Jinan Finer Chemical Co., Ltd
Product Description Product name Sulfolane CAS 7681-52-9 Assay 13% 10% 5% Appearance Light Yellow Liqui
Cas:7681-52-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryPUYANG WANQUAN CHEMICAL CO.,LTD.
good factory price and good quality Main uses: used for water purification, as well as for disinfectant, pulp bleaching, pharmaceutical industry to make chloramines and so on. Appearance:Micro-yellow solution Storage:dry condition Package:25kg/
Cas:7681-52-9
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:7681-52-9
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Hebei yanxi chemical co.,LTD.
1) Solid sodium chlorite is white or light yellow green, crystalline, alkaline, lightly absorptve humidity and easily solvable in water and alcohol. 2) The product is an oxidizng agent with high efficiency and an excellent bleaching ingredent, it
Cas:7681-52-9
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
CAS:7681-52-9 EINECS No.:231-668-3 Molecular formula: NaClO Main Active Ingredients and Content:This product is a bleach disinfectant with sodium hypochlorite as the main active ingredient. The content of effective Appearance: Colorless and cl
Cas:7681-52-9
Min.Order:5 Metric Ton
FOB Price: $1.0 / 2.0
Type:Other
inquiryHebei Mojin Biotechnology Co.,Ltd
Hebei Mojin Biotechnology Co., Ltd, Our company is a professional in lead acetate, diphenyl ethylamine and other chemical raw materials and chemical reagents research and development production enterprises. Our business covers more than 30 countrie
Cas:7681-52-9
Min.Order:1 Kilogram
FOB Price: $150.0
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Triumph International Development Limilted
Triumph has the complete production of G- KG - MT service chain,we can make the new technology into productivity quickly in the research and development of new products. Main Service 1.Own made fine chemical products 2.Out sourcing and quality c
Cas:7681-52-9
Min.Order:100 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:7681-52-9
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:7681-52-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Product Name: Sodium hypochlorite Synonyms: TEEPOL BLEACH;SODIUM HYPOCHLORITE;Sodium hypochlorite solution;SODA BLEACHING LYE;b-kliquid;carrel-dakinsolution;caswellno776;chloros CAS: 7681-52-9 MF: ClNaO MW: 74.44 EINECS: 231-668-3 Product Cat
Cas:7681-52-9
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:7681-52-9
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: Sodium Hypochloride CAS No.:7681-52-9 Molecule Formula:NaClO Molecule Weight:68.46 Purity: 20% Package: 25kg/drum Description:Colorless liquid with strong odor Manufacture Standards:Enterprise Standard
Cas:7681-52-9
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Nanjing Yuance Industry&Trade Co., Ltd.
Product name: 2, 3-Dihydroxy naphthalene MF: C10H8O2 Appearance: Gray-white or slight red powder Purity: 99% min Packing: 25 kgs or 50 kgs carton
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Cas:7681-52-9
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:7681-52-9
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:7681-52-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHubei Langyou International Trading Co., Ltd
Sodium hypochlorite CAS.7681-52-9 Application:Sodium hypochlorite CAS.7681-52-9
Hunan Longxianng Runhui Trading Co.,Ltd
Sodium hypochlorite CAS.7681-52-9Appearance:ask jude for details Storage:Dry and ventilated Package:in foil bag or customized Application:Pharmaceutical intermediates/Food additive Transportation:By express (Door to door) such as FEDEX, DHL, EMS for
Cas:7681-52-9
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Beantown Chemical
in stock Application:7681-52-9
Cas:7681-52-9
Min.Order:0
Negotiable
Type:Trading Company
inquirySynthetic route

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sulfuric acid In water ice cooling; distn. with H2SO4; | A n/a B n/a C 9% |
| With NaOH; H2SO4 In water ice cooling; distn. with H2SO4; | A n/a B n/a C 9% |

