-
Name
Chloral hydrate
- EINECS 206-117-5
- CAS No. 302-17-0
- Article Data27
- CAS DataBase
- Density 1.814 g/cm3
- Solubility 660 g/100ml in water
- Melting Point 57 °C(lit.)
- Formula C2H3Cl3O2
- Boiling Point 96.3 °C at 760 mmHg
- Molecular Weight 165.404
- Flash Point 57.3 °C
- Transport Information UN 3286 3/PG 2
- Appearance colourless crystals with a pentrating odour
- Safety 26-45-25-23-36/37-16-27
- Risk Codes 25-36/38-39/23/24/25-23/24/25-11-36/37/38
-
Molecular Structure
-
Hazard Symbols
 T,
T,  F
F
- Synonyms Chloralhydrate (8CI);1,1,1-Trichloro-2,2-dihydroxyethane;2,2,2-Trichloro-1,1-ethanediol;Bi 3411;Chloral, monohydrate;Chloraldurat;TCA;
- PSA 40.46000
- LogP 0.66730
Synthetic route

| Conditions | Yield |
|---|---|
| In tetrachloromethane | 45% |
| With water Thermodynamic data; ΔH; | |
| With water | |
| In water |
-
-
473-34-7
N,N-dichloro-p-toluenesulfonamide

-
-
79-01-6
Trichloroethylene

-
A
-
302-17-0
chloral hydrate

-
B
-
83790-89-0
N-(2,2,2-trichloro-1-hydroxyethyl)4-methylbenzenesulfonamide

-
C
-
93018-67-8
N-(2,2,2-trichloro-1-p-toluenesulfonamidoethyl)-p-toluenesulfonamide

-
D
-
70-55-3
toluene-4-sulfonamide

| Conditions | Yield |
|---|---|
| With water; tin(IV) chloride at 20 - 22℃; for 1560h; Product distribution; other time, temperature, Lewis acid (AlCl3); | A .11 g B 37.9% C 2.7% D 2.9% |


| Conditions | Yield |
|---|---|
| With water; chlorine |

| Conditions | Yield |
|---|---|
| With sodium hypochlorite; water; boric acid |

| Conditions | Yield |
|---|---|
| With water Chlorieren; |

| Conditions | Yield |
|---|---|
| With water Chlorieren; |

| Conditions | Yield |
|---|---|
| With water; chlorine at 90℃; | |
| With water; iodine; chlorine at 90℃; |

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride at 170℃; |
-
-
75-87-6
chloral

-
-
147624-92-8
2,2-Dihydroxy-3-oxo-pent-4-enoic acid tert-butyl ester

-
A
-
302-17-0
chloral hydrate

-
B
-
117917-40-5
2,3-Dioxo-pent-4-enoic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| In acetone Equilibrium constant; Thermodynamic data; ΔH; |


| Conditions | Yield |
|---|---|
| at 170℃; |
-
-
593-63-5
chloroacetylene

-
-
7732-18-5
water

-
-
13460-50-9
metaboric acid

-
A
-
302-17-0
chloral hydrate

-
B
-
79-11-8
chloroacetic acid

-
-
15687-05-5
acetic acid-[4-(2,2,2-trichloro-1-hydroxy-ethoxy)-anilide]

-
-
7732-18-5
water

-
A
-
302-17-0
chloral hydrate

-
B
-
103-90-2
4-acetaminophenol


-
A
-
302-17-0
chloral hydrate

-
B
-
102603-74-7
(2,2-diphenyl-ethylidene)-(2,2-diphenyl-vinyl)-amine

-
-
20752-32-3
1-bornyloxy-2,2,2-trichloro-ethanol

-
-
7732-18-5
water

-
A
-
302-17-0
chloral hydrate

-
-
6627-72-1
rac-endo-borneol

| Conditions | Yield |
|---|---|
| chloral-l-borneolate; | |
| chloral-d-borneolate; |

-
-
79-01-6
Trichloroethylene

-
A
-
115-20-8
1,1,1-trichloroethanol

-
B
-
302-17-0
chloral hydrate

-
C
-
76-03-9
trichloroacetic acid

| Conditions | Yield |
|---|---|
| In acetone at 37℃; for 1h; Enzyme kinetics; Oxidation; hydroxylation; Enzymatic reaction; |

-
-
93018-67-8
N-(2,2,2-trichloro-1-p-toluenesulfonamidoethyl)-p-toluenesulfonamide

