0
p-hydroxyphenethyl alcohol

-
-
107-92-6
butyric acid

-
-
386263-87-2
2-(4-hydroxylphenyl)ethyl butyrate

| Conditions | Yield |
|---|---|
| With Candida antarctica at 45℃; for 24h; | 100% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 0 - 20℃; for 24h; |
-
-
1438397-77-3
N-(2-aminoethyl)-4-methyl-N-(prop-2-yn-1-yl)benzenesulfonamide

-
-
107-92-6
butyric acid

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B at 50℃; for 4h; Molecular sieve; Ionic liquid; Green chemistry; Enzymatic reaction; | 99.9% |
| copper methanesulfonate In cyclohexane at 130 - 135℃; for 2.5h; | 96% |
| With salicylic acid resin supported FeCl3 In benzene at 125℃; for 1.96667h; | 95.8% |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B at 50℃; for 4h; Molecular sieve; Ionic liquid; Green chemistry; Enzymatic reaction; | 99.9% |
| In n-heptane at 40℃; for 24h; lipozyme IM 20 (immobilized Mucor miehi lipase); | 94.66% |
| In water at 30℃; for 18h; lipase from Aspergillus niger; Yield given; |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B at 50℃; for 4h; Molecular sieve; Ionic liquid; Green chemistry; Enzymatic reaction; | 99.9% |
| With sodium hydroxide at 80℃; for 8h; | 66.94% |
| In hexane at 30℃; for 72h; Corynebacterium sp. S-401; | 23% |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B at 50℃; for 4h; Molecular sieve; Ionic liquid; Green chemistry; Enzymatic reaction; | 99.9% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane Steglich Esterification; |

| Conditions | Yield |
|---|---|
| With aluminum(III) sulphate octadecahydrate at 110℃; for 0.166667h; Sealed tube; Microwave irradiation; | 99.7% |
| With NiO/SiO2 at 360℃; Reagent/catalyst; | 97.98% |
| With 1-methyl-3-(4-sulfobutyl)-1H-imidazol-3-ium hydrogensulfate at 80℃; for 2h; | 96% |
-
-
107-92-6
butyric acid