-
-
3811-04-9
potassium chlorate

-
-
7647-14-5
sodium chloride

-
A
-
7681-52-9
sodium hypochlorite

-
B
-
80937-33-3
oxygen

-
C
-
14866-68-3, 80434-34-0
chlorate

| Conditions | Yield |
|---|---|
| With Na2Cr2O7 In water Electrolysis; influencce of electrode material;; |


| Conditions | Yield |
|---|---|
| In water Electrolysis; |


| Conditions | Yield |
|---|---|
| In water Electrolysis; | |
| In water Electrolysis; chlorate formation as side reaction;; |


| Conditions | Yield |
|---|---|
| In water Electrolysis; | |
| In water Electrolysis; Schuckert-process, monopolar arrangement, vessel of enamelled iron, cooled by water; carbon electrodes or carbon electrodes covered with Pt, electrodes used as stirrer;; | |
| With CrO4(1-) In water byproducts: H, chromium chromate; Electrolysis; electrolysis without diaphragm, cathode reaction; additive alkalichromates, formation of chromium chromate, which act as diaphragm;; |

| Conditions | Yield |
|---|---|
| With chlorine In not given Electrolysis; NaOH formed at cathode and chlorine formed at anode; gas proofed anode region, asbestos diaphragm; mixing of NaOH and chlorine in a special region;; | |
| With chlorine In not given Electrolysis; NaOH formed at cathode and chlorine formed at anode; anode: coal plates or rods with coke pieces; passing catholyte into anode region;; | |
| With chlorine In not given Electrolysis; NaOH formed at cathode and chlorine formed at anode; cell of ungazed tone plates filled with pieces of C; alternative connection to positive and negative pole;; |
-
-
7782-50-5
chlorine

-
-
1310-73-2
sodium hydroxide

-
A
-
7681-52-9
sodium hypochlorite

-
B
-
7647-14-5
sodium chloride

| Conditions | Yield |
|---|---|
| In neat (no solvent) 170kg NaOH, 63kg Cl2, 60°C, cyclindric rotating vessel, 1.5h;; product-mixture:24% NaClO, 25.5% NaCl, 43% NaOH;; | |
| In neat (no solvent) 170kg NaOH, 63kg Cl2, 60°C, cyclindric rotating vessel, 1.5h, sintering of product to increase stability;; product-mixture:24% NaClO, 25.5% NaCl, 43% NaOH;; | |
| In not given byproducts: H2O; react. is reversible;; | |
| In neat (no solvent) reaction of finely atomized alkaline hydroxide soln. with Cl2;; mixture;; | |
| In water |


| Conditions | Yield |
|---|---|
| In sodium hydroxide Cl2 passed through carbonate-free soln. of NaOH at -5...0°C; | |
| In water sparging chlorine into 0.3 M aq. soln. of NaOH; UV monitoring; | |
| In water residual alkalinity: 1g/l; not isolated;; |

| Conditions | Yield |
|---|---|
| With Na-phosphates In neat (no solvent) | |
| With NaHSO4 or NaH2PO4 or NaO2CCO2H In water other solvents possible; addn. of the solvent to the mixture of the solid components just before use;; | |
| With NaHSO4 or NaH2PO4 or NaO2CCO2H In water | |
| With Na-phosphates In neat (no solvent) | |
| With NaHSO4 or NaH2PO4 or NaO2CCO2H In water other solvents possible; addn. of the solvent to the mixture of the solid components just before use;; |

| Conditions | Yield |
|---|---|
| In water react. at ion exchange resin;; solns. with 1-3% active chlorine;; | |
| In water react. at ion exchange resin;; solns. with 1-3% active chlorine;; |

| Conditions | Yield |
|---|---|
| With boric acid In water boric acid+Ca(ClO)2 in water distillation, first 100 ml distillate colected in 200 ml containing NaOH; |
-
-
7778-54-3
calcium hypochlorite

-
-
497-19-8
sodium carbonate

-
A
-
7681-52-9
sodium hypochlorite

-
B
-
1310-73-2
sodium hydroxide

| Conditions | Yield |
|---|---|
| In water byproducts: CaCO3; dissolving mixture of dry Ca(OCl)2, containing Ca(OH)2, and Na2CO3 in H2O;; pptn. of CaCO3; formation of a NaOCl and NaOH soln.;; |


| Conditions | Yield |
|---|---|
| With (borax or MgCO3) In water byproducts: CaCO3; hot Na2CO3-soln. which may contain borax or MgCO3;; not isolated;; | |
| In neat (no solvent) | |
| With (borax or MgCO3) In water byproducts: CaCO3; hot Na2CO3-soln. which may contain borax or MgCO3;; not isolated;; | |
| With sodium hydrogencarbonate In water pH=9.5; | |
| In neat (no solvent) |
-
-
7778-54-3
calcium hypochlorite