-
A
-
302-17-0
chloral hydrate

-
B
-
70-55-3
toluene-4-sulfonamide

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; for 0.5h; Product distribution; |
-
-
75457-08-8
N-(2,2,2-trichloro-1-hydroxyethyl)benzenesulfonamide

-
A
-
98-10-2
benzenesulfonamide

-
B
-
302-17-0
chloral hydrate

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; for 0.5h; Product distribution; |
-
-
107905-40-8
N-(2,2,2-trichloro-1-p-chlorobenzenesulfonamidoethyl)-p-chlorobenzenesulfonamide

-
A
-
98-64-6
4-Chlorobenzenesulfonamide

-
B
-
302-17-0
chloral hydrate

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; for 0.5h; Product distribution; |
-
-
85095-84-7
N-(2,2,2-trichloro-1-benzenesulfonamidoethyl)benzenesulfonamide

-
A
-
98-10-2
benzenesulfonamide

-
B
-
302-17-0
chloral hydrate

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 20℃; for 0.5h; Product distribution; |
-
-
56-41-7
L-alanin

-
A
-
109-06-8
α-picoline

-
B
-
302-17-0
chloral hydrate

-
D
-
5625-46-7
cyclo(-DL-Ala-DL-Ala-)

| Conditions | Yield |
|---|---|
| With copper dichloride at 250℃; for 0.00555556h; Pyrolysis; Inert atmosphere; |


| Conditions | Yield |
|---|---|
| at 250℃; for 0.00555556h; Pyrolysis; Inert atmosphere; |

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
A
-
302-17-0
chloral hydrate

-
B
-
67-66-3
chloroform

-
C
-
513-88-2
1,1-Dichloroacetone

-
D
-
918-00-3
1,1,1-trichloroacetone

| Conditions | Yield |
|---|---|
| With chlorine In aq. phosphate buffer pH=7; Kinetics; pH-value; UV-irradiation; |

-
-
302-17-0
chloral hydrate

-
-
1118-12-3
1,1-dimethylethylurea

-
-
96010-48-9
N-tert-butyl-N'-(2,2,2-trichloro-1-hydroxyethyl)urea

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water for 15h; | 100% |
-
-
302-17-0
chloral hydrate

-
-
150208-33-6
1-[2-(3,4-Dichloro-phenyl)-ethyl]-[1,4]diazepane

-
-
150208-82-5
1-<2-(3,4-dichlorophenyl)ethyl>-4-formylhomopiperazine

| Conditions | Yield |
|---|---|
| In toluene at 90℃; for 1.5h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: chloral hydrate; 4-propylaniline With hydrogenchloride; hydroxylamine hydrochloride; sodium sulfate In water for 0.0166667h; Reflux; Stage #2: With sulfuric acid at 50 - 80℃; | 100% |
-
-
302-17-0
chloral hydrate

-
-
1309089-16-4
ethyl 2-[4-[[2-hydroxyiminoacetyl]amino]phenyl]cyclobutanecarboxylate

| Conditions | Yield |
|---|---|
| Stage #1: chloral hydrate With sodium sulfate In water at 23℃; for 0.166667h; Stage #2: ethyl 1-(4-aminophenyl)cyclobutanecarboxylate hydrochloride With hydroxylamine hydrochloride In water at 60℃; for 5h; | 100% |
-
-
302-17-0
chloral hydrate

-
-
55289-36-6
3-bromo-2-methylaniline

-
-
129833-54-1
6-bromo-7-methyl-1H-indole-2,3-dione

| Conditions | Yield |
|---|---|
| Stage #1: chloral hydrate; 3-bromo-2-methylaniline With hydrogenchloride; water; sodium sulfate at 20℃; Stage #2: With hydroxylamine hydrochloride Reflux; | 100% |
| Stage #1: chloral hydrate; 3-bromo-2-methylaniline With hydrogenchloride; sodium sulfate In water at 20℃; Stage #2: With hydroxylamine hydrochloride In water Reflux; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: chloral hydrate; 4-fluor-2-methylaniline With hydrogenchloride; sodium sulfate In water at 20℃; Stage #2: With hydroxylamine hydrochloride In water Reflux; | 100% |
| With hydroxylamine hydrochloride at 80℃; for 1h; | 47% |
-
-
302-17-0
chloral hydrate

