 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
Synthetic route

| Conditions | Yield |
|---|---|
| In not given evapn. of filtrated soln.; |

| Conditions | Yield |
|---|---|
| In water heating;; | |
| In water heating;; |


| Conditions | Yield |
|---|---|
| In not given evapn.of filtrated soln.; |

| Conditions | Yield |
|---|---|
| In further solvent(s) Electrolysis; in 5-20 w.-% NaOH soln.; | |
| In not given Electrolysis; in dild. NaOH soln.; |

| Conditions | Yield |
|---|---|
| In not given Electrolysis; |

-
-
144-55-8
sodium hydrogencarbonate

-
B
-
10101-50-5
sodium permanganate

| Conditions | Yield |
|---|---|
| In water byproducts: Na2CO3; stirring (1 h), refluxing (30 min); filtration, crystn.; IR spectroccopy, X-ray diffractometry; |

-
-
7681-52-9
sodium hypochlorite

-
-
10101-50-5
sodium permanganate

| Conditions | Yield |
|---|---|
| With Mn(II)-silicate In water alk. soln.;; | |
| With Mn(II)-silicate In water alk. soln.;; |

| Conditions | Yield |
|---|---|
| In water Kinetics; Na2MnO4*10H2O was reacted with aq. OH(1-) at 0; 25 and 45°C; reaction was study at 45°C with varied pressure (0-180 MPa); |

-
-
10101-50-5
sodium permanganate

| Conditions | Yield |
|---|---|
| dehydratation at 160°C or quicker at 180°C; | |
| dehydratation at 70°C in vac. over P2O5; | |
| dehydratation at 160°C or quicker at 180°C; | |
| dehydratation at 70°C in vac. over P2O5; | |
| dehydratation in vac. at 70-80°C; |

| Conditions | Yield |
|---|---|
| In water |

| Conditions | Yield |
|---|---|
| Stage #1: manganese(IV) oxide; boric acid; magnesium oxide; sodium silicate solution of Ludox 30 With lithium hydroxide In water at 110℃; for 12h; Stage #2: at 900℃; for 12h; Calcination; |

-
-
10101-50-5
sodium permanganate

-
-
7664-41-7
ammonia

| Conditions | Yield |
|---|---|
| In ammonia aq. NH3; Cd sulfate dissolved in aq. NH3, cooled to 5°C, then aq. NaMnO4 cooled to 5°C added; cooled to 0°C; | 86% |


| Conditions | Yield |
|---|---|
| In water at 159.84℃; for 12h; Autoclave; High pressure; | 83% |

| Conditions | Yield |
|---|---|
| In water | 63% |
| In water |
-
-
10101-50-5
sodium permanganate

-
-
141-82-2
malonic acid

| Conditions | Yield |
|---|---|
| With CH3OH In acetonitrile to a soln. of malonic acid in actonitrile was added NaMnO4 under N2, stirred for 20-30 min, methanol was added; evapn.; elem. anal.; | 38% |
-
-
10101-50-5
sodium permanganate

-
-
7463-35-6
N-(3-chloro-2-methylphenyl)acetamide

-
-
19407-42-2
2-(acetylamino)-6-chlorobenzoic acid

| Conditions | Yield |
|---|---|
| In water |

-
-
10101-50-5
sodium permanganate

-
-
7664-93-9
sulfuric acid

-
-
551-16-6, 1079-37-4, 3115-55-7, 21794-93-4, 145920-51-0
6-aminopenicillanic acid

-
-
36091-15-3
(5R,6R)-6-amino-2,2-dimethyl-1,1-dioxopenam-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With phosphoric acid; ammonia In water; acetonitrile |


| Conditions | Yield |
|---|---|
| In not given first treating of NaMnO4 soln. with chlorine or air then pptg. with KCl; |
-
-
10101-50-5
sodium permanganate