-
-
15944-13-5
Chlorkalk

-
-
497-19-8
sodium carbonate

-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| In water byproducts: CaCO3; not isolated;; | |
| In water byproducts: CaCO3; not isolated;; |


| Conditions | Yield |
|---|---|
| With (borax or MgCO3) In water byproducts: CaCO3; hot NaHCO3-soln. which may contain borax or MgCO3;; not isolated;; | |
| With (borax or MgCO3) In water byproducts: CaCO3; hot NaHCO3-soln. which may contain borax or MgCO3;; not isolated;; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium dichromate; sodium chromate(VI) In water Kinetics; Electrolysis; in a glass cylindrical cell without a diaphragm; constant pH maintainedwithin 0.05-0.1 by buffering (Na2CrO4 and Na2Cr2O7 with addn. of NaOH or HCl); expts. conducted at c.d. 10 A/l by periodic method or with continuous introduction of NaCl feed soln.; not isolated, detected by polarization curves; gas anal.; |


| Conditions | Yield |
|---|---|
| In water Electrolysis; electrolysis of sea-water, graphite-electrodes;; not isolated;; | |
| In water Electrolysis; 65-100g NaCl/l, 15°C;; | |
| In water Electrolysis; apparatus described;; not isolated;; |

| Conditions | Yield |
|---|---|
| In water byproducts: NaHCO3; Na2CO3 suspended in satd. soln., filtration of NaHCO3;; solns. with 5-12% active chlorine; not isolated;; | |
| In water 100g Na2CO3/l;; <20g active chlorine per l; not isolated;; | |
| In water 100g Na2CO3/l;; <20g active chlorine per l; not isolated;; | |
| In water byproducts: NaHCO3; Na2CO3 suspended in satd. soln., filtration of NaHCO3;; solns. with 5-12% active chlorine; not isolated;; |


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In water; toluene satn. of toluene with a ClO2/N2-mixture (ratio 20:80) at 0°C, neutralization with NaOH, introduction of ClO2/N2, addn. of 30% NaOH and 15-20% H2O2, separation of layers, evapn. of lower layer;; 90% NaClO;; |

-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| With chloroamine In water react. with a chloroamine sol. in the aq. soln. of Na-salt;; chloride-free soln.;; | |
| With chloroamine In water react. with a chloroamine sol. in the aq. soln. of Na-salt;; chloride-free soln.;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) react. of solid NaOH with Cl2O in mixture with inert gases, temp. up to 100°C;; | |
| In tetrachloromethane dissolving Cl2O in CCl4, react. with NaOH at 30-50°C;; |
-
-
7791-21-1
hypochlorous anhydride

-
-
7782-50-5
chlorine

-
-
1310-73-2
sodium hydroxide

-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| In tetrachloromethane dissolving Cl2O and Cl2 in CCl4, react. with NaOH at 30-50°C;; |


| Conditions | Yield |
|---|---|
| In water byproducts: H, Ca(OH)2; Electrolysis; electrolysis without diaphragm, cathode reaction; additive CaCl2; formation of acidic soln. and therefore formation of ClO3 because of pptn. of Ca(OH)2;; | |
| In water byproducts: H, Ca(OH)2; Electrolysis; electrolysis without diaphragm, cathode reaction; additive CaCl2; formation of acidic soln. and therefore formation of ClO3 because of pptn. of Ca(OH)2;; |

| Conditions | Yield |
|---|---|
| In water Electrolysis; cell with diaphragm, anode compartment filled with satd. NaHCO3 soln. (not soda), cathode compartment satd. NaCl soln., low temp.;; soln. free of chloride but not stable at slightly elevated temp.;; |
-
-
13973-88-1
chlorine azide

-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| With sodium hydroxide In not given byproducts: NaN3, H2O; | |
| With NaOH In not given byproducts: NaN3, H2O; |

| Conditions | Yield |
|---|---|
| In not given byproducts: H2O; | |
| In not given byproducts: H2O; passing N3Cl into NaOH soln.;; |