-
-
55289-36-6
3-bromo-2-methylaniline

| Conditions | Yield |
|---|---|
| Stage #1: chloral hydrate; 3-bromo-2-methylaniline With hydrogenchloride; sodium sulfate In water at 20℃; Stage #2: With hydroxylamine hydrochloride In water Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With phosphorus pentoxide; sulfuric acid | 99% |
| With diethyl ether; water; chlorine at 80℃; | |
| With sulfuric acid |
-
-
302-17-0
chloral hydrate

-
-
97-67-6
(S)-Malic acid

-
-
196512-77-3
(2RS,5S)-5-carboxymethyl-2-trichloromethyl-4-oxo-1,3-dioxolane

| Conditions | Yield |
|---|---|
| With sulfuric acid for 10h; Ambient temperature; | 99% |
| With sulfuric acid at 20℃; for 3h; | 93% |
| With sulfuric acid at 20℃; for 3h; Inert atmosphere; | 93% |
| In sulfuric acid at 0 - 20℃; | 52% |
-
-
58534-95-5
3-bromo-2-fluorobenzenamine

-
-
302-17-0
chloral hydrate

-
-
1336963-99-5
N-(3-bromo-2-fluorophenyl)-2-(hydroxyimino)acetamide

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydroxylamine hydrochloride; sodium sulfate In water at 55 - 90℃; for 3h; | 99% |
| Stage #1: 3-bromo-2-fluorobenzenamine; chloral hydrate With hydrogenchloride; sodium sulfate In water at 35℃; Stage #2: With hydroxylamine hydrochloride at 90℃; for 16h; | 61% |
| Stage #1: chloral hydrate With sodium sulfate In water at 35℃; Stage #2: 3-bromo-2-fluorobenzenamine With hydrogenchloride; hydroxylamine hydrochloride In water at 90℃; for 16h; | 61% |
-
-
302-17-0
chloral hydrate

-
-
17275-82-0
ethyl 3,5-dimethoxybenzoate

-
-
63585-71-7
4,6-dimethoxy-3-trichloromethyl-1-benzo[c]furanone

| Conditions | Yield |
|---|---|
| With sulfuric acid for 4h; | 98% |
| With sulfuric acid |
-
-
302-17-0
chloral hydrate

-
-
79-05-0
Propionamid

-
-
34243-50-0
N-(2,2,2-trichloro-1-hydroxyethyl)propionamide

| Conditions | Yield |
|---|---|
| at 90 - 100℃; | 98% |
-
-
302-17-0
chloral hydrate

-
-
17792-50-6
Phenyl-phosphonic acid bis-(2,2,3,3,4,4,5,5-octafluoro-pentyl) ester

-
-
122348-12-3
Phenyl-(2,2,2-trichloro-1-hydroxy-ethyl)-phosphinic acid 2,2,3,3,4,4,5,5-octafluoro-pentyl ester

| Conditions | Yield |
|---|---|
| In benzene temp. not higher than 40 deg C; | 98% |
-
-
302-17-0
chloral hydrate

-
-
172422-37-6
Phenyl-phosphonic acid bis-(2,2,2-trifluoro-ethyl) ester

-
-
122348-13-4
Phenyl-(2,2,2-trichloro-1-hydroxy-ethyl)-phosphinic acid 2,2,2-trifluoro-ethyl ester

| Conditions | Yield |
|---|---|
| In benzene temp. not higher than 40 deg C; | 98% |
-
-
302-17-0
chloral hydrate

-
-
1309089-22-2
ethyl 2-[4-[[(2E)-2-hydroxyiminoacetyl]amino]phenyl]-2-methylpropanoate

| Conditions | Yield |
|---|---|
| Stage #1: chloral hydrate With sodium sulfate In water at 23℃; for 0.166667h; Stage #2: 2-(4-amino-phenyl)-2-methyl-propionic acid ethyl ester; hydrochloride With hydroxylamine hydrochloride In water at 60℃; for 5h; | 98% |
-
-
302-17-0
chloral hydrate

-
-
586-38-9
3-Methoxybenzoic acid

-
-
82735-28-2
6-methoxy-3-(trichloromethyl)isobenzofuran-1(3H)-one

| Conditions | Yield |
|---|---|
| With sulfuric acid at 20℃; for 12h; | 97% |
| With sulfuric acid at 20℃; for 12h; | 83.6% |
| With sulfuric acid at 20℃; for 24h; | 63% |
-
-
302-17-0
chloral hydrate