-
-
13122-71-9
peroxybutyric acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; KU-2 x 8 cation-exchange resin In 1,4-dioxane at 19.9℃; Mechanism; Product distribution; Kinetics; other temperatures; | 99.2% |
| With tetraacetyl diborate; xylene unter vermindertem Druck unter Entfernen von Essigsaeure und Behandeln des Reaktionsprodukts mit Wasserstoffperoxid in Aether; | |
| With dihydrogen peroxide | |
| With sulfuric acid; dihydrogen peroxide at -10 - -5℃; Oxidation; |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In chlorobenzene at 80℃; Friedel-Crafts Acylation; | 99% |
| With zinc(II) chloride In toluene Reflux; | 89.3% |
| With PPA |
Butyric acid Specification
The IUPAC name of this chemical is Butyric acid. With the CAS registry number 107-92-6 and EINECS registry number 203-532-3, it is also named as ethylacetic acid. In addition, the molecular formula is C4H8O2. It is an oily colorless liquid that is easily soluble in water, ethanol, and ether, and can be separated from an aqueous phase by saturation with salts such as calcium chloride. And it has an unpleasant smell and acrid taste. Besides, it is found in butter, parmesan cheese, vomit, and as a product of anaerobic fermentation (including in the colon and as body odor).
Physical properties about this chemical are: (1)ACD/LogP: 0.70; (2)# of Rule of 5 Violations: 0; (3)ACD/BCF (pH 5.5): 1; (4)ACD/BCF (pH 7.4): 1; (5)ACD/KOC (pH 5.5): 8.756; (6)ACD/KOC (pH 7.4): 1; (7)#H bond acceptors: 2; (8)#H bond donors: 1; (9)#Freely Rotating Bonds: 2; (10)Polar Surface Area: 37.3 Å2; (11)Index of Refraction: 1.411; (12)Molar Refractivity: 22.145 cm3; (13)Molar Volume: 89.188 cm3; (14)Polarizability: 8.779 ×10-24cm3; (15)Surface Tension: 32.584 dyne/cm; (16)Density: 0.988 g/cm3; (17)Flash Point: 69.712 °C; (18)Enthalpy of Vaporization: 42.395 kJ/mol; (19)Boiling Point: 164.348 °C at 760 mmHg; (20)Vapour Pressure: 1.351 mmHg at 25°C.
Preparation of Butyric acid: It is industrially prepared by the fermentation of sugar or starch, brought about by the addition of putrefying cheese, with calcium carbonate added to neutralize the acids formed in the process. In addition, it can be prepared by butan-1-ol. The other product is butyraldehyde. This reaction will need reagent NaBrO3 and catalyst Ru2(dmnapy)Cl4. The reaction time is 15 hours with ambient temperature. The yield is about 91.5%.
![]()
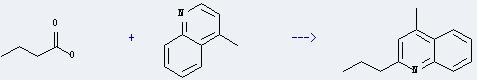
Uses of Butyric acid: it can be used as food, perfume and fishing bait additives, an animal feed supplement, an approved food flavouring. In addition, it is used in the preparation of various butyrate esters. Besides, it can react with 4-methyl-quinoline to get 4-methyl-2-propyl-quinoline. This reaction will need reagents sulfuric acid and iron(III) sulfate, and solvent H2O. The reaction time is 20 hours with irradiation. The yield is about 76%.
When you are using this chemical, please be cautious about it as the following:
This chemical can cause burns. During using it, wear suitable protective clothing. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. And in case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.).
You can still convert the following datas into molecular structure:
(1)SMILES: CCCC(=O)O
(2)InChI: InChI=1/C4H8O2/c1-2-3-4(5)6/h2-3H2,1H3,(H,5,6)
(3)InChIKey: FERIUCNNQQJTOY-UHFFFAOYAP
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LC | inhalation | > 500mg/m3 (500mg/m3) | LUNGS, THORAX, OR RESPIRATION: STRUCTURAL OR FUNCTIONAL CHANGE IN TRACHEA OR BRONCHI | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 23(5), Pg. 31, 1958. |
| mouse | LD50 | intraperitoneal | 3180mg/kg (3180mg/kg) | Journal of Pharmacy and Pharmacology. Vol. 21, Pg. 85, 1969. | |
| mouse | LD50 | intravenous | 800mg/kg (800mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Acta Pharmacologica et Toxicologica. Vol. 18, Pg. 141, 1961. |
| mouse | LD50 | subcutaneous | 3180mg/kg (3180mg/kg) | Journal of Pharmacy and Pharmacology. Vol. 21, Pg. 85, 1969. | |
| mouse | LDLo | oral | 500mg/kg (500mg/kg) | GASTROINTESTINAL: NECROTIC GHANGES KIDNEY, URETER, AND BLADDER: OTHER CHANGES BLOOD: CHANGES IN SPLEEN | Toksikologiya Novykh Promyshlennykh Khimicheskikh Veshchestv. Toxicology of New Industrial Chemical Substances. For English translation, see TNICS*. Vol. 4, Pg. 19, 1962. |
| rabbit | LD50 | skin | 530uL/kg (0.53mL/kg) | Union Carbide Data Sheet. Vol. 4/10/1968, | |
| rat | LC | inhalation | > 500mg/m3 (500mg/m3) | LUNGS, THORAX, OR RESPIRATION: STRUCTURAL OR FUNCTIONAL CHANGE IN TRACHEA OR BRONCHI | Toksikologiya Novykh Promyshlennykh Khimicheskikh Veshchestv. Toxicology of New Industrial Chemical Substances. For English translation, see TNICS*. Vol. 4, Pg. 19, 1962. |
| rat | LD50 | oral | 2gm/kg (2000mg/kg) | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 30, 1982. |
Related Products
- Butyric acid
- Butyric acid hydrazide
- Butyric acid, 2-(5-nitro-alpha-iminofurfuryl)hydrazide
- Butyric acid, 4-fluoro-2-hydroxythio-, S-methyl ester
- Butyric acid, p-methoxyphenyl ester
- Butyric anhydride
- 107929-91-9
- 107933-72-2
- 107934-68-9
- 107-93-7
- 107937-17-7
- 107947-17-1
- 107-94-8
- 107949-21-3
- 107-95-9
- 107-96-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View