-
-
17341-25-2
sodium cation

-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| With chloride lime In not given reaction of chloride lime with sodium salts;; |

| Conditions | Yield |
|---|---|
| In water from strongly alk. soln. by evapn. at low temp.;; solid NaClO obtained;; |

| Conditions | Yield |
|---|---|
| In not given passing Cl into NaOH lye at temp. below 27°C; stirring; pptd. NaCl is removed; addn. of solid NaOH;; soln. of chloride chlorine and hypochlorite chlorine;; | |
| With sodium carbonate In not given passing Cl into Na2CO3 soln.; formation of HClO and NaCl; reaction of HClO with acetanilide or p-nitroacetanilide to a chlorinated base, which decomposes with NaOH in NaOCl and the anilides;; | |
| In not given reaction of Cl with concd. soln. of NaOH; concn. of NaOH soln. may be equivalent to NaOCl soln. or dild. NaOH soln. and addn. of NaOH to form concd. NaOCl soln.;; separation of pptd. NaCl from reaction vessel; concd. NaOCl soln. containing small amounts of chloride;; | |
| With sodium carbonate In not given passing Cl into Na2CO3 soln.; formation of HClO and NaCl; reaction of HClO with acetanilide or p-nitroacetanilide to a chlorinated base, which decomposes with NaOH in NaOCl and the anilides;; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) reaction of chlorine with powdered NaOH at 0°C;; | |
| With air In not given passing Cl (dild. with 4-9 Vol. air) into NaOH lye to decrease formation of NaClO3;; |


| Conditions | Yield |
|---|---|
| In water pH=9.8 - 13.5; Product distribution / selectivity; | 100% |
| With sodium hydroxide; urea In water pH=6.5 - 13.4; Product distribution / selectivity; | 1.5% |
| With urea In water pH=5.6; Product distribution / selectivity; |

| Conditions | Yield |
|---|---|
| In water at -11 - -8℃; pH=~ 10; | 100% |
| In water at -15 - -7℃; pH=~ 10; Product distribution / selectivity; | 100% |
| In diethyl ether; water at -20 - -10℃; for 0.5h; |


| Conditions | Yield |
|---|---|
| copper(II) sulfate In not given 80-90°C, 20 min; not isolated; UV/VIS spectrometry; | 98.5% |
| cobalt(II) nitrate In not given 80-90°C, 20 min; not isolated; | 73% |
| silver nitrate In not given 80-90°C, 20 min; not isolated; | 72% |
| iron(III) sulfate In not given 80-90°C, 20 min; not isolated; | 65% |

-
-
7681-52-9
sodium hypochlorite

-
-
40365-16-0
n-C3H7SO2CH3

-
-
40365-20-6
heptafluoro-n-propyl(trichloromethyl)sulfone

| Conditions | Yield |
|---|---|
| In water 20°C, 72 h; | 98% |
| In water 20°C, 72 h; | 98% |
-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| In water react. of 23ml NaClO-soln. (0.294g Cl per ml soln.) with 12g Na3(PO2NH)3 within 3min (stirring, ice-cooling), dropwise addn. of 18ml aq. acetic acid (0.39g acid per ml soln.) at 12-18°C within 45min, addn. of an excess ethanol, pptn.;; extractn. with glacial acetic acid, washing with ethanol;; | 97% |
| In water react. of 23ml NaClO-soln. (0.294g Cl per ml soln.) with 12g Na3(PO2NH)3 within 3min (stirring, ice-cooling), dropwise addn. of 18ml aq. acetic acid (0.39g acid per ml soln.) at 12-18°C within 45min, addn. of an excess ethanol, pptn.;; extractn. with glacial acetic acid, washing with ethanol;; | 97% |
-
-
74115-13-2
5-bromopyridine-3-ol

-
-
7681-52-9
sodium hypochlorite

-
-
286946-77-8
5-bromo-2-chloro-3-hydroxypyridine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water | 96% |
-
-
7647-01-0
hydrogenchloride

-
-
7681-52-9
sodium hypochlorite

-
-
12305-41-8
tetramethylammonium 3,3’-commo-bis(undecahydro-1,2-dicarba-3-cobalta-closo-undecaborate)(1-)

| Conditions | Yield |
|---|---|
| In water addn. of excess 10% aq. NaOCl and 6 M HCl to Co-complex, stirring (25°C, 24 h); MeCN addn., shaking, sepn. of org. layer, evapn; | 85% |