-
-
598-50-5
N-Methylurea

-
-
1954-79-6
N-methyl-N'-(2,2,2-trichloro-1-hydroxyethyl)urea

| Conditions | Yield |
|---|---|
| In water for 48h; Substitution; | 97% |
| With water |

| Conditions | Yield |
|---|---|
| With 3 A molecular sieve In 1,4-dioxane at 70℃; for 168h; Substitution; | 97% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydroxylamine hydrochloride; sodium sulfate at 90℃; | 97% |
-
-
302-17-0
chloral hydrate

| Conditions | Yield |
|---|---|
| With sodium hydride In diethyl ether for 3h; | 97% |
-
-
302-17-0
chloral hydrate

-
-
7149-75-9
3-methyl-4-chloroaniline

-
-
637347-65-0
N-(4-chloro-3-methylphenyl)-2-(hydroxyimino)acetamide

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydroxylamine hydrochloride; sodium sulfate In water at 100℃; for 1h; | 97% |
-
-
302-17-0
chloral hydrate

-
-
940054-43-3
3-chloro-5-fluoroaniline hydrochloride

-
-
940054-44-4
N-(3-chloro-5-fluorophenyl)-2-hydroxyimino-acetamide

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium sulfate In water at 20℃; for 16.6667h; Heating / reflux; | 97% |

| Conditions | Yield |
|---|---|
| In benzene at 30 - 40℃; Abramov reaction; Inert atmosphere; | A n/a B 97% |

-
-
302-17-0
chloral hydrate

-
-
51085-49-5
2-fluoroaniline hydrochloride

-
-
349-24-6
N-(2-fluorophenyl)-2-(hydroxyimino)acetamide

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium sulfate In water for 0.166667h; Reflux; | 97% |
-
-
348-54-9
2-Fluoroaniline

-
-
302-17-0
chloral hydrate

-
-
349-24-6
N-(2-Fluorophenyl)-2-hydroxyiminoacetamide

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydroxylamine hydrochloride; sodium sulfate In water for 0.166667h; Heating; | 96% |
| With hydrogenchloride; hydroxylamine; sodium sulfate | |
| With hydrogenchloride; hydroxylamine hydrochloride; sodium sulfate at 80 - 90℃; for 2h; | |
| Stage #1: 2-Fluoroaniline; chloral hydrate With hydrogenchloride; sodium sulfate In water at 20℃; for 5h; Stage #2: With hydroxylamine hydrochloride In water at 60℃; for 1h; |

| Conditions | Yield |
|---|---|
| With sulfuric acid | 96% |
| With sulfuric acid at 20℃; -chloralide; |

| Conditions | Yield |
|---|---|
| at 90 - 100℃; | 96% |
| Heating; |

| Conditions | Yield |
|---|---|
| In benzene at 30 - 40℃; Abramov reaction; Inert atmosphere; | A n/a B 96% |

-
-
239122-51-1
5-bromo-3-fluoro-2-methoxyaniline

-
-
302-17-0
chloral hydrate

-
-
1616831-12-9
N-(5-bromo-3-fluoro-2-methoxy-phenyl)-2-[(E)-hydroxyimino]-acetamide

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydroxylamine hydrochloride; sodium sulfate In 1,4-dioxane; water at 50 - 70℃; for 15h; | 96% |
-
-
302-17-0
chloral hydrate

