-
Name
2-METHYLGLUTARIC ACID
- EINECS 210-521-7
- CAS No. 18069-17-5
- Article Data101
- CAS DataBase
- Density 1.246 g/cm3
- Solubility
- Melting Point 80-82 °C(lit.)
- Formula C6H10O4
- Boiling Point 332.7 °C at 760 mmHg
- Molecular Weight 146.143
- Flash Point 148.8 °C
- Transport Information
- Appearance white to almost white fine crystalline powder
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2-Methylglutaric acid;2-Methylpentanedioic acid;
- PSA 74.60000
- LogP 0.57190
Synthetic route

| Conditions | Yield |
|---|---|
| With hexafluoroantimonic acid at 30℃; for 1h; | 100% |
-
-
4553-62-2
2-methylglutaronitrile

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid at 90℃; for 16h; Reagent/catalyst; Temperature; Inert atmosphere; | 96% |
| With hydrogenchloride | |
| acid hydrolysis; | |
| With sulfuric acid; water at 105 - 125℃; for 8.42h; Inert atmosphere; |
-
-
26093-63-0
3-methyltetrahydropyran

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With sodium bromate; potassium hydrogensulfate In water at 25 - 30℃; for 120h; Oxidation; | 87% |
-
-
500-05-0
2H-pyran-2-one-5-carboxylic acid

-
A
-
5807-38-5
6-oxo-tetrahydro-pyran-3-carboxylic acid

-
B
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With methanol; palladium on activated charcoal Hydrogenation; |
-
-
6018-41-3
methyl coumalate

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With methanol; platinum(IV) oxide Hydrogenation.Verseifung der NaHCO3-loeslichen Anteile des Reaktionsprodukts mit konz. HCl; |
-
-
74457-59-3
2,6-dimethyl-5-oxo-heptanoic acid

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| bei weiterer Oxidation; α-methyl-isobutyryl-butyric acid from dihydrocamphorphorone; |

| Conditions | Yield |
|---|---|
| durch Oxidation; substance from carvenone; |
-
-
67-66-3
chloroform

-
-
6784-16-3
5-methyl-2-isopropylidenecyclopentanone

-
A
-
18069-17-5
2-methylglutaric acid

-
B
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| bei der Ozonspaltung; |

-
-
54088-65-2
rac-2,6-dimethylhept-5-enenitrile

-
A
-
498-21-5, 636-60-2
2-methylbutanedioic acid

-
B
-
18069-17-5
2-methylglutaric acid

-
C
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| bei der Oxydation und nachfolgender Verseifung des Reaktionsproduktes; |

-
-
74650-83-2
methyl α-methyl-γ-carboethoxy-γ-acetylbutyrate

-
A
-
18069-17-5
2-methylglutaric acid

-
B
-
14203-46-4
4-methyl-cyclohexane-1,3-dione

| Conditions | Yield |
|---|---|
| With sodium methylate anschliessend Erwaermen mit wss. KOH; |
-
-
74650-83-2
methyl α-methyl-γ-carboethoxy-γ-acetylbutyrate

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With methanol; sodium methylate anschliessendes Erhitzen mit wss.Kalilauge; |
-
-
35299-16-2
cyano-2 methyl-4 glutarate de diethyle

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| mit verd. Mineralsaeuren; |
-
-
132467-55-1
1,1,2-triethyl 2-methylpropane-1,1,2-tricarboxylate

-
-
141-52-6
sodium ethanolate

-
A
-
18069-17-5
2-methylglutaric acid

-
B
-
597-43-3
2,2-dimethylsuccinic acid

| Conditions | Yield |
|---|---|
| at 100℃; im Rohr und, Kochen des Reaktionsprodukts mit 20 prozentiger Salzsaeure; |

-
-
23733-68-8
p-menth-3-en-2-one

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With potassium permanganate |
-
-
14203-46-4
4-methyl-cyclohexane-1,3-dione

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With air |
-
-
66984-18-7
(+/-)-1-methyl-2-oxo-1-cyclopentanecarbonitrile

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With nitric acid |
-
-
6784-16-3
5-methyl-2-isopropylidenecyclopentanone

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With potassium permanganate |

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide; dihydrogen peroxide |
-
-
996-82-7
sodium diethylmalonate

-
-
80-62-6
methacrylic acid methyl ester

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| anschliessende Hydrolyse und Decarboxylierung; |
-
-
141-52-6
sodium ethanolate

-
-
105-53-3
diethyl malonate

-
-
20609-71-6
3-bromo-2-methyl-propionic acid methyl ester

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| Verseifen des entstandenen Esters mit HCl; |

-
-
18852-51-2
sodium cyanoacetic acid ethyl ester

-
-
80-55-7
ethyl 2-hydroxy-2,2-dimethylethanoate

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With ethanol man behandelt das saure Reaktionsprodukt mit kalter konzentrierter Schwefelsaeure, verduennt mit Wasser und kocht; |
-
-
23886-52-4
4-cyanopentanoic acid

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide |
-
-
14035-94-0
dimethyl 2-methylglutarate