-
-
7681-52-9
sodium hypochlorite

-
-
40365-39-7
1,1,1,2,3,3,3-heptafluoro-2-methanesulfinyl-propane

-
-
40365-20-6
heptafluoro-n-propyl(trichloromethyl)sulfone

| Conditions | Yield |
|---|---|
| 82% | |
| 82% |
-
-
7681-52-9
sodium hypochlorite

-
-
40365-18-2
Dodecafluor-5-trifluormethylhexyl-methyl-sulfon

-
-
40365-22-8
5-trifluoromethyldodecafluorohexyl(trichloromethyl)sulfone

| Conditions | Yield |
|---|---|
| In water 20°C, 36 h; | 81% |
| In water 20°C, 36 h; | 81% |

| Conditions | Yield |
|---|---|
| In not given in very weakly acidic soln.; | A 20% B 80% |
| In not given in very weakly acidic soln.; | A 20% B 80% |

-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| In water stirred for 24 h; collected, washed with water; elem. anal.; | 80% |
-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| In water stirred for 2-3 h at room temp.; filtered off, washed (cold water), dried (vac.); elem. anal.; | 80% |

-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| In water stirred for 2-3 h at room temp.; filtered off, washed (cold water), dried (vac.); elem. anal.; | 80% |

-
-
7681-52-9
sodium hypochlorite

| Conditions | Yield |
|---|---|
| With NaOH In sodium hydroxide aq. NaOH; stirring (room temp., 2 - 3 h, pH.apprx.10); filtn., washing (cold water), drying (vac.); | 80% |
-
-
7681-52-9
sodium hypochlorite

-
-
1365832-59-2
[(99)TcO(ethylene glycol)(N-benzyl-2-(1,4,7-triazononan-1-yl)acetamide)]Br

-
-
1365832-77-4
[(99)TcO3(N-benzyl-2-(1,4,7-triazononan-1-yl)acetamide)]Br*H2O

| Conditions | Yield |
|---|---|
| With HCl In water according to Braband H., et al., Chem.-Eur. J., 2009, 15, 633; (N2); acidified soln. of NaOCl was added to aq. soln. of Tc compd.; reacted for 30 min; slowly evapd.; filtered; washed (satd. KBr); elem. anal.; | 80% |

| Conditions | Yield |
|---|---|
| In water stirred for 30 min; slowly evapd.; elem. anal.; | 78% |


| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 1h; Inert atmosphere; | 78% |
-
-
7681-52-9
sodium hypochlorite

-
-
150736-72-4
(S)-tert-butyl 1-hydroxybutan-2-ylcarbamate

-
-
153371-25-6
(2S)-2-(tert-butoxycarbonyl)aminobutyraldehyde

| Conditions | Yield |
|---|---|
| Stage #1: (S)-tert-butyl 1-hydroxybutan-2-ylcarbamate With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; sodium hydrogencarbonate; sodium bromide In dichloromethane; water at 0 - 20℃; Stage #2: sodium hypochlorite In dichloromethane; water at 0℃; for 1.16667h; | 77% |
-
-
7681-52-9
sodium hypochlorite

-
-
13580-38-6
3-(prop-2-yn-1-yloxy)propan-1,2-diol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; Ki; aqueous H2SO4 In water | 76% |
| With sodium hydroxide; Ki; aqueous H2SO4 In water | 76% |

| Conditions | Yield |
|---|---|
| In not given Fe-complex was reacted with NaOCl at 50°C for 5 h; | 75% |
-
-
7681-52-9
sodium hypochlorite

-
-
40365-17-1
1,1,1,2,3,3,3-heptafluoro-2-methanesulfonyl-propane

-
-
40365-21-7
heptafluoroisopropyl(trichloromethyl)sulfone

| Conditions | Yield |
|---|---|
| In water 20°C, 36 h; | 75% |
| In water 20°C, 36 h; | 75% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View



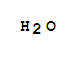


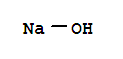
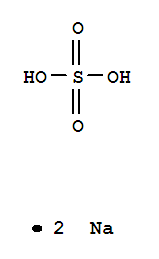






 C,
C, Xi
Xi