-
-
151-10-0
1,3-Dimethoxybenzene

-
-
138002-56-9
1,1,1-trichloro-2,2-bis(2,4-dimethoxyphenyl)ethane

| Conditions | Yield |
|---|---|
| 95% |
Chloral hydrate Chemical Properties
IUPAC Name: 2,2,2-Trichloroethane-1,1-diol
Following is the structure of Chloral hydrate (CAS NO.302-17-0):
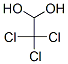
Molecular Formula: C2H3Cl3O2
Molecular Weight: 165.4
EINECS: 206-117-5
Storage temp.: 0-6 °C
Water Solubility: 660 g/100 mL
Index of Refraction: 1.545
Molar Refractivity: 28.85 cm3
Molar Volume: 91.1 cm3
Density: 1.814 g/cm3
Flash Point: 57.3 °C
Melting point: 57 °C(lit.)
Surface Tension: 59.3 dyne/cm
Enthalpy of Vaporization: 39.15 kJ/mol
Boiling Point: 96.3 °C at 760 mmHg
Vapour Pressure: 25.6 mmHg at 25 °C
Product Categories of Chloral hydrate (CAS NO.302-17-0): Miscellaneous; Organics; Method 551EPA; Method 8240; 500 Series Drinking Water Methods; 8000 Series Solidwaste Methods; EPA
Stability of Chloral hydrate (CAS NO.302-17-0): Stable, but may be air or light sensitive. Incompatible with alcohols, cyanides, iodine, strong bases, carbonates.
Canonical SMILES: C(C(Cl)(Cl)Cl)(O)O
InChI: InChI=1S/C2H3Cl3O2/c3-2(4,5)1(6)7/h1,6-7H
InChIKey: RNFNDJAIBTYOQL-UHFFFAOYSA-N
Chloral hydrate Uses
Chloral hydrate (CAS NO.302-17-0) is mainly used as pesticides,pharmaceutical intermediates, also used to produce Chloroform and chloral.
Chloral hydrate Toxicity Data With Reference
| 1. | mrc-smc 15 mmol/L | MUREAV Mutation Research. 141 (1984),19. | ||
| 2. | sce-hmn:lym 54 mg/L | AGTQAH Annales de Genetique. 24 (1981),105. | ||
| 3. | orl-mus TDLo:10 mg/kg:CAR | CDPRD4 Cancer Detection and Prevention. 9 (1986),279. | ||
| 4. | orl-wmn TDLo:465 mg/kg:EYE,BPR | AMSVAZ Acta Medica Scandinavica. 223 (1988),269. | ||
| 5. | orl-hmn LDLo:4 mg/kg | PHBTH* Pharmacology: Basis of Therapy. 3 (1967),132. | ||
| 6. | orl-hmn TDLo:300 mg/kg:CNS,CVS | BMJOAE British Medical Journal. 2 (1977),994. | ||
| 7. | unr-cld TDLo:48 mg/kg/6H-I:BPR AACRAT 65,691,86 | |||
| 8. | unk-man LDLo:103 mg/kg | 85DCAI Poisoning; Toxicology, Symptoms, Treatments. 2 (1970),73. | ||
| 9. | unr-inf TDLo:160 mg/kg/3D-I:CVS AACRAT 65,691,86 | |||
| 10. | rec-cld TDLo:97 mg/kg:CVS AACRAT 65,691,86 | |||
| 11. | orl-rat LD50:479 mg/kg | TXAPA9 Toxicology and Applied Pharmacology. 18 (1971),185. | ||
| 12. | skn-rat LD50:3030 mg/kg |
Chloral hydrate Consensus Reports
Reported in EPA TSCA Inventory. EPA Genetic Toxicology Program.
Chloral hydrate Safety Profile
A human poison by ingestion and possibly other routes. Poison experimentally by ingestion, intravenous, and rectal routes. Moderately toxic by subcutaneous, parenteral, and intraperitoneal routes. Experimental reproductive effects. Human systemic effects by ingestion: general anesthetic, cardiac arrhythmias, blood pressure depression, eye effects, coma, pulse rate increase, arrhythmias. Human mutation data reported. Questionable carcinogen with experimental carcinogenic and tumorigenic data by skin contact. A sedative, anesthetic, and narcotic. Combustible when exposed to heat or flame. When heated to decomposition it emits toxic fumes of Cl−.
Hazard Codes:  T
T F
F
Risk Statements: 25-36/38-39-23/24/25-11-36/37/38
R25 :Toxic if swallowed.
R36/38:Irritating to eyes and skin.
R39:Danger of very serious irreversible effects.
R23/24/25:Toxic by inhalation, in contact with skin and if swallowed.
R11:Highly flammable.
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-45-25-23-36/37-16-27
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S25:Avoid contact with eyes.
S23:Do not breathe vapour.
S36/37:Wear suitable protective clothing and gloves.
S16:Keep away from sources of ignition.
S27:Take off immediately all contaminated clothing.
RIDADR: UN 3286 3/PG 2
WGK Germany: 2
RTECS: FM8750000
HazardClass: 6.1(b)
PackingGroup: III
Chloral hydrate Specification
Chloral hydrate (CAS NO.302-17-0) is also named as ,1,1-trichloro-2,2-ethanediol; 2,2,2-Trichloro-1,1-ethanediol; aquachloral; bi 3411; chloraldurat; dormal; Escre; felsules; hydral; trichloroacetaldehyde hydrate; Trichloroacetaldehyde Monohydrate; TCA,and so on.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View