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| (hydrolysis); |
-
-
89898-62-4
butane-1,3,3-tricarboxylic acid

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| at 150℃; |
-
-
39555-01-6
diethyl 2-ethoxycarbonyl-2-methylglutarate

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
37810-29-0
2-methyl-4-cyanobutyric acid

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
292638-85-8
acrylic acid methyl ester

-
-
609-08-5
Diethyl methylmalonate

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| (i) , (ii) (decarboxylation); Multistep reaction; |

| Conditions | Yield |
|---|---|
| Yield given. Multistep reaction; |
-
-
1679-47-6
3-methyltetrahydro-2-furanone

-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| at 270℃; im geschlossenen Rohr anschliessend Behandeln mit Kalilauge; |
-
-
18069-17-5
2-methylglutaric acid

-
-
31468-33-4
2-methylglutaric anhydride

| Conditions | Yield |
|---|---|
| With acetic anhydride for 24h; Heating; | 100% |
| With acetyl chloride Heating; | 98.4% |
| With acetic anhydride at 140℃; for 7h; | 92.2% |
-
-
18069-17-5
2-methylglutaric acid

-
-
88150-42-9
2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-3-ethoxycarbonyl-5-methoxycarbonyl-6-methyl-1,4-dihydropyridine

| Conditions | Yield |
|---|---|
| In diethyl ether; tert-butyl alcohol at 5 - 20℃; for 4h; | 95% |
-
-
110-00-9
furan

-
-
18069-17-5
2-methylglutaric acid

-
-
4124-30-5
dichloroacetic anhydride

-
-
91061-95-9
methyl 4-furoylbutyrate

| Conditions | Yield |
|---|---|
| In toluene | 94.5% |

-
-
18069-17-5
2-methylglutaric acid

-
-
42856-62-2
2-methyl-1,5-pentanediol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0℃; for 16h; Reflux; | 93% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 0℃; for 16h; Inert atmosphere; Reflux; | 93% |
| With dimethylsulfide borane complex In tetrahydrofuran at 0 - 25℃; for 16.15h; Inert atmosphere; | 91% |
-
-
18069-17-5
2-methylglutaric acid

-
-
79-03-8
propionyl chloride

-
-
20990-14-1
2,4-dimethyl-1,3-cyclohexanedione

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylglutaric acid With aluminum (III) chloride; nitromethane at 20℃; for 1h; Inert atmosphere; Stage #2: propionyl chloride at 80℃; for 3h; Inert atmosphere; | 89% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In water at 60℃; for 3h; Temperature; Inert atmosphere; | 86.1% |

-
-
18069-17-5
2-methylglutaric acid

-
-
103129-82-4
amlodipine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 1h; | 84% |
-
-
18069-17-5
2-methylglutaric acid

-
-
29553-51-3
3-methyl-piperidine-2,6-dione

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylglutaric acid With acetic anhydride at 20 - 60℃; for 8h; Stage #2: With ammonia In tetrahydrofuran; water at 0 - 20℃; for 8h; Stage #3: With acetic anhydride at 60℃; for 8h; | 78% |
| With acetyl chloride Behandeln des erhaltenen Saeureanhydrids mit Ammoniak bei 120grad und Erhitzen des Reaktionsprodukts auf 200grad; | |
| With ammonia 1.) 150 deg C, 1.5 h, 2.) 200 deg C, 0.5 h; Yield given. Multistep reaction; | |
| Multi-step reaction with 2 steps 1: Ac2O 2: NH4OH View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylglutaric acid With aluminum (III) chloride; nitromethane In 1,2-dichloro-ethane at 20℃; for 1h; Inert atmosphere; Stage #2: butyryl chloride In 1,2-dichloro-ethane at 80℃; for 3h; Inert atmosphere; | 73% |
-
-
18069-17-5
2-methylglutaric acid

-
-
124-40-3
dimethyl amine

-
-
1033820-68-6
N,N,N',N'-tetramethyl-2-methylglutaramide

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylglutaric acid With thionyl chloride at 60℃; for 5h; Stage #2: dimethyl amine With triethylamine In toluene at 0 - 20℃; | 67% |
-
-
18069-17-5
2-methylglutaric acid

-
-
109-89-7
diethylamine

-
-
100071-13-4
N,N,N',N'-tetraethyl-2-methylglutaramide

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylglutaric acid With thionyl chloride at 60℃; for 5h; Stage #2: diethylamine With triethylamine In toluene at 0 - 20℃; | 66% |

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 20℃; | 65% |

-
-
18069-17-5
2-methylglutaric acid

-
-
75-36-5
acetyl chloride

-
-
134746-37-5
2-acetyl-4-methylcyclohexane-1,3-dione

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylglutaric acid With aluminum (III) chloride In nitromethane at 20℃; for 1h; Inert atmosphere; Stage #2: acetyl chloride In nitromethane at 80℃; for 2h; Inert atmosphere; | 65% |

| Conditions | Yield |
|---|---|
| With urea In ethanol at 20℃; for 168h; | 60% |