-
-
1313-13-9
manganese(IV) oxide

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In not given dropwise addn. of H2O2 at 70 °C to NaMnO4-soln. (contg. NaOH or KOH);; MnO1.99-MnO2;; | |
| With hydrogenchloride In hydrogenchloride redn. of NaMnO4 with HCl;; drying at 120 °C in O2-atmosphere yields Na0.22MnO1.97;; | |
| In neat (no solvent) thermal decompn.;; | |
| With HCl In hydrogenchloride redn. of NaMnO4 with HCl;; drying at 120 °C in O2-atmosphere yields Na0.22MnO1.97;; |

| Conditions | Yield |
|---|---|
| In not given excess of soln. of NaMnO4 used; | |
| In not given excess of soln. of NaMnO4 used; |

| Conditions | Yield |
|---|---|
| With ethanol In not given redn. of a soln. of sodium paramolybdate and NaMnO4 by alcohol; addn. of KCl;; | |
| With alcohol In not given redn. of a soln. of sodium paramolybdate and NaMnO4 by alcohol; addn. of KCl;; |

-
-
10101-50-5
sodium permanganate

| Conditions | Yield |
|---|---|
| With NaOH; H2O dissolved 20g of Na2{Fe(CN)5NO}*2H2O, 10g of NaMnO4, 2.5g of NaOH in 100cm^3 water; stayed (often shaked); added 30 cm^3 of ethanol; filtered; mixed with acetic acid, then with alcohol-ether; washed 5 times in water, pptd. with alcohol; |

| Conditions | Yield |
|---|---|
| In sodium hydroxide byproducts: H2O; aq. NaOH; in 30-40 % NaOH soln., heating, mixing; | |
| In sodium hydroxide byproducts: H2O; in 30-40 % NaOH soln., heating, mixing; |


| Conditions | Yield |
|---|---|
| excess of NaOH used, at 125°C; | |
| In neat (no solvent) N2 atmosphere; molar ratio Na-compd. : Mn-compd. = 1 : 1, heating (150°C, 10 h); detd. by X-ray powder diffraction and IR-spectroscopy; | |
| In not given | |
| excess of NaOH used, at 125°C; |
-
-
10101-50-5
sodium permanganate

-
-
44584-78-3
bismuth oxide perchlorate

| Conditions | Yield |
|---|---|
| In not given in soln. of perchloric acid; |
-
-
10101-50-5
sodium permanganate

-
-
44584-78-3
bismuth oxide perchlorate

| Conditions | Yield |
|---|---|
| In not given in soln. of perchloric acid; |
-
-
10101-50-5
sodium permanganate

-
-
44584-78-3
bismuth oxide perchlorate

| Conditions | Yield |
|---|---|
| In not given in soln. of perchloric acid; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: O2; heating to 120 - 180°C; reaction followed by X-ray powder diffraction and IR-spectroscopy; | |
| byproducts: O2; thermal decompn. at 330°C; |
-
-
10101-50-5
sodium permanganate

-
-
14265-44-2
Phosphate

| Conditions | Yield |
|---|---|
| With phosphorous In water byproducts: hypophosphate, triphosphate, tetraphosphate; phosphorus dissolved in CS2; further byproduct: MnO2-hydrate; detn. of products by paper chromatography; | |
| With white phosphorus In water byproducts: hypophosphate, triphosphate, tetraphosphate; phosphorus dissolved in CS2; further byproduct: MnO2-hydrate; detn. of products by paper chromatography; |
-
-
10101-50-5
sodium permanganate

| Conditions | Yield |
|---|---|
| With LiI In not given redox reaction between NaMnO4 and LiI in 6-fold excess at room temp. according to A. Ibarra-Palos et al., Solid State Ionics 138 (2001) 203-212 and A. Ibarra-Palos, et al., J. Power Sources 146 (2005)294-299; washed with distd. H2O, filtered, dried under vac. at >=150 °C; small contents of I (2-4%); |

-
-
10101-50-5
sodium permanganate

| Conditions | Yield |
|---|---|
| In neat (no solvent) formation of a diadochous compound between BaSO4 and NaMnO4;; | |
| In neat (no solvent) formation of a diadochous compound between BaSO4 and NaMnO4;; | |
| In not given satd soln. used; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating in air (800°C, 8 h); |

| Conditions | Yield |
|---|---|
| excess of NaOH used, at 125°C; recrystn from heavy basic soln.; | |
| excess of NaOH used, at 125°C; recrystn from heavy basic soln.; |

| Conditions | Yield |
|---|---|
| excess of NaOH used, at 125°C, cooling in mixture of ice and NaCl; | |
| excess of NaOH used, at 125°C, cooling in mixture of ice and NaCl; |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View


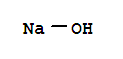




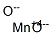

 O,
O,  C
C