-
-
18069-17-5
2-methylglutaric acid

-
-
2528-61-2
Heptanoic acid chloride

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylglutaric acid With aluminum (III) chloride; nitromethane at 20℃; for 1h; Inert atmosphere; Stage #2: Heptanoic acid chloride at 80℃; for 3h; Inert atmosphere; | 57% |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride for 0.0833333h; microwave irradiation; | 34% |

| Conditions | Yield |
|---|---|
| With sulfuric acid | |
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide |
-
-
18069-17-5
2-methylglutaric acid
-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| With thionyl chloride; chlorine Eingiessen des Reaktionsprodukts in Methanol; |
-
-
18069-17-5
2-methylglutaric acid

-
-
474327-99-6
2-bromo-4-methyl-glutaric acid dimethyl ester

| Conditions | Yield |
|---|---|
| With thionyl chloride man setzt bei 40grad Brom hinzu, erwaermt die Reaktions-Loesung dann auf 60grad und giesst das Reaktionsprodukt in Methanol; |
-
-
18069-17-5
2-methylglutaric acid

| Conditions | Yield |
|---|---|
| /BRN= 1783854/; |
-
-
18069-17-5
2-methylglutaric acid

-
-
10171-93-4
N,N'-diphenyl-2-methylpentane-1,5-dicarboxamide
-
-
18069-17-5
2-methylglutaric acid
Pentanedioic acid,2-methyl- Specification
The Pentanedioic acid,2-methyl-, with the CAS registry number 18069-17-5, is also known as alpha-Methylglutaric acid. It belongs to the product categories of Miscellaneous Biochemicals; C6; Carbonyl Compounds; Carboxylic Acids. Its EINECS registry number is 210-521-7. This chemical's molecular formula is C6H10O4 and molecular weight is 146.1412. Its IUPAC name is called 2-methylpentanedioic acid. What's more. this chemical is white to almost white fine crystalline powder.
Physical properties of Pentanedioic acid,2-methyl-: (1)ACD/LogP: -0.69; (2)ACD/LogD (pH 5.5): -2.35; (3)ACD/LogD (pH 7.4): -5.31; (4)ACD/BCF (pH 5.5): 1; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 1; (7)ACD/KOC (pH 7.4): 1; (8)#H bond acceptors: 4; (9)#H bond donors: 2; (10)#Freely Rotating Bonds: 4; (11)Index of Refraction: 1.473; (12)Molar Refractivity: 32.93 cm3; (13)Molar Volume: 117.2 cm3; (14)Surface Tension: 50.1 dyne/cm; (15)Density: 1.246 g/cm3; (16)Flash Point: 148.8 °C; (17)Enthalpy of Vaporization: 63.26 kJ/mol; (18)Boiling Point: 332.7 °C at 760 mmHg; (19)Vapour Pressure: 2.76E-05 mmHg at 25°C.
Preparation of Pentanedioic acid,2-methyl-: this chemical can be prepared by 3-Methyl-tetrahydro-pyran. This reaction is a kind of Oxidation. It will need reagents NaBrO3, KHSO4 and solvent H2O. The reaction time is 120 hours with reaction temperature of 25 - 30 °C. The yield is about 87%.
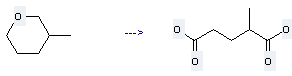
Uses of Pentanedioic acid,2-methyl-: it can be used to produce 2-methyl-pentanedioic acid anhydride. This reaction will need reagent acetyl chloride.

When you are using this chemical, please be cautious about it as the following:
This chemical may cause inflammation to the skin or other mucous membranes. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. Whenever you will contact it, please wear suitable protective clothing.
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: CC(CCC(=O)O)C(=O)O
(2)InChI: InChI=1S/C6H10O4/c1-4(6(9)10)2-3-5(7)8/h4H,2-3H2,1H3,(H,7,8)(H,9,10)
(3)InChIKey: AQYCMVICBNBXNA-UHFFFAOYSA-N
Related Products
- Pentanedioic acid, 2-bromo-, diethyl ester
- Pentanedioic acid, 2-hydroxy-, sodium salt (1:2)
- Pentanedioic acid, 2-methyl-, (2S)-
- Pentanedioic acid, 3-[[(1,1-dimethylethyl)dimethylsilyl]oxy]-, 1-(1,1-dimethylethyl) 5-ethyl ester,(3R)-
- Pentanedioic acid, 3-oxo-, monomethyl ester
- Pentanedioic acid,1,5-bis(2-methylpropyl) ester
- Pentanedioic acid,1,5-didecyl ester
- Pentanedioic acid,1-ethyl ester
- Pentanedioic acid,2,2,3,3,4,4-hexafluoro-
- Pentanedioic acid,2,2,4-trimethyl-4-phosphono-
- 180694-97-7
- 180695-79-8
- 180698-14-0
- 180698-18-4
- 180698-19-5
- 180729-93-5
- 18073-32-0
- 180748-30-5
- 18075-40-6
- 18075-